Team:SJTU-BioX-Shanghai/Project network
From 2009.igem.org
Pnstontale (Talk | contribs) m |
Pnstontale (Talk | contribs) m |
||
| Line 41: | Line 41: | ||
| - | Next, go to [[Team:SJTU-BioX-Shanghai/Modelling#bannerTop| | + | Next, go to [[Team:SJTU-BioX-Shanghai/Modelling#bannerTop|Modelling]]. |
{{Template:SJTU09_enddiv}} | {{Template:SJTU09_enddiv}} | ||
Revision as of 05:28, 21 October 2009

Project introduction. Inspired by the natural regulator of circadian bioclock exhibited in most eukaryotic organisms, our team has designed an E.coli-based genetic network with the toxin-antitoxin system so that the bacterium oscillates between two states of dormancy and activity (more...)
Contents |
Network construction
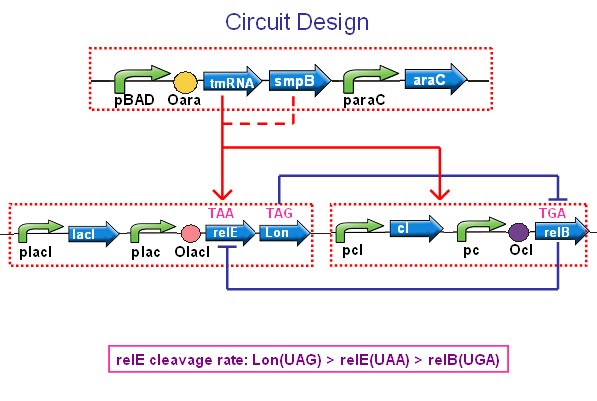
Overview of whole circuit and gene relationship:
Our whole network can be devided into three parts:
- RelE and Lon-toxin system;
- RelB –antitoxin system;
- tmRNA-rescue system;
Part I: RelE and Lon-toxin system: Hypnos Curse
In this part, the expressions of Lon protease and RelE toxin is under the regulation of Lac promoter. At first, LacI-inhibiting protein is continously expressed, thus blocks the transcription of Lon and RelE. Once we use IPTG to induce the genetic circuit, the free LacI will be inactive, which eventually causes coexpression of RelE and Lon.
This part can be also called: Hypnos’ Curse. The relE toxin is an RNase that preferentially cleaves mRNAs binding to the ribosomal A site at the second position of stop codons, so the excessive protein accumulation of RelE toxin will severely inhibit translation and prevent colony formation. Meanwhile, the Lon protease will hydrolysis RelB-antitoxin.
Most importantly, the stop condon of RelE gene is mutated from UGA(wild type) to UAA, which cause moderate self-inhibition of RelE protein. The result of self-inhibition can prevent RelE from accumulation.
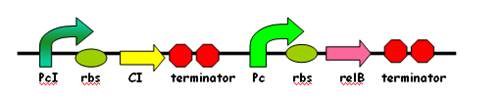
Part II:RelB-antitoxin system: Hypnos Mercy
Part II gives the chance for E.coli to revive from dormancy. The RelB-antitoxin is under control of Lamda CI promