Project > AND Gate 1 > Design
Mechanism
Many AND Gates have been designed and constructed recently. In our project, We adopted the design of an existing AND gate(Anderson et al. Molecular Systems Biology) and improved its efficiency
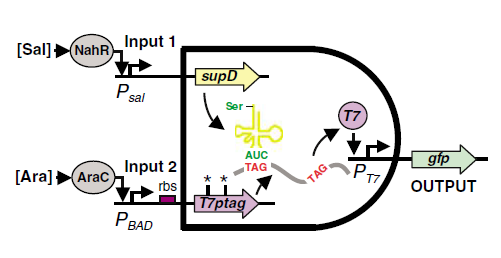 fig1. The mechanism of the AND Gate Anderson, Christopher JC Voigt CA and Arkin AP. Environmental signal integration by a modular AND gate. 2007, Molecular Systems Biology 3:133Fig.1 illustrates the mechanism of the design. One of the inputs of the AND gate leads to the expression of T7ptag mRNA which is a T7 polymerase coding sequence with amber mutations inside. The other input of the AND gate leads to the expression of supD tRNA, which acts as an amber mutation suppressor. The concurrence of the mRNA and the tRNA can make functional T7 polymerase, which can activate the downstream T7 promoter.
The AND Gate can be further decoupled into an input sensor, a core, and an output.
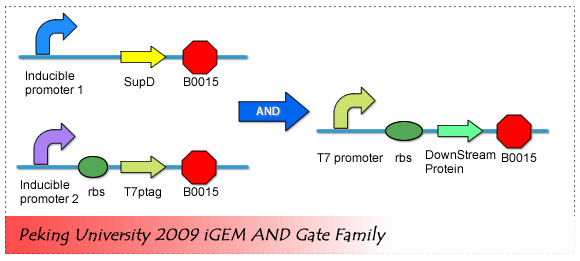 fig2. AND Gate Biological Components The input interface includes two inducible promoter which can be induced to expression by two different environmental small molecules. Core of the AND Gate is the T7 polymerase coding sequence with amber mutations and supD tRNA. And the Output interface is actually a T7 promoter, which regulate the expression of the downstream component.
Sensor Module (Connected to the input interface of the first AND Gate)
The first AND Gate directly detects the two environmental signals, so the input interface of the first AND Gate was connected to two inducible promoter which can be induced to expression by two different environmental small molecules.
We actually prepared five inducible promoter, and make combinations among the five promoters. The five promoters are tetR repressible promoter inducible by aTc, PBad promoter by arabinose, Lac Promoter inducible by IPTG, Salicylate inducible promoter and Lux promoter that responses to HomoSerine Lactone.
All of these inducible promoters can only work in the presence of activators or repressors, so we cloned all the activators and the repressors downstream of a constitutive promoter to make a protein generator. Then the generator is placed upstream of the corresponding inducible promoter.
There are two ways, actually, to put the generator upstream of the inducible promoter.
As shown in fig3.
1. The constitutive promoter is cloned back to back with the inducible promoter, thus transcription of the activator or the repressor is opposite of the inducible promoter. 2. The generator transcripts in the same direction of the inducible promoter, the constitutive promoter is prevented to interfere with the inducible promoter by the terminator. However, any terminator is leaky, the first way is the better way to prevent interference.
This construct combines repressors (activators) together with its promoter. It is actually a quite useful part even outside of the AND gate Module. We call it an environmental small molecule sensor. It provides a sensor for environmental signals and a interface to intracellular circuits. Actually, a similar construct(repressor protein only) without the constitutive promoter is called an inverter, but we think it is more proper to be called a sensor when the promoter upstream of the repressor is a constitutive promoter. Its signaling molecule like aTc, IPTG, arabinose, HomoSerineLactone and salicylate diffuse into the cell and take function. It is like the some of the hydrophobic hormones in high organisms.
The promoters we chose at first is the LuxR-LuxP construct induced by Homoserinelactone, the tet operon induced by aTc, the Lac operon induced by IPTG, and the Arabinose inducible promoter PBad. So we designed the following sensors:
The aTc Sensor is a tet promoter repressible by tetR protein. The repression is released in the presence of aTc.
The IPTG Sensor is a lac promoter repressible by lacI repressor.
And because the LuxR-LuxP sensor has already been constructed by iGEM2006_PennState. So we didn’t reverse the LuxR coding sequence but just use their construct.
The araC protein is expressed upstream of PBad to make a arabinose sensor which can express downstream gene when induced by arabinose.
The salicylate Sensor:
The salicylate Sensor is not made up of standard parts, it is originally on the pSal-SupD plasmid that Voigt Lab mailed us. We got the sequence from NCIBI there is no standard enzymes inside of its sequence. We designed primers and PCR it from the plasmid. Then the promter is placed upstream of part E0840 to characterize its induction curve.
AND Gate 1 Module
SupD tRNA and T7ptag mRNA makes the core of this AND Gate.
T7ptag gene is a T7 polymerase coding gene with two amber mutation at it Serine codon. The mutated T7 polymerase gene can only be transcribed but cannot be successfully expressed for the failure of translation.
