Project > AND Gate 1 > Sensor Result
Construction result
Input promoters construction:
Before the construction of AND gates, five different inducible promoters were chosen as inputs: pBad (respond to L-arabinose), pSal (respond to salicylate), pLac (respond to IPTG), pTet (respond to tetracycline) and luxP (respond to homoserine lactone). When the inducing small molecules are in presence, the corresponding promoter is activated, for example, IPTG can induce pLac.
We have constructed four of these inducible systems (including the activator or repressor protein and promoter), from basic parts, and used one directly from other composite parts. The general pattern is like this:
They are:
BBa_K228004: NahR-pSal system;
BBa_K228009: AraC-pBad system;
BBa_K228817: lacI-pLac system;
BBa_K228818: tetR-pTet system.
They are not only composite parts which can be used in our project, but also valuable devices which can be employed in other designs or circuits.
For the luxR-luxP system, we simply utilized the parts: BBa_J09855, which is designed and constructed by iGEM2006_Pennstate.
Input promoters test:
In order to characterize the AND gate (e.g. the transfer function), the activities of these promoters should be measured independently by constructing GFP (with strong RBS B0034) downstream the promoters and testing the output in response to the small-molecular inducers.
Therefore we successfully fused these promoters to GFP and test results are following:
Salicylate Sensor
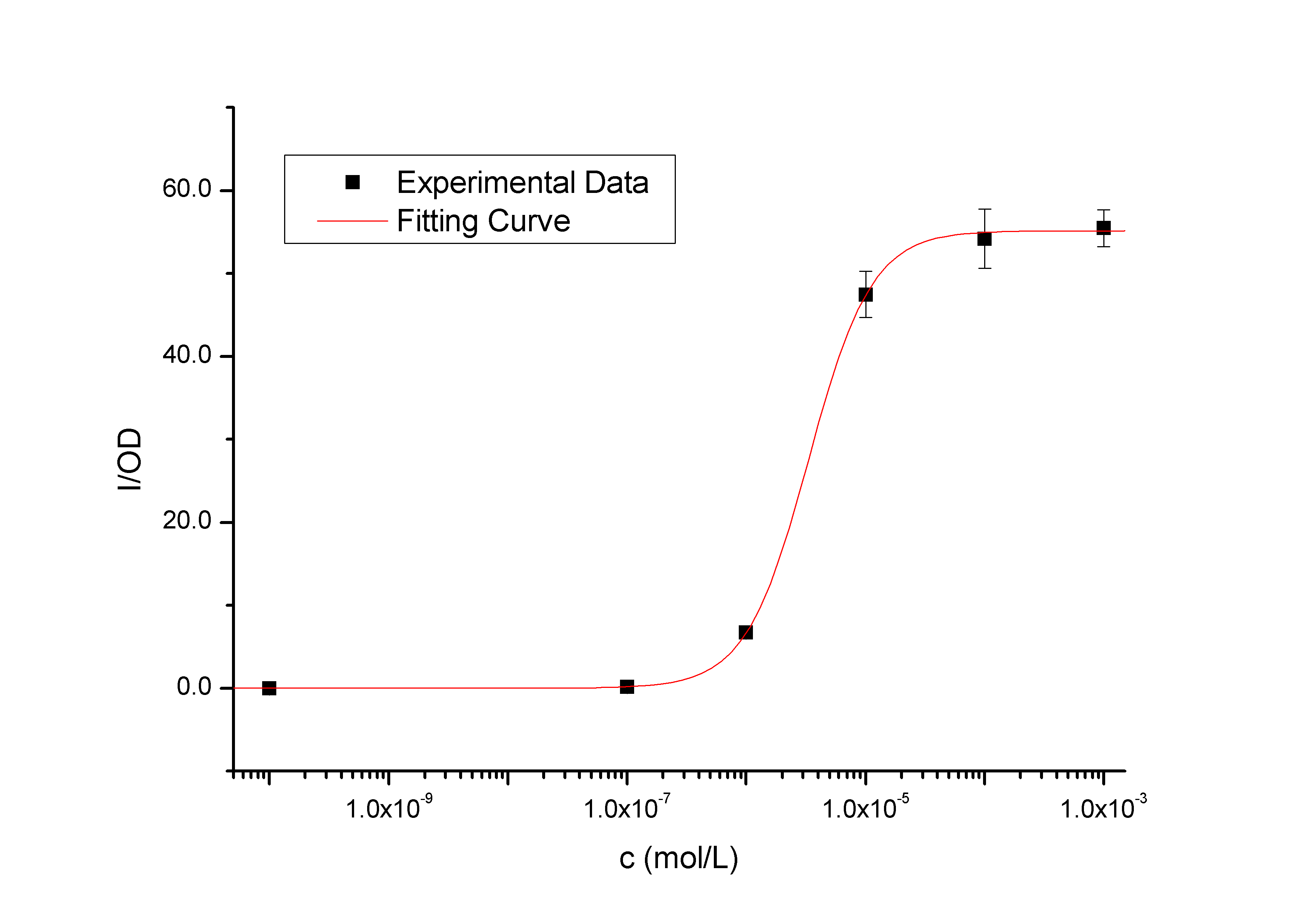 Fig1 - Transfer function of BBa_K228004. This transfer function is the 120 min time-slice from the time and dose dependent input-output surface. The data points represent the mean of 6 individual measurements. The corresponding error bars represent the 95% confidence interval in the mean of the independent measurements. The solid black curve was calculated by fitting a simple Hill function to the experimental measurements. The Y axis denotes the value of fluorescence normalized by the OD600 value, and the X axis denotes the concentration of the inducer.
In this salicylate inducible promoter part, the activator protein (NahR) gene is in the upstream but in an opposite direction of the regulated promoter. In the presence of salicylate, the promoter can be activated. We placed a generator on its downstream; the transcription of the coding sequence will be triggered by the salicylate-binded positive transcriptional activator protein which is encoded by NahR.
For the purpose to test the promoter, we connected BBa_E0840 downstream of the salicylate promoter to make the composite part, BBa_K228850. This report system allowed us to test the salicylate inducible promoter indirectly via the fluorescence output of BBa_E0840, while the input is salicylate solution at a gradient of concentration.
We tested the promoter (act as a sensor) through the protocol described in the Tramsfer Function page. The time between repeated samplings is 20 minutes. 6 replicates were measured for each concentration of salicylate, and the concentration 0 group is to measure fluorescent background.
We converted the OD600 value into the concentration of cells (/ml). Then we normalized the GFP fluorescence by the OD600 values, to obtain the ratio of GFP fluorescence to the OD600 values. The mean for each concentration-time group was then averaged across 6 replicates to obtain a population mean.
The transfer function in Figure 1 is the 120 min time-slice from the time and dose dependent surface. The data points represent the mean of 6 individual measurements. The corresponding error bars denote the 95% confidence interval in the mean of the independent measurements. In