Team:Berkeley Wetlab/Cell Surface Display Parts
From 2009.igem.org
Jennbrophy (Talk | contribs) (→Passengers) |
Jennbrophy (Talk | contribs) |
||
| Line 9: | Line 9: | ||
<font color="#41383C" size="2">Follow any of the links below to see assay information for each of the passengers we made.</font> | <font color="#41383C" size="2">Follow any of the links below to see assay information for each of the passengers we made.</font> | ||
| - | {| cellpadding="25" cellspacing=" | + | {| cellpadding="25" cellspacing="0" |
| <html> | | <html> | ||
| - | <div align=" | + | <div align="center"> |
<a href="https://2009.igem.org/Team:Berkeley_Wetlab/Passenger:_MGFP"> | <a href="https://2009.igem.org/Team:Berkeley_Wetlab/Passenger:_MGFP"> | ||
<img width="175" src="https://static.igem.org/mediawiki/2009/6/63/Mgfp5button.png"> | <img width="175" src="https://static.igem.org/mediawiki/2009/6/63/Mgfp5button.png"> | ||
| Line 17: | Line 17: | ||
</div> | </div> | ||
</html><br> | </html><br> | ||
| - | [[Team:Berkeley_Wetlab/Passenger: MGFP | '''The Sticky Protein''']]<br> | + | <center>[[Team:Berkeley_Wetlab/Passenger: MGFP | '''The Sticky Protein''']]</center><br> |
A protein used by mussels to stick to rocks. An underwater bio-glue! | A protein used by mussels to stick to rocks. An underwater bio-glue! | ||
|| <html> | || <html> | ||
Revision as of 02:09, 21 October 2009
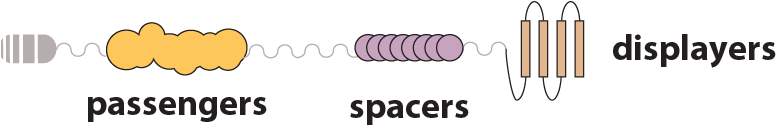
Passengers
Follow any of the links below to see assay information for each of the passengers we made.
|
A protein used by mussels to stick to rocks. An underwater bio-glue! |
The Enzyme |
The scFv |
|
The Tag |
The Heterodimeric Binding Peptide |
The Other Binding Peptide |
Spacers
Multiple spacer elements are typically present in natural display systems suggesting that they are important components of cell surface display systems. However, the precise role of spacers in the functionality of outer membrane proteins has not been extensively characterized. We hoped to characterize the effects of the inclusion of spacer elements within our passenger-display design. There were five spacer domains introduced to our surface display system: INP-repeats, beta roll, beta helix, Gly-Ser repeats and GFP-LVA. These elements are further described below.
1. INP - Repeats-repeated portion of ice nucleation protein (INP) sequence.
2. Beta Roll - protein tertiary structure that is natively found in autotransporters and other outermembrane proteins.
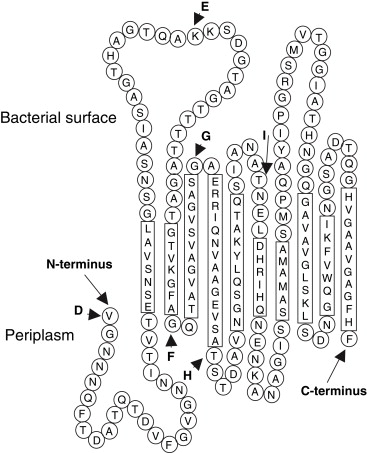
3. Beta Helix - protein helical structures stabilized by hydrogen bonds and protein-protein interactions. The resulting structure contains two to three faces formed by the association of parallel beta strands.
4. Gly-Ser Repeats - a flexible 14-mer amino acid sequence of glycine and serine residues repeated.
5. GFP-LVA - modified green fluorescent protein
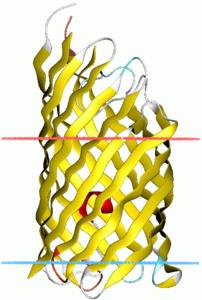
Displayers
A displayer is defined as an outmembrane protein that carries another protein to the extracellular space of the cell.
