Team:SupBiotech-Paris/Concept1
From 2009.igem.org
Contents |
Tissue vector
Tissue vector function is to carry cell vector to reach the organ of interest, the one containing the target cell.
It should contain three essential characteristics. As a bacterial vector, it should not be pathogen for the patient, or at least not much harmful. It should have an interesting tropism, which means to be able to attain the target tissue. And finally, the most important characteristic, it must be able to foil the immune system, in order not to be destructed or eliminated before reach its objectives.
During this chapter you will discover on which characteristics we based our reasoning to choose the best bacterium in term of efficiency.
Then, you will discover in more details its properties through different constraint imposed by DVS concerning its
pathogenicity, its resistivity and its tropism.
Finally, we will present you an improvement brought to the bacterium in order to make an optimal tissue vector.
The tissue vector: Mycobacterium avium [1,2]
We studied several bacterial types to determine the best ratio between a low pathogenicity, an interesting tropism and a good resistance to the immune system, to choose the best tissue vector.
A hard task, cause in general rules, a microorganism which resists to the immune system is potentially dangerous for its host. We had to find a resistant microorganism but which does not have a high pathogenicity for humans. Indeed it is easier to reduce the pathogenicity than improve the resistance, we have chosen this last factor for the selection.
At first, we list bacteria presenting a resistance to the immune system. The four most known are:
- Mycobacterium tuberculosis, responsible agent for tuberculosis;
- Legionella pneumophila, responsible agent for Legionnaire's disease;
- Listeria monocytogenes, responsible agent for meningitis;
- Coxiella burnetii, responsible agent for Q fever.
These four are of course highly pathogen, we looked then at their homologous or under-species less pathogen for humans.
Mycobacterium avium (M.avium) is the bacterium which retains the most our attention. This last one belongs to Mycobacterium specie, and more precisely to the Mycobacterium Avium Complex (MAC).
M. avium is less pathogen than its homologous Mycobacterium tuberculosis. The only clinical manifestations it can induce are: productive cough, tiredness, fever, loss of weight and night sweat. These symptoms result from the colonization of the respiratory or gastrointestinal tractus and the possible diffusion in other parts of the body. Immunocompetent subjects having not a subjacent disease have a low risk to present infection symptoms by M. avium. Epidemiologic reports recently show that we find more and more the presence of M. avium in subjects, notably women who seem to have no pulmonary or immune predisposing trouble. Most of healthy subjects infected by M. avium have a localized infection; in contrast, we notice infection by M. avium disseminated in an important percentage of patients infected by AIDS (80% of colonized patients), as in other population of which the immune system is deficient, like subjects having a severe combined immunodeficiency syndrome or which received a transplantation and in patients treated by corticosteroids or cytotoxic medicines.
M. avium seems to be one of the most interesting candidates to be the tissue vector. This bacterium resists to immune system and is few pathogen for Man.
Immune system resistance of the tissue vector [3,4,5]
Immune system can be a terrible enemy, detecting and eliminating any element considered as foreign for the organism. Impossible to be stealth to it, it must be “hardened”.
For that the tissue vector has two assets, a system to divert the immune system and a robust wall.
Immune system resistance mechanism
Bacteria like Mycobacterium avium developed resistance strategies highly specific to foil the Immune system.
So, M. avium, facultative intracellular microorganism, has the capacity to infect some cells of the immune system like macrophages or dendritic cells, responsible for phagocytosis.
Infection generates phagocytosis death, and then we observe in the host a decrease of its non specific immunity, which permits to M. avium to persist in its host.
