Team:TorontoMaRSDiscovery/Project
From 2009.igem.org
| Home | The Team | The Project | BioBricks | Modelling | Bioinformatics | Safety | Notebook |
|---|
Engineering bacterial micro-compartments to investigate metabolic channeling and its potential uses in biotechnological applications
Contents |
Background
Proteins operate as parts of integrated biochemical pathways and exhibit varying degrees of spatial organization ranging from sub-cellular compartmentalization to the formation of discrete complexes. In metabolic processes, the co-localization of enzymatic components has been shown to promote the transfer of substrates between consecutive reactions in a process termed “channeling”[1]. Channeling results in the efficient translocation of substrates between enzymes and has been proposed to result in the following benefits:
- Optimization of catalytic efficiency by decreasing transit time for intermediates
- Relief of the effects of product inhibition
- Protection from the creation of potentially toxic or unstable intermediates
- Regulation of substrate flux through mediating pathway cross-talk
We have taken an interdisciplinary approach to systematically investigate how nature implements metabolic channeling and how this knowledge may be exploited for biotechnological applications such as the production therapeutic molecules and biofuels [3] and the degradation of toxic wastes[4].
Design
Considerations
From an engineering perspective a successful implementation requires:
- A subcellular space for co-localizing enzymes (and intermediates) which would restrict free diffusion.
- A method of targeting chosen enzymes to this space
- A pair of enzymes that are expected to benefit from the effects of channeling.
Our search for a suitable platform to effect channeling led us to bacterial microcompartments. A detailed structural analysis of encapsulin nano-compartments has recently been reported [6]. Of particular interest to us is the fact that a discernible targeting sequence is known; a fact that distinguishes this compartment from other microcompartments. Our ongoing search for alternative microcompartments as well as our search for enzymes is described on our bioinformatics page.
We are evaluating enzymes for use in our system which are likely to be amenable to channeling as predicted by C. Sanford [7]. We want to identify associated pathways containing substrates and products that are 1) easily assayed (e.g. using colorimetric or spectrophotometric tests) and 2) potentially commercially relevant (i.e. produces a desirable product or breaks down an undesirable compound). Short-listed pathways will be modeled using SimBiology [8] and Cell++[9] (the latter was developed in our host laboratory) to predict the effect of channeling on pathway intermediates and products. Here, we present our proof-of-concept system based on an encapsulin microcompartment and fluorescent (eCFP) 'probe'.
Objectives
- Design, construct and characterize a micro-compartment expression system in E. coli.
- Demonstrate in vivo assembly of the expressed micro-compartments.
- Target a fluorescent marker (eCFP) to the micro-compartment.
- Identify and prioritize candidate enzyme pairs for channeling.
- Apply channeling to selected enzyme pairs.
Components
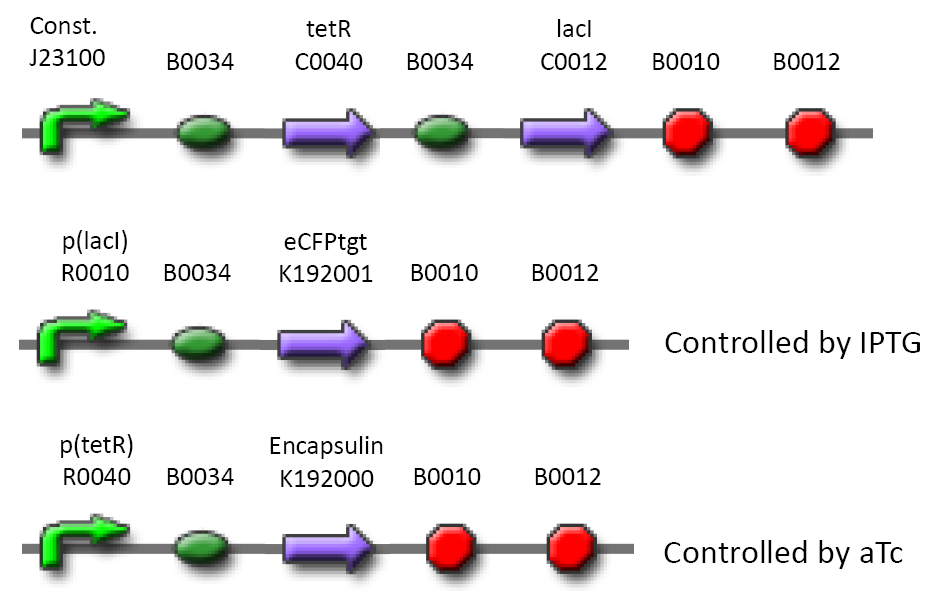
Using two new parts (see below) and several existing BioBricks [5] we have designed and are in the process of constructing an expression system capable of producing functional, T. maritima derived, encapsulin micro-compartments in E. coli (Figure 1). We include a 'control module' expressing two repressors under control of a constitutive promoter as well as the structural, microcompartment protein, encapsulin () and a fluorescent 'probe' () with a c-terminal extension corresponding to the conserved targeting sequence described by Sutter et al [6]. By varying the amounts of inducers aTc and IPTG in the system it is possible to vary the expression of encapsulin and the targeted fluorescent probe (eCFP). To determine whether encapsulin correctly assembles in this system we plan to use a standard negative staining (transmission EM) approach to visualize the microcompartments. To further establish that these compartments are functional, we will test our ability to target peptides to this compartment using the recombinant, fluorescent marker protein (eCFP) by fluorescence microscopy. This 'probe' carries an LVA tag which targets the protein for degradation. We hypothesize that encapsulation of eCFP will prolong its half-life and that by varying the amounts of the inducers aTc and IPTG in the expression system it will be possible to obtain a state where sufficient encapsulin is produced to enclose detectable amounts of eCFP and where background signal is minimized (see model). By measuring the difference in fluorescence between bacteria expressing encapsulin + eCFP versus those expressing only eCFP at a given optical density we hope to obtain an estimate of the amount of eCFP being protected from degradation and therefore localized to the micro-compartments.