The SupD tRNA is an amber mutation suppressor, this tRNA recognizes the TAG stop codon and mandatorily translate it into a Serine. Thus with supD, the function of the T7 polymerase gene is recovered. According to the Paper "Environmental signal integration by a modular AND Gate", SupD tRNA has little side effect on E.coli, although it can suppress normal termination of translation.
We Use PCR to make T7ptag and SupD into standard biobrick. The primer is designed according to the paper, we keep the complementary sequence in the original primer, and replace the enzyme cut site in the original primer with standard prefix and suffix. The original plasmid where we get SupD sequence and T7ptag sequence is shown below:
 fig8. The Original Plasmid Where We Got the SupD and T7ptag Gene Thus the T7ptag and supD gene are made into standard parts. Because our project employs many parts and devices, it is important for us to prevent these parts to interfere with each other. So the First thing after the core parts are constructed, we put a strong terminator BBa_B0015 downstream of the two parts.
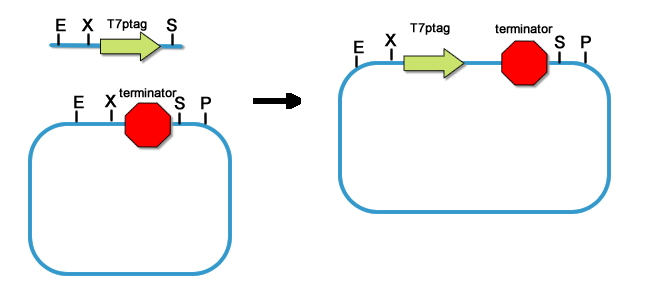 fig9. Add Terminator downstream of T7ptag 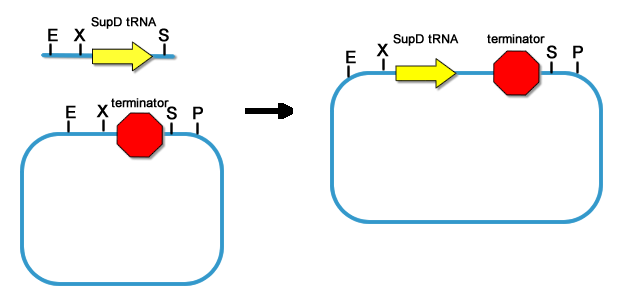 fig10. Add Terminator downstream of T7ptag  fig11. Placing rbs upstream of T7ptag-terminator
For T7ptag, we clone it downstream of 9 different rbs. While SupD is just tRNA, we don’t need to add rbs. So far, the core of the AND Gate is constructed. Before the AND Gate is functional, standard regulatory parts should be placed upstream of the T7ptag and SupD construct.
In order to pass signal from this AND gate to downstream modules, a T7 promoter is needed as an output interface. So the downstream protein can be regulated by the T7 promoter. Which is described at the end of this page.
Integration of the Sensor to the input interface of AND Gate 1
Actually, the sensors we constructed is not all successful, for example, the aTc, IPTG sensor (tetP and LacP with its repressor constitutively expressed) is leaky and shows little induction. Later, we got four inducible promoter that shows induction in the presence of the inducer. The arabinose inducible promoter is successfully constructed, and the salicylate inducible promoter is also PCRed and well characterized, the LuxR system, although leaky, show a maximum induction of 8 fold. The lac promoter can only work in a E.coli Strain that has lacIq mutation, we chose JM109 strain.
For those successfully constructed, either two of the inputs are combined to make an AND Gate. Theorectically, there are 12 combinations in total.
| | LacP-SupD | LuxP-SupD | PBad-SupD | SalP-SupD
|
| LacP-T7ptag | N/A | Shan Shen | Shan Shen | Siheng He
|
| LuxP-T7ptag | Not Made | N/A | Rencheng Gao | Not Made
|
| PBad-T7ptag | Shuke Wu | Not Made | N/A | Haoqian Zhang
|
| SalP-T7ptag | Not Made | Not Made | Guosheng Zhang | N/A
|
AND the variation of rbs makes a total of 108 AND Gates.
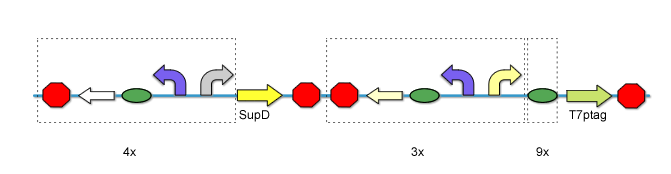 fig12. The total Variation of AND Gates We constructed and tested 8x9 of them, and finally got two AND Gates that best fits our need.
Directing the Output Interface
In our project, we need to pass signal from AND Gate 1 to CI repressor protein to trigger the bistable module. We also constructed T7 promoter-rbs-CI-terminator library for later system assembly. At the same time, in order to test our AND Gate, we constructed a T7 promoter-GFP reporter output. The AND Gate module is moved to the pSB4K5 plasmid, and we constructed a T7 promoter-GFP reporter on a Amp resistant and colE1 origin plasmid. By transforming both plasmids, and measured the fluorescence by plate reader after induction, we successfully selected two AND Gates that best fits our need.
^Top
|