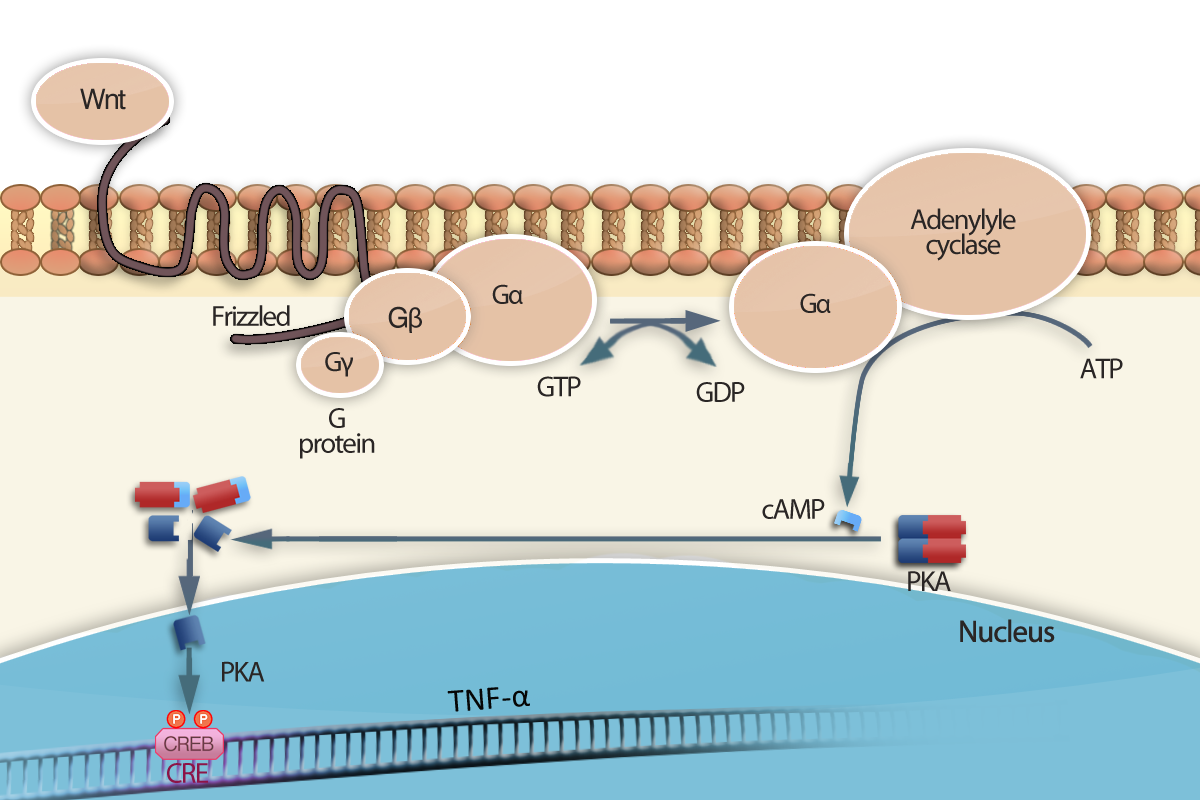
To fight against macrophages, M. avium inhibits phagolysosomes maturation, initially responsible of bacteria lysis. This mechanism paradigm is not already determined, but the principal hypothesis involves a cell wall glycolipid, the lipoarabinomannan.
To fight against dendritic cells, M. avium upsets cellular transduction signals by releasing some enzymes regulating the second messenger’s rate.
Indeed, M. avium has, in its genome, 12 genes containing classes III cyclase adenylate domains, the most important is Rv0386. This one facilitates the AMPc formation in macrophages. AMPc is a second messenger leading to the pro-inflammatory TNF-α cytokines synthesis.
But, at high concentration, TNF-α stops dendritic cells maturation and then the immune response sets off.
M. avium does not “fear anything” in front of the immune system, its elimination is low. It has the capacity to persist in the organism and therefore has more chance to attempt its target and to establish itself.
Cell wall and tissue vector envelope
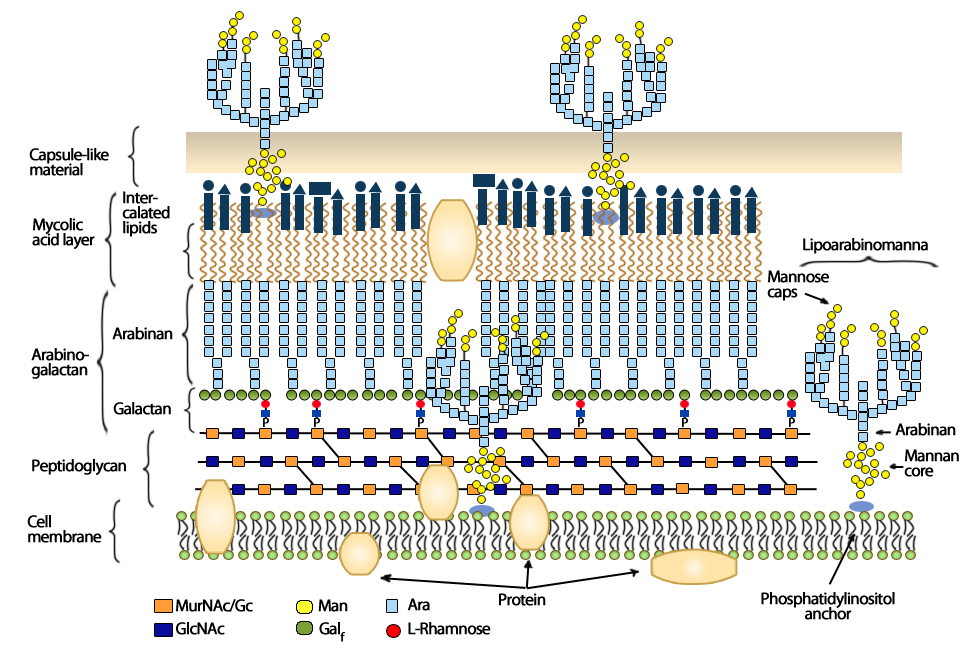
Mycobacterium capacity to resist to the immune system is essentially due to its wall and cellular envelope. These both present a complex structure and is specific to Mycobacterium.