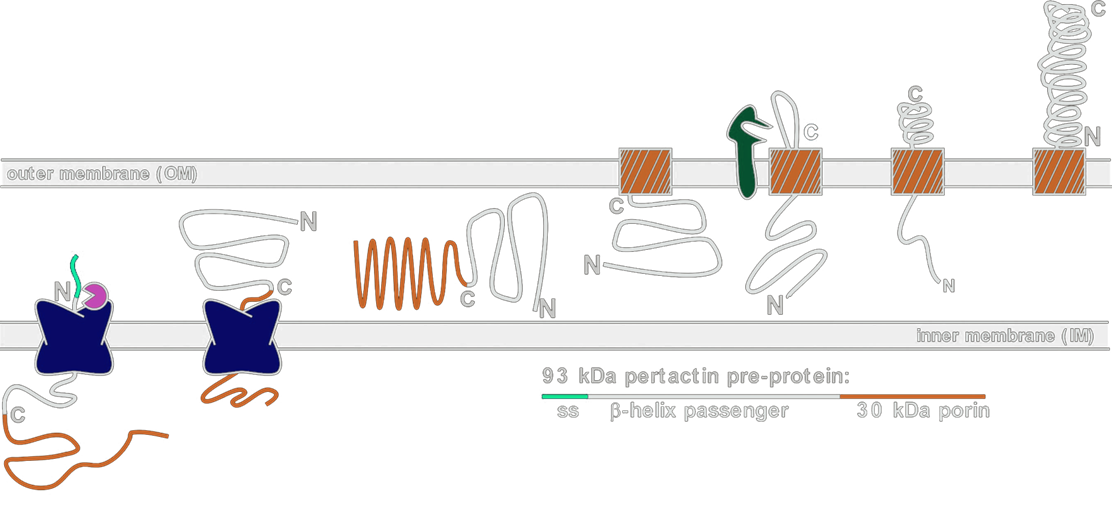
For successful cell surface display of proteins, there must be an effective protein localization mechanism. Gram-negative bacteria such as E. Coli have two membranes, which present a problem for transporting proteins synthesized in the cytoplasm to the outside of the cell. Various transport schemes exist in gram-negative bacteria to effectively localize proteins to the outermembrane. The most common schemes are TypeI, TypeIII, and TypeV secretion.
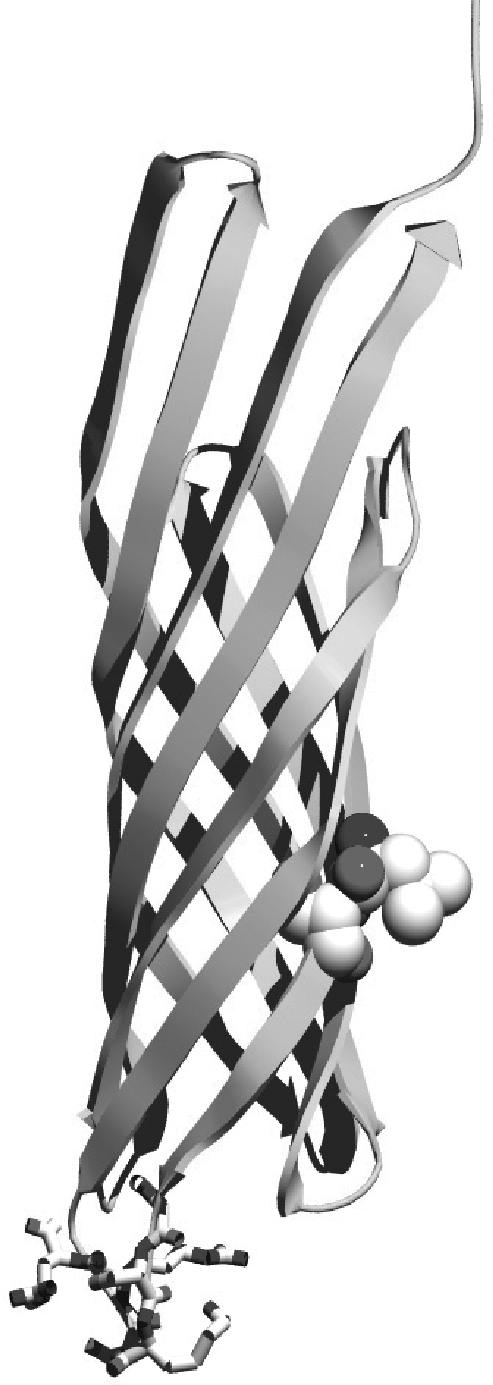
In our display systems, we chose a class of outermembrane proteins called autotransporters that localizes proteins via the TypeV secretion mechanism. Over 700 autotransporters have been sequenced, many of which are used to export virulence factors to the outside of the cell. We decided to harvest this localization system for cell surface display because the outermembrane protein (aka displayer) spontaneously inserts into the outermembrane and pulls the protein it is covalently linked to (aka passenger) into the extracellular space. Moreover, autotransporters are capable of pulling through large proteins, such as enzymes and single-chain variable fragments. We have removed the native passengers in our autotransporter constructs and fused heterologous passengers of interest to the N-terminus of these autotransporters.