Progress
We have made substantial progress in the construction of all components as detailed in our notebook. Specifically, the encapsulin biobrick was completed, sequenced and contributed to the parts registry. Also, as the construction of the fluorescent probe was not trivial, we took advantage of the offer from Mr. Gene to have this part synthesized. We have not yet completed standard assembly of the control module and are awaiting confirmation of this assembly before transforming the synthesized probe, at which point this part will also be contributed. There has been some difficulty obtaining the encapsulin assembly and we are beginning to suspect that our attempts to obtain the construct (initially with a constitutive promoter) may be killing the cells due to the effects of over-expression of the protein.
Our study of the natural biology of microcompartments across bacteria suggests that their occurrence is confined to a protective function, most commonly isolating toxic or reactive intermediates in certain redox reactions. While other microcompartments exist, the lack of a discernible targeting sequence makes them unsuitable for use in our system and encapsulin remains our best option at present. We have short-listed a series of enzyme pairs that we feel are amenable to manipulation by enzyme channeling based on thermodynamic and other properties for use in our future work.
Future Work
We continue to work on completing the assembly of our proof-of-concept system at which point we will proceed with the characterization of the system as planned. Modeling of candidate enzyme pairs for a downstream application will proceed using the results of our enzyme search. We anticipate that this will lead to several candidate applications. For these applications, we will construct recombinant enzymes fused to the targeting sequence. These enzymes will be co-expressed in the presence and absence of encapsulin and the effect on pathway intermediates and/or products will be assayed to determine the effects of channeling and to compare them with our modeled prediction. The nature of the assays will depend on the chosen system. It is possible that few commercially relevant pathways have products that are easily assayed by colorimetric or spectrophotometric means. If this turns out to be the case, we propose to first test an alternative, readily assayable system regardless of commercial relevance, to meet our scientific objectives. We note that there are several such pathways related to glycolysis that would meet this objective, including one previously modeled by C Sanford [7]. As a further experiment, we would like to apply channeling to a branching pathway to evaluate the potential role of channeling in dynamic pathway switching.
References
- Jorgensen, K. et al. Metabolon formation and metabolic channeling in the biosynthesis of plant natural products. Curr Opin Plant Biol, 8 280-91 (2005).
- Spivey, H.O. & Ovadi, J. Substrate channeling. Methods 19, 306-21 (1999).
- Atsumi, S. & Liao, J.C. Metabolic engineering for advanced biofuels production from Escherichia coli. Curr Opin Biotechnol (2008).
- Villas-Boas, S.G. & Bruheim, P. The potential of metabolomics tools in bioremediation studies. Omics 11, 305-13 (2007).
- Canton, B., Labno, A. & Endy, D. Refinement and standardization of synthetic biological parts and devices. Nat Biotechnol 26, 787-93 (2008).
- Sutter, M. et al. Structural basis of enzyme encapsulation into a bacterial nanocompartment. Nat Struct Mol Biol (2008).
- Sanford, C. M.Sc. Thesis: Modeling and Computational Prediction of Metabolic Channeling (2009).
- SimBiology 3.0 - Model, simulate, and analyze biological systems. Vol. 2009 (The MathWorks).
- Sanford, C., Yip, M.L., White, C. & Parkinson, J. Cell++--simulating biochemical pathways. Bioinformatics 22, 2918-25 (2006).
 "
"