Figure 2, the level of induction triggered by the same concentration of inducer (of which concentrations are higher than 10^-7 mol/L, causing concentration of 10^-10 mol/L group has no significantly positive differences with the blank group) will be enhanced as the induction time extended, so it is the same with the saturation of fluorescence, as expected.
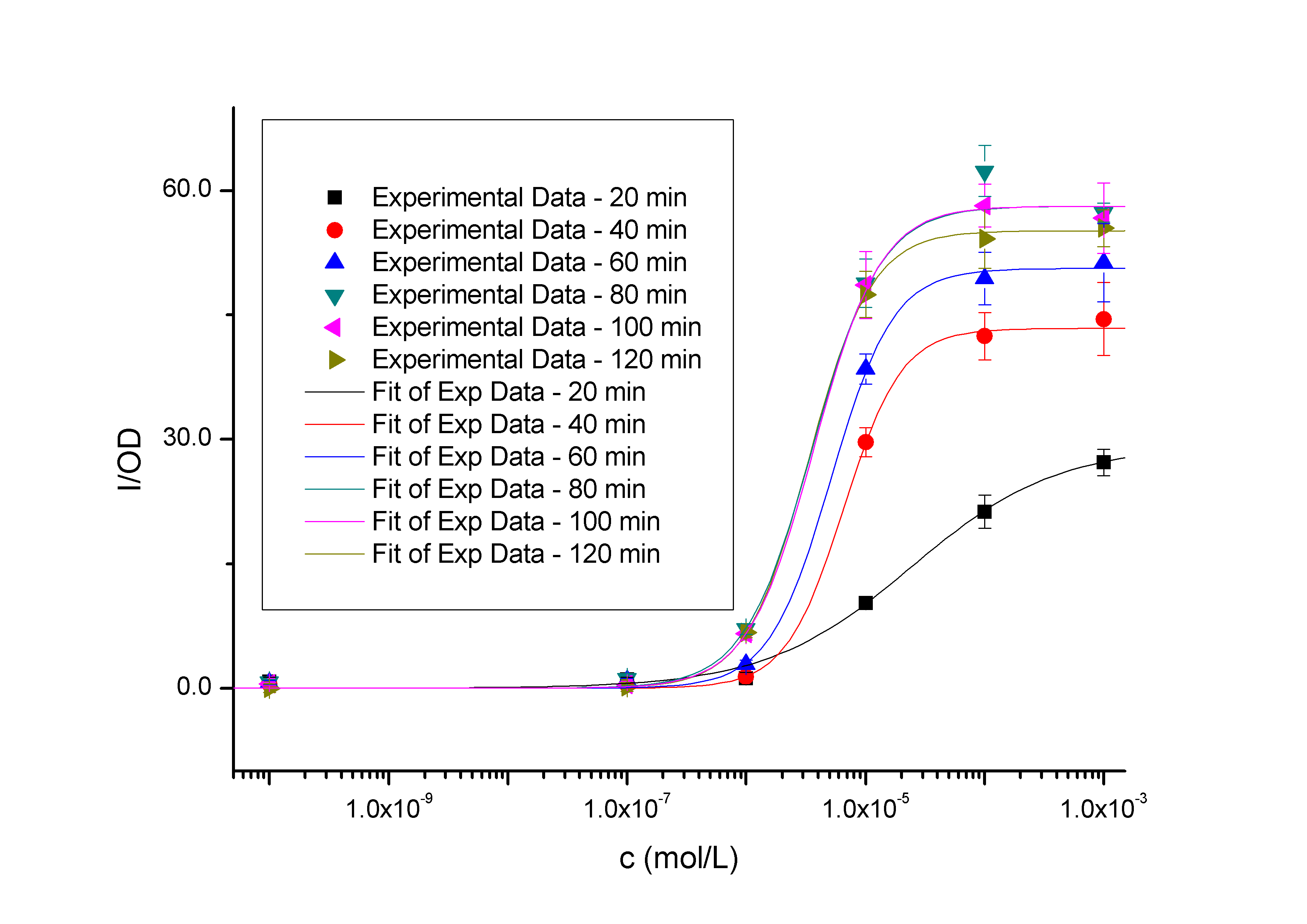 Fig2 – Transfer functions of BBa_K228004 in different time slices from the time and dose dependent input-output surface. The data points represent the mean of 6 individual measurements. The corresponding error bars denoting the 95% confidence interval in the mean of the independent measurements. These solid curves were calculated by fitting simple Hill functions to the experimental measurements. The Y axis denotes the value of fluorescence normalized by the OD600 value, and the X axis denotes the concentration of the inducer. As we can see, the level of induction triggered by the same concentration of inducer (higher than 10^-7 mol/L, causing concentration of 10^-10 mol/L has no significantly positive differences with the blank) will be enhanced as the induction time being extended, so it is the same with the out-put saturation. We used least squares estimation to fit a simple model to the data assuming that GFP is not degraded. Hill equations derived from simple biochemical equations describe the data well (Table 1). Pmax is the maximum output level, K is the switch point, and n is the hill coefficient describing the steepness of the transition from low output to high output.
