Team:Minnesota/Modeling
From 2009.igem.org
| Home | The Team | The Project | Submitted Parts | Modeling | SynBioSS Designer | Parts Characterization | Experiments |
|---|
Contents |
Our Modeling Goals
We want to improve the user interface for SynBioSS (see below), which was developed for last year's competition and also create mathematical models of our experimental results. Our computational output will guide future research and is the key to determining what is happening at the molecular level of our logical AND gate.
Computational Tools
Synthetic Biology Software Suite (SynBioSS)
The entire software suite can be found via the SynBioSS home page. The software suite is open source that runs on Windows, Macintosh and Linux. It does not require command-line programming and thus facilitates the modeling of biochemical reactions.
SynBioSS Designer
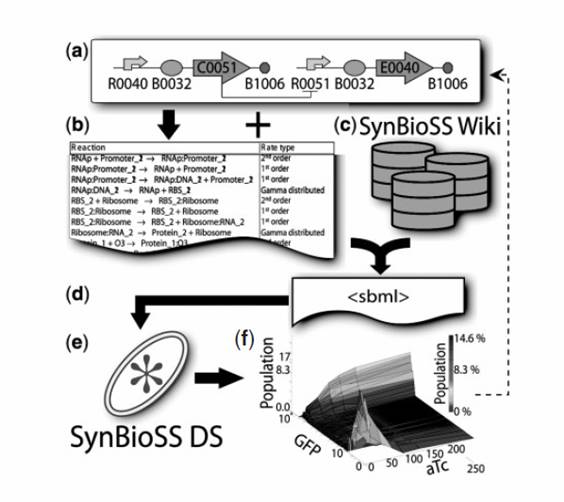
SynBioSS Designer is an application for the automatic generation of sets of biomolecular reactions. This software allows a user to input the molecular parts involved in gene expression and regulation (e.g. promoters, transcription factors, ribosomes, etc.) The software then generates complete networks of reactions that represent transcription, translation, regulation, induction and degradation of those parts. To facilitate the creation of detailed kinetic models of synthetic gene networks composed of BioBricks, we have adapted SynBioSS Designer to automatically generate a kinetic model from a construct composed entirely of BioBricks. A NetCDF or SBML file is generated for simulations.
Our goal for this year's iGEM competition is to overhaul the user interface for the SynBioSS Designer. After updating the modeling component to account for polycistronic strands of Coding DNA, our primary goal is to integrate a search box that will allow users to query the BioBricks database and retrieve relevant characteristics. Currently, the user must manually input all information about every BioBrick (e.g. for promoters, the user must input whether the promoter is constitutively on or off, whether the promoter has operator sites, where these operator sites are located, etc.). Once completed, users will be able to search the Registry for BioBricks that fit search parameters, pull them into a convenient and user-friendly interface in SynBioSS, and export a NetCDF or SBML file for computation, constructing the entire complex biological system of reactions in a matter of minutes.
SynBioSS Wiki
The inaccessibility of requisite kinetic data complicates the generation of detailed mechanistic models. We address this barrier by creating a web accessible database curated by users in a “Wiki” format. SynBioSS Wiki is a significant extension of the open-source Mediawiki software. The Wiki stores reaction kinetic data in a formatted and searchable scheme with references to the relevant literature. This framework allows for the input of reactions whose rates are described either by elementary first & second order rate equations or any arbitrarily complex rate equation defined using MathML (e.g. Hill type reactions). Reactions may be searched via participating molecules which may be proteins, DNA sequences, small molecules, etc. Once located, reactions of interest (along with their associated kinetic data) may be collected. The completed model may be exported in SBML format for additional editing or simulation.
It is also through the SynBioSS Wiki databases that SynBioSS Designer can access and proliferate kinetic information related to the simulation of BioBricks, thus extending the utility of the database for the benefit of the greater modeling community. To jumpstart the process, we have entered the known biomolecular interactions in the expression and regulation of well-studied operons, such as the lactose, the tetracycline and the arabinose operons.
