Wisconsin-Madison/28 June 2009
From 2009.igem.org
Calendar
|
|
|
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
June 28, 2009
Ex 17: GFP Regulation by ProU Promoter
Test 3: Results: Fail, problems with the Plate Reader (connected to simulator)
Ex 15: Inducing Various Modified (triple) E.Coli Strains
Induction: 1. Irrabinose - 0.8 – 1% g/100mL
2. Wait 30 minutes
3. 1mM conc. IPTG (100uL/100mL)
4. Layer of Dodecane to trap isopentenol
Placed 5 ml of sample to into fresh 100ml LB, 50uL Amp, 73.5uL Cm, 100uL Tet
OD 600
Control / BL21 (DE3) / MG1655 (W) / MG1655 Delta Ara
.395 / .290 / .184 / .249
.364 / .246 / .174 / .211
.398 / .266 / .183 / .227
.425 (I) / .225 / .219 / .227
.564 / .240 / .214 / .229
.916 / .305 / .239 / .303
1.35 / .322 / .33 / .332
2.039 / .429 (I) / .427 (I) / .432 (I)
1.0087 / 1.048 / 1.086 / 1.0085
40uL IPTG solution? (0.002g/ culture)
Results: It took 2 hours to reach OD
Maybe IPTG and Arabinose kill cell
Maybe Arabinose slows maetablolis,
Note - Cultures grew extraordinarily fast – USE less see culture (2.5ml)
Ex 16: MiniPrep of Choline Colonies A,C
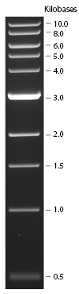
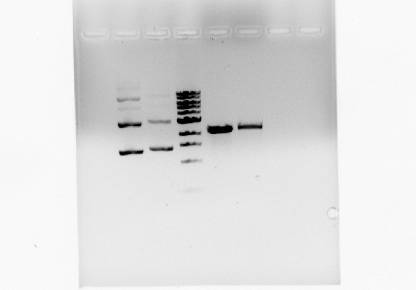
Ran on 1% Agarose Gel
Undigested (A, C), 1kb Ladder, Digested (A, C)
Pc: PEAMT cut
Pu: PEAMT uncut
Expected size: 4150bp
Cut with: Pst1
Ex 16:
Growth:
Streaked and Inoculated: multiple colonies from multiple plats for Miniprep tomorrow
Both iGEM Plasmid: pSB1A3 different digestions
P1 - cut Ecor1and Spe1
P2 - cut Xba1 and Pst1
Also transformed NudF an ProU from last ligation :
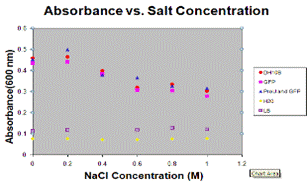
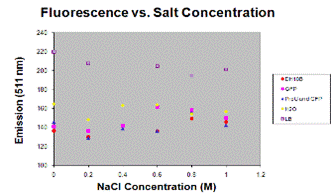
Ex; Absorbance and fluorescence taken, man
Autoclaving Medium:
- Standard is to fill bottle half way full
- It is OK to fill bottle full unless Agar
- Agar needs to be half, will bubble
- Better to make half bottle to avoid contamination of medium
Ex 10: Cyanobacteria
added 8mL to 2mL 6803 cultures to achieve 10mL cultures - NP
 "
"