Table 1. Parameters of the measured transfer functions
| | Pmax | K | n | R^2
|
| 20 min | 29.2±3.6 | 2.42E-05±1.07E-05 | 0.710±0.144 | 0.98124
|
| 40 min | 43.4±4.4 | 6.54E-06±1.48E-06 | 1.792±0.275 | 0.97309
|
| 60 min | 50.6±6.6 | 5.13E-06±1.71E-06 | 1.677±0.335 | 0.96184
|
| 80 min | 58.1±2.5 | 3.59E-06±1.16E-06 | 1.520±0.373 | 0.99088
|
| 100 min | 58.1±3.2 | 3.70E-06±1.05E-06 | 1.567±0.316 | 0.98898
|
| 120 min | 55.2±0.3 | 3.32E-06±8.29E-08 | 1.649±0.032 | 0.99999
|
We also observed salicylate may decrease the growth rate of E.coli, for more detail refer to our favorite parts: K228004
Arabinose Sensor
The araC-pBad system: (Part: )
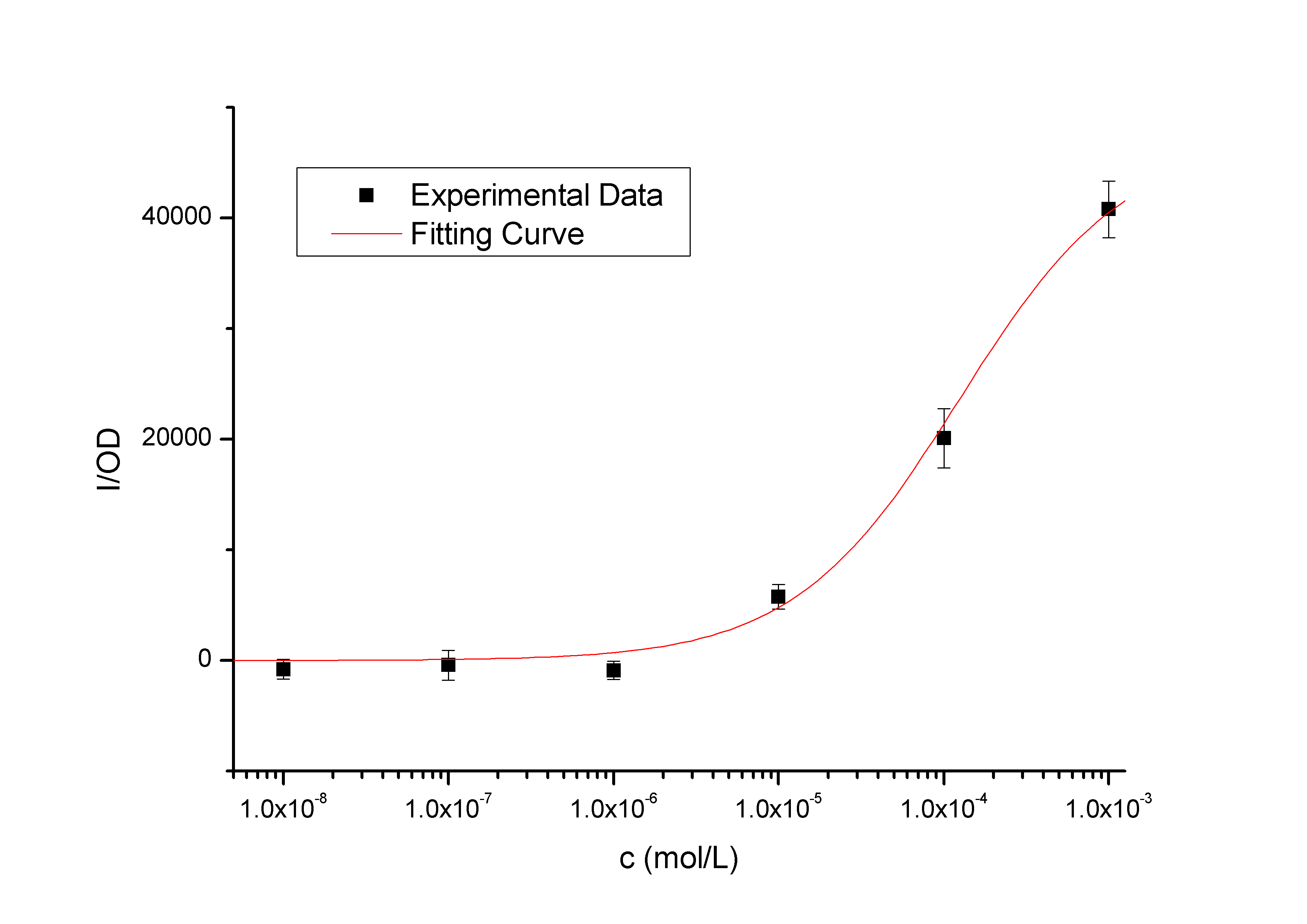 Fig3. Transfer function of arabinose sensor. This transfer function is a 120 min time-slice from the time and dose dependent input-output surface. Data points represent the mean of 6 individual measurements. The corresponding error bars represent the 95% confidence interval in the mean of the independent measurements. The Y axis denotes the value of fluorescence normalized by the OD600 value, and the X axis denotes the concentration of arabinose. We have constructed the inducement system composed of L-Arabinose inducible Pbad promoter and constitutively expressed AraC regulatory protein. When arabinose is absent, the AraC protein binds to and represses Pbad very efficiently. Thus, the transcription of any downstream coding gene is restricted. On the other hand, the import of arabinose could change the conformation of AraC protein, successfully prevent it repressing Pbad promoter and activate the transcription of downstream coding gene, such as GFP.
The direction of AraC coding sequence is opposite compared to the Part . We have PCRed with primers which include standard enzyme-cutting sites. The aim of this reversion is to avoid unexpected expression leaking of downstream sequences.
The transfer function describes the equilibrium relationship between input (L-Arabinose solution at a gradient of concentration) and output signals (GFP fluorescence). For the purpose of test, we placed GFP coding gene (Part BBa_E0840) downstream of Pbad promoter to contruct a reporter system, which allows us to use Microplate Reader to test the arabinose inducible promoter indirectly via the green fluorescence output according to the concentration gradient of arabinose.
aTc Sensor
 Fig4. the result of tet promoter system. This is a picture of tet system, the colonies are green under blue light, even without induction, the upper plate is for comparison. The tetR-pTet system (parts: )
The colonies which contain the aTc sensor - GFP plasmid become very green without induction and showed no induction after aTc is added to the base. The evidence that it is green even without induction is on Fig1, and the data after induction are not showed here.
Discussion: the leakage is obvious and the reasons may lie in the constitutive promoter we used to drive the expression of tetR and the LVA tag of tetR coding sequence. The constitutive promoter we used is pCat(BBa_I14033) which is a medium promoter, while the natural promoter of tetR is pTet itself. It is likely that the expression of tetR is insufficient to repress pTet. Another possible reason is the LVA tail of the tetR(For detail see BBa_C0040), and its function is for rapid degradation the protein. As a consequence, there are not enough TetR in presence to repress the pTet.
IPTG Sensor
The lacI-pLac system (Part: )
The result is similar to the tetR system. The colonies which contain the IPTG sensor - GFP plasmid expressed GFP even without induction, and the GFP flourescence didn't increase even after IPTG induction. The evidence that this construct is leaky without induction is on Fig5, and the data after induction are not showed here.
Nevertheless, we found another way to bypass the problem: take advantage of the lacIq on the F plasmid of E. coli strain JM109. Since the F plasmid is one copy per cell, pLac () should be on a low copy plasmid, such as pSB4K5. The pLac can be repressed by endogenous lacI even without its represssor coexpressed on the same plasmid. Thus we constructed the simplified system (Part: ) and induced it by different concentration of IPTG. The result is shown in Fig5, judging from the induction curve, pLac promoter may be qualified in constructing the AND Gate.
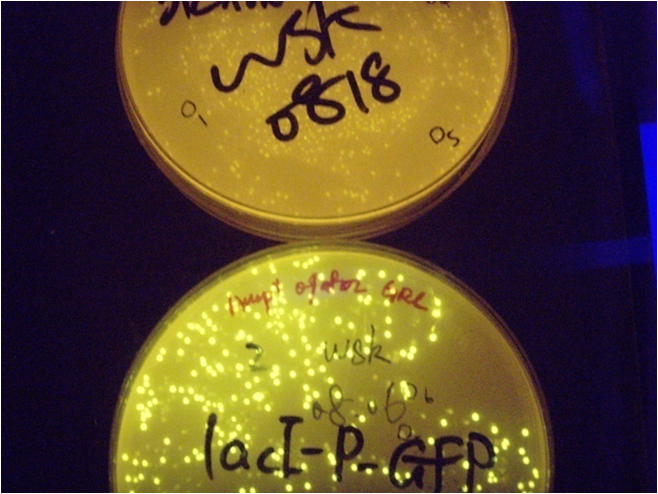 Fig5. the result of pLac promoter system. The colonies are green under blue light, even without induction, the upper plate is for comparison. 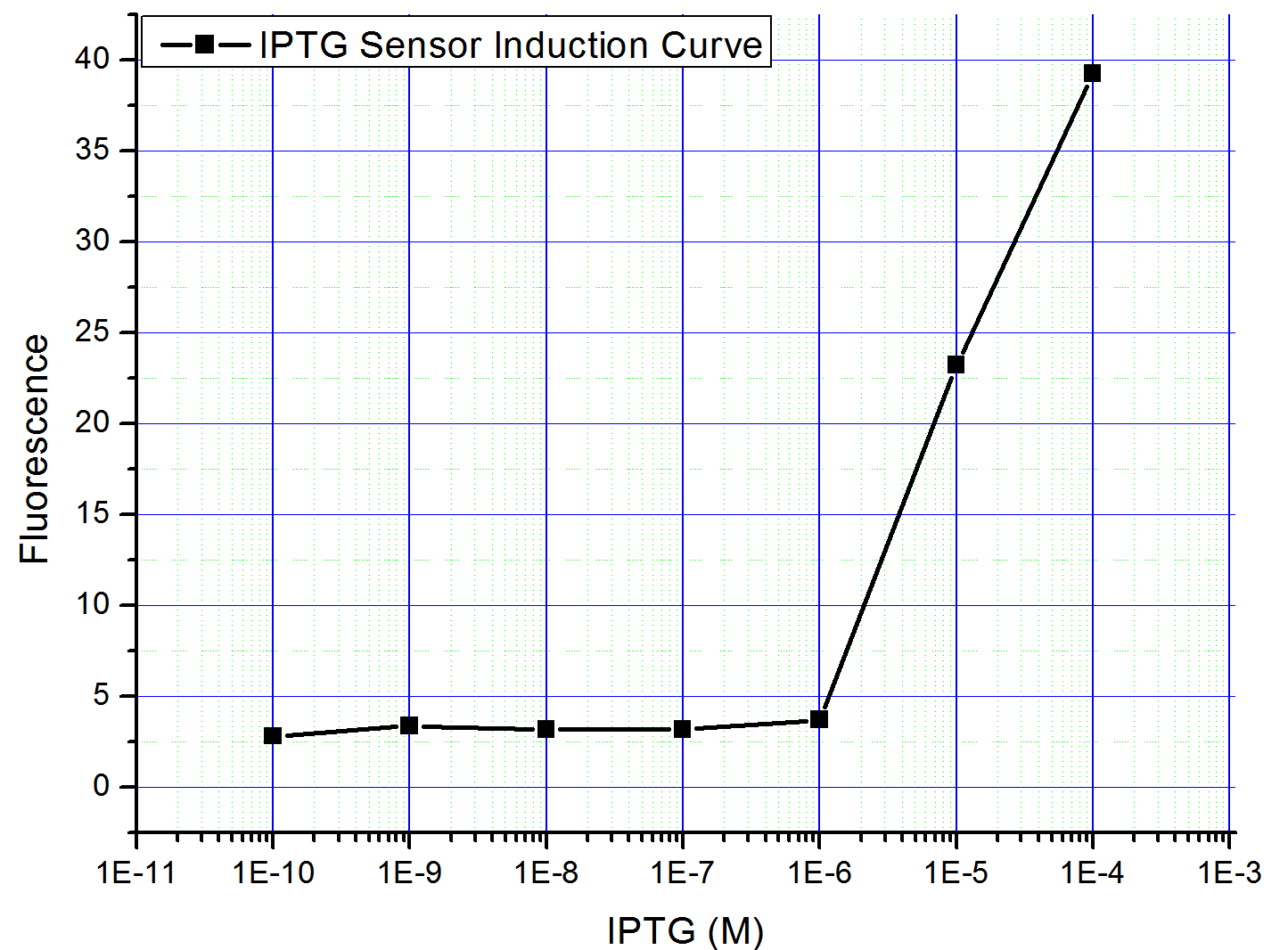 Fig6. lac promoter induction curve. The Bacteria is induced with 10^-9~10^-3 M IPTG, the fluorescence is measured by plate-reader
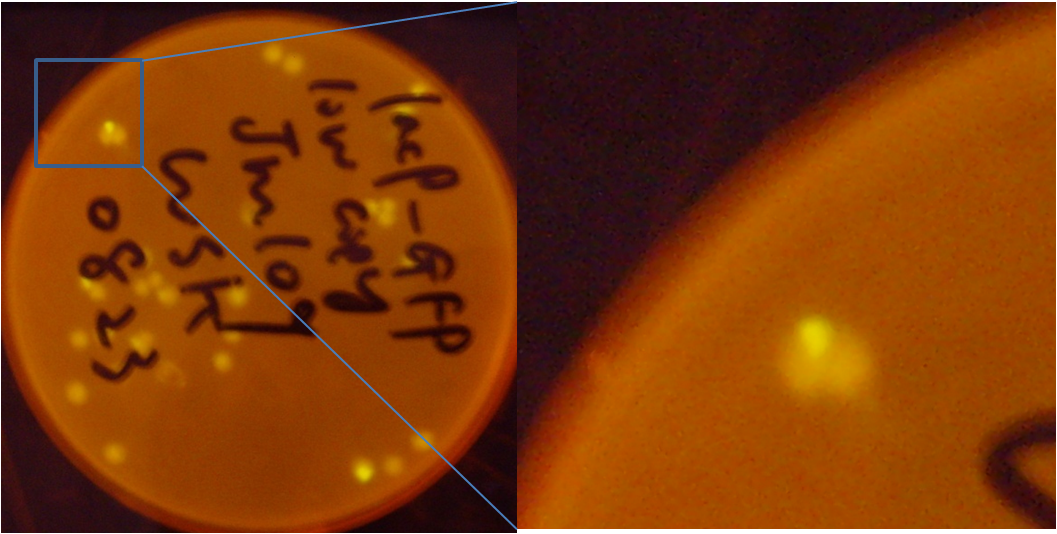 Fig7. the JM109 colonies that contain low copy pLac-GFP plasmid. Center of the colonies turns green, while in the other area GFP expression is suppressed. It suggests that some E.coli lost their F plasmids
However, there is still another problem in this simplified system: the loss of F plasmid of JM109 may lead to activation of promoter pLac without induction. From the plate (without induction), we found that part(especially the central part) of a singal colony turned green, while they are supposed to stop express GFP in the presence of lacIq. A stronger evidence is the data from flowcytometry: The strength of GFP fluorescence obviously shows double peaks. All these suggest that some of the cell retains the F plasmid while the the others lost it.
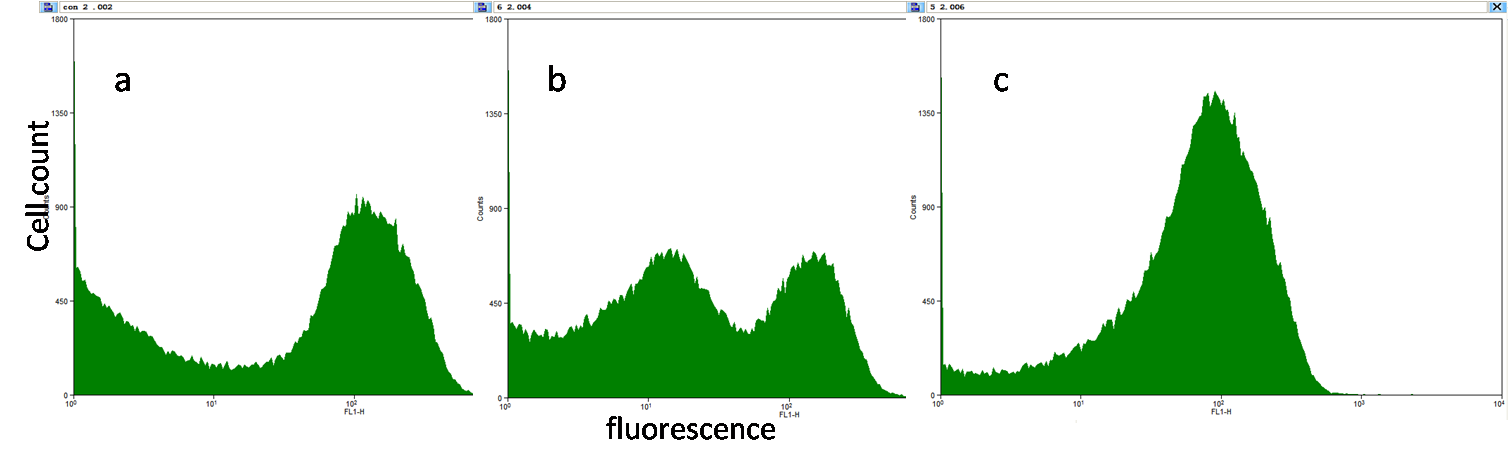 Fig8a, b & c. The fluorescence was measured by flowcytometry to see the fluorescence of each cell in a population. Fig5a illustrates that there are leaky cells and normal cells without induction. Fig8b shows the data from the cells induced by 10^-6M IPTG, and Fig8c show those induced by 10^-5M IPTG. Double peaks can be observed on both 8a and 8b, which means there are two groups of E.coli, but as the induction saturates, two peaks merge into one peak. In a word, the pLac promoter is not a perfect promoter to construct AND Gate. However, if a more stable source of lacI can be supplied, we believe it would be better.
HSL Sensor
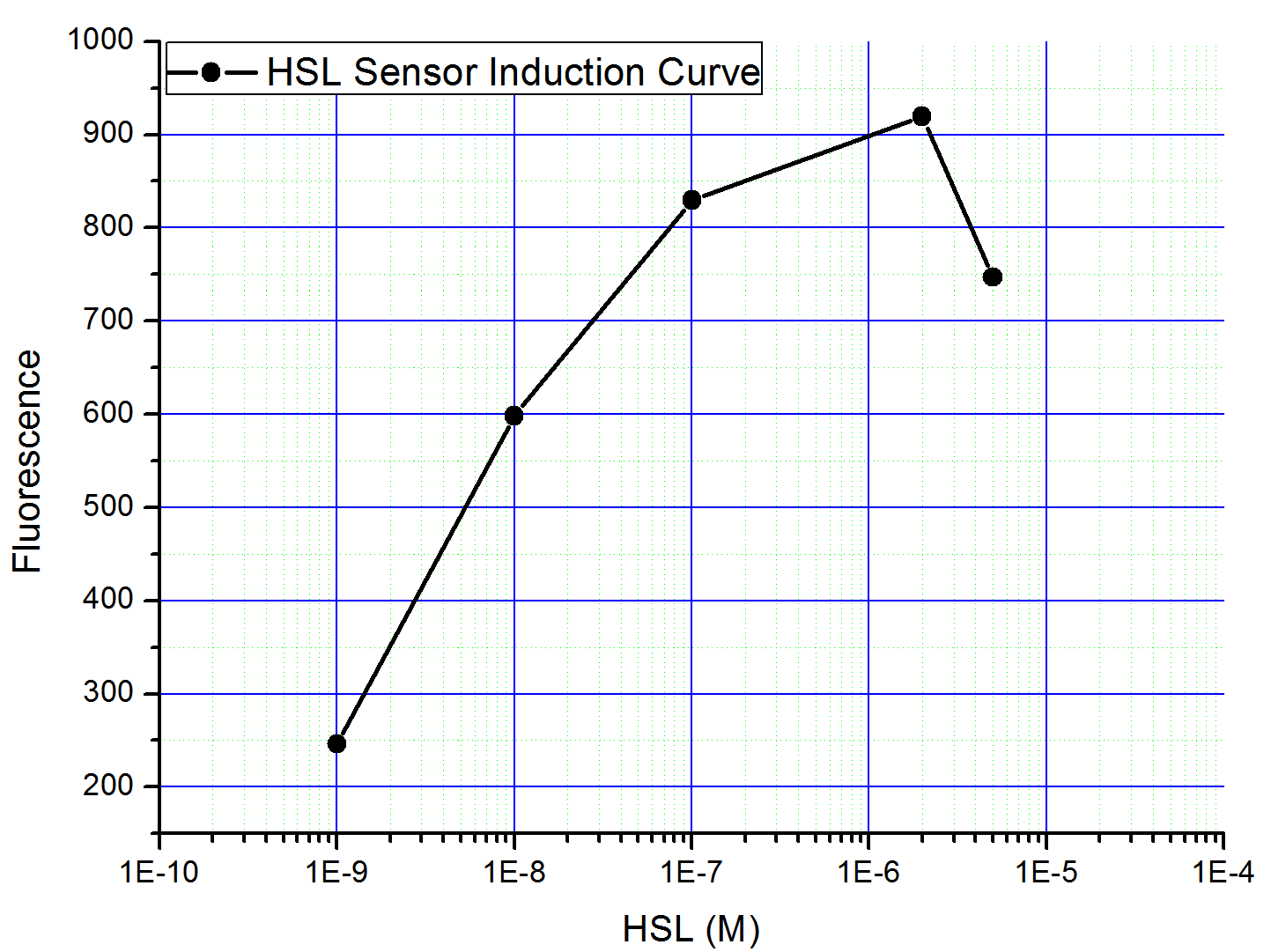 fig9. The Induction Curve of HSL Sensor
The luxR-luxP system (Parts: K228010, mainly by Min Lin)
We have constructed the plasmid (based on the part:, constructed by iGEM2006_Pennstate. ), and used different concentration of homoserine lactone to induce (See Protocols). The fluorescence of individual cell is tested by flowcytometry. The result shows that this HSL Sensor can be induced to 8 fold with HSL(Fig 9).
Although induction can be observed when HSL is added to the medium, the basal of this promoter is quite high. And it is later that we found this promoter is not qualified enough to construct our AND Gate(See the AND Gate 1 Result Section).
^Top
|