Real breastplate, M. avium envelope is composed by several elements. Some, like soluble proteins, carbohydrates, and lipids are common to all bacteria.
On the other hand others, specific to M. avium, are the most interesting, like these three insoluble macromolecules:
- arabinogalactan,
- peptidoglycan,
- mycolic acid.
Together, these 3 constituents form the mycoylarabinogalactanpeptidoglycane (MAGP) heart of the cellular membrane. MAGP represents one of the two lipopolysaccharides (LPS) common to all mycobacteria, but M. avium possesses a second LPS: the lipoarabinomannan.
We observe as well, in M. avium, a glycopeptidolipids (GPL) lay as specific.
All this structure (MAGP, GPL and lipoarabinomannan) is highly immunogen like LPS is in others bacteria. Unlike to other bacteria, this complex structure gives a special resistance to M. avium. The network formed by the membrane, based on parallel hydrocarbonates, permits to the bacterium to be impermeable to several agent like ethanol, detergent or classical antibiotics.
Tissue vector wall and its faculty to synthesize cellular transduction hecklers permit itself to be one of the only vector to be resistant to the immune system. This is probably one of the major asset of the DVS.
Targeted tissu of the DVS [6,7]
DVS can be adapted to several pathologies in function of its tissue vector tropism. These last are linked to the tropism of M. avium “Wild type”, but it is possible to transform M. avium to make it sensible to some organic chemotactisms. Now, let’s discover what possibilities offer M. avium “Wild type”.
M. avium can infect an organism by oral and intravenous way. If its homologous M. tuberculosis is often seen like a pulmonary pathogen agent, M. avium can eventually infect all organs. This affirmation is verified in immunodepressed patients in which the decreased immunity permits to the bacterium to propage in the entire organism.
Deeper researches on tropism revealed some targeted organs. It is shown that weeks after infection, bacterium concentration reaches a plate in two organs: spleen and liver and later in a third one: lungs.
Figure 2: Lung, spleen, liver and thymus invasion by M. avium from 0 to 52 weeks in log 10 (6 to 10 mice)
We observe as well that during several weeks post-infection, the bacterial amount still increases. Although, this increase in targeted tissues is not correlated with a Mycobacterium increase in blood. So, the increase is due to targeted tissues invasion and not to the entire body invasion.
So, there is a tropism as well as an implantation of the vector in targeted tissues. This characteristic is really interesting because the vector is not circulating but resident.
The tissue vector first function is to deliver the cell vector in the targeted organ. If the liberation is too early, the cellular vector has less chance to reach the interested cell. Alone, the cell vector is eliminated by the organism, but in the tissue vector, it is safe.
The tissue vector possesses three interesting and identified tropisms. The DVS offers a therapeutic application for lungs, spleen and liver pathophysiologies.
The tissue vector can target these organs, implant them, and develop inside of these tissues, until receving the delivery signal of cell vector.
Delivery system of the cell vector
An implanted system in bacterium permits to control expression and then cell vector liberation.
This control system by doxycycline, implanted in tissue vector is made by two elements:
- A synthesis plasmid of LacI, controlled by doxycycline.
This first plasmid code for LacI repressor under the control of the repressed promoter (pTET). pTET activity, and then LacI expression, depends totally on the doxycycline fixation on the TetR repressor.
- A LacP/O operator is implanted in the cell vector genome and controls the gene expression via control expression of phage repressor.
LacI is attached on phage genome LacP/O promoter, inhibiting its repressor expression.
Repression of the cell vector expression is achieved constantly by a phage repressor. In presence of doxycycline, phage repressor is inhibited, promoting cell vector expression.
Delivery system permits the cell vector to be maintained inside the tissue vector under the control of doxycycline. The M. avium monitoring and the realization of statistic way to determine the propagation kinetic are essentials. These information permit to determine injection times. Doxycycline injection causes the cell vector release which lyses the tissue vector and diffuses in the targeted tissue.
To summarize…
...DVS possesses a tissue vector having the following characteristics:
- Low pathogenicity, so low toxicity,
- Immune system resistance, so low clearance,
- Tissue targeting, so first specificity,
- Cell vector synthesis and delivery.
These characteristics, specific to DVS, give solutions to recurrent issues, of specificity and elimination by the immune system, meet by vectors.
 "
"