Mathematical Modeling
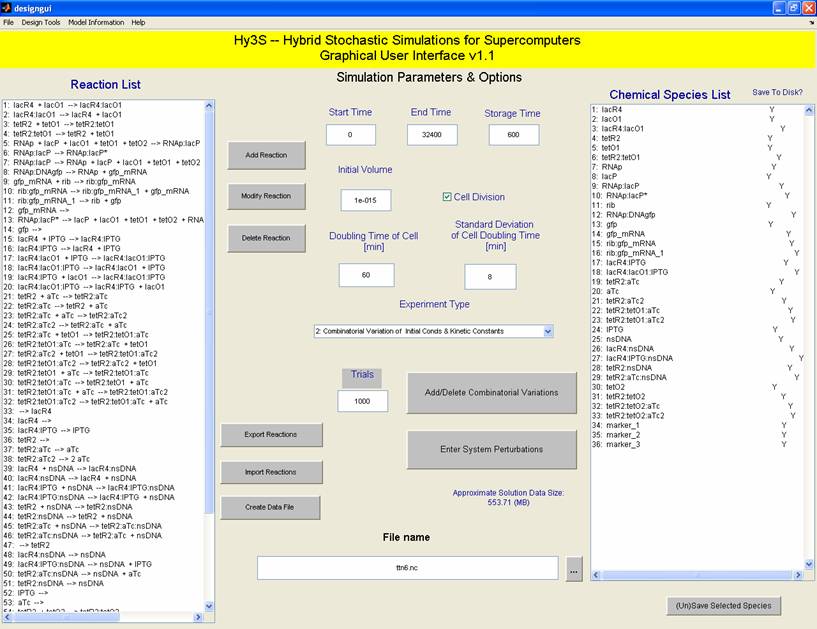
It is important to be able to observe the fidelity of the AND gate in silico as well as in vivo. Using our in-house software suite, SynBioSS, we used networks of sixty or more reactions to model the AND gate with different constructs and mutations. Similar to the experimental procedures, we quantified the fidelity of our AND gate with GFP. We also used a MATLAB Graphical User Interface (GUI) to analyze our experimental data. We used our empirical data from the wet lab to perfect our model.
Some Basic Modeling
We are working with four different combinations of lac andtet (L and T, respectively) operators in our promoter design project. These constructs are: T--, TT-, TTL and -T-. Here, we will demonstrate a little basic modeling we have done of the TTL construct using SynBioSS MATLAB and a design GUI.
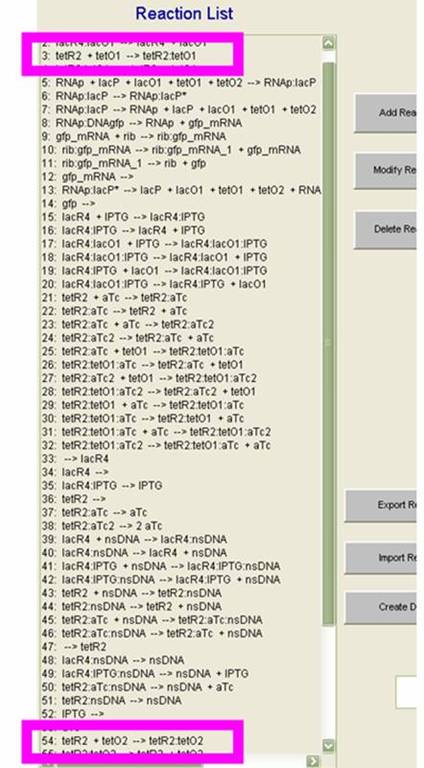
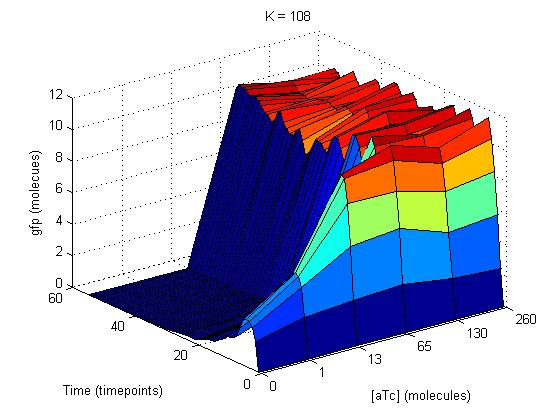
On the SynBioSS wiki, the TTL model is available for download in a .nc format. Upon opening this with the MATLAB GUI, the list of 65 reactions that constitute the repression and induction of transcription. In this model, there are two tet operators TO1 and TO2 and one lac operator LO1. The reactions included in this list account for the repression and induction of transcription in the absence and presence of aTc and IPTG, respectively. In this simple model, we examined reactions 3 and 54, which involve the tet operators being bound by the repressor. (These reactions are shown in the GUI on the left.) We varied the reactions constants from the original 108 as low as 0 and as high as 109 to see if the fidelity of the AND gate could depend on the reaction rates for repression. We quantified our results by counting molecules of GFP. This not only gave us a clearer idea of what was happening at the molecular level at this promoter site, but also served as a preparation for matching the experimental results computationally.