As depicted in the diagram above, autotransporter transport begins with localization to the periplasm via the Sec secretion pathway. The translocated protein remains unfolded in the periplasm until it inserts into the outermembrane by forming a beta barrel with its C-terminal 250-300 amino acyl residues. The N-terminus of the protein (containing our passenger of interest) is then pulled through the barrel to the outside of the cell. Passengers of displayers are often cleaved for extracellular secretion. In our systems, however, we removed the signal sequence that signals for peptide cleavage so our passengers remain attached to the transmembrane displayer protein.
In constructing our parts, we looked into a broad range of autotransporters, some well characterized and others putative, to explore the spectrum of display machinery and to establish the functionality of novel autotransporters for cell surface display.
1. azo1653 AtD (putative) - organism Azoarcus sp. (strain BH72)
Autotransporter type: AT-1 family
2. OprF AtD - organism Pseudomonas fluorescens
Structure: an 8-stranded beta barrel in the outermembrane
3. Cl02365 AtD (putative) - organism Neisseria meningitidis
Autotransporter type: AT-1 family
4. VtaA11 AtD - organism Haemophilus parasuis
Autotransporter type: AT-2 family
5. Hag AtD - organism Moraxella catarrhalis
Autotransporter type: dimeric family
Structure: 200kDa protein with 10-stranded beta barrel
6. Pcryo_1225 AtD (putative) - organism Psychrobacter cryohalolentis
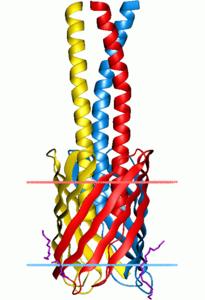
7. Hia AtD - organism Haemophilus influenzae
Autotransporter domain: trimeric family
Structure: modular segments containing repeats of structurally distinct domains
8. upaG_short - organism Escherichia Coli
Autotransporter type: trimeric family
9. espP(beta) - organism Escherichia coli
Structure: 12-stranded beta barrel
10. ehaB - organism Escherichia coli
Features: primary sequence alone is sufficient for crossing the bacterial membrane
11. TshA - organism Escherichia coli
Autotransporter type: serine protease subfamily (because of the 7AA serine protease motif)
12. VirG(IcsA) - organism Shigella flexneri
13. YuaQ AtD (putative) - organism Escherichia coli
Features: bears sequence similarity to the confirmed autotransporters AIDA and Ag43
14. AIDA-I - organism Escherichia Coli Features: identified to be similar to IgA1, the first autotransporter used for surface display. Occurs naturally in the host organism, E. coli, and is a robust tool for surface display
15. Ag43_short - organism Escherichia Coli MG1655
Features: expression of Ag43 is evenly distributed around the bacterial cell
Structure: 14 antiparallel beta strands each composed of about 12AA residues
16. eCPX (circularly permuted OmpX)- organism Escherichia Coli
Features: protein is an enhanced CPX variant located in the outermembrane that joins the N- and C-termini of OmpX.
17. CPG_L2 (circularly permuted OmpG)- organism Escherichia Coli
Features: protein is circularly permuted with its backbone opening in loop 2, allowing both the N- and C- termini to be present in the extracellular space.
18. CPG_L6 (circularly permuted OmpG)- organism Escherichia Coli
Features: protein is circularly permuted with its backbone opening in loop 6, allowing both the N- and C- termini to be present in the extracellular space.
Some of these proteins are putative autotransporters that have sequence homology to confirmed autotransporters. We chose these proteins because we wanted to test their functionality and expand the range of displayers available for surface display.
References
Pina, S et al. Trimeric Autotransporters of Haemophilus parasuis: Generation of an Extensive Passenger Domain Repertoire Specific for Pathogenic Strains. J Bacteriol. January 2009; 191(2): 576–587. Available Online: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2620822/ (Accessed: 20 October 2009).
Kostakioti, M et al. Functional analysis of the Tsh autotransporter from an avian pathogenic Escherichia coli strain. Infect Immun. October 2004;72(10):5548-54. Available Online: http://www.ncbi.nlm.nih.gov/pubmed/15385451?ordinalpos=5&itool=EntrezSystem2.PEntrez.Pubmed.Pubmed_ResultsPanel.Pubmed_DefaultReportPanel.Pubmed_RVDocSum (Accessed: 20 October 2009).
http://www.uniprot.org/uniprot/?query=H81868&sort=score
http://pfam.sanger.ac.uk/family?acc=PF05736
http://www.genome.jp/dbget-bin/www_bget?azo:azo1653
http://www.genome.jp/dbget-bin/www_bget?pcr:Pcryo_1225
http://www.uniprot.org/uniprot/Q9JMS3
 "
"