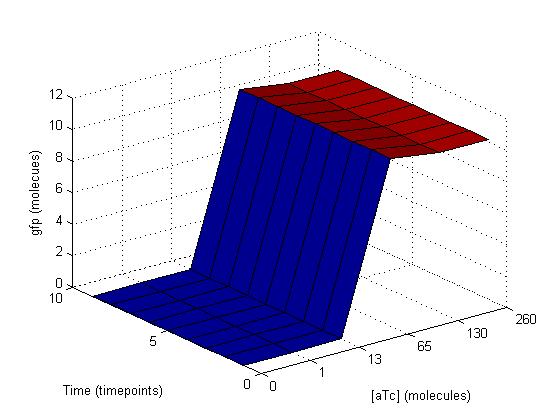
The initial results of our modification of the reaction constants were graphs that showed a steady increase in GFP with time and aTc concentration that eventually plateaued and held GFP constant around 12 molecules. At the plateau as you can see on the graph on the below top left, the gfp oscilated with time despite aTc concentration because the cells were splitting every hour. To smooth out this oscillation, we chose timepoints when the cells were not doubling and graphed gfp again with respect to time and aTc concentration. This graph is shown at the below top right.
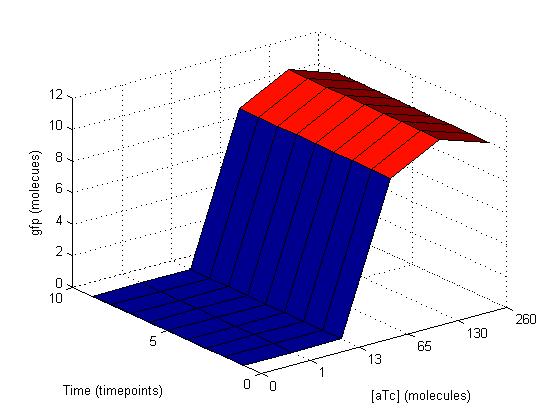
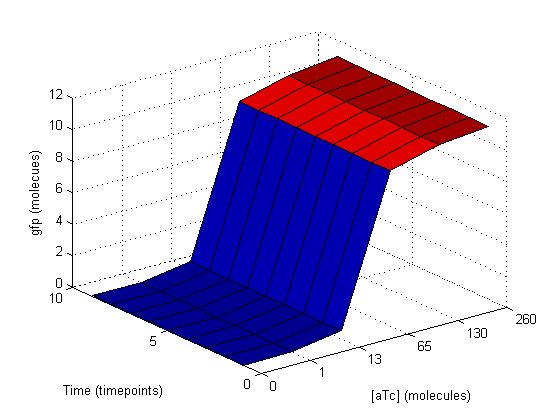
These similar graphs below with k=109 (bottom left) and 106 (bottom right) reinforce the idea of biological complexity. The fidelity of the AND gate does not appear to be dependent on these two reactions alone.
Of the numerous other reactions included in this model, four others involved the binding of one or two molecules of aTc to the tet repressor and thereby inducing transcription. So we decided to investigate the effects of different reaction constants for these reactions on GFP production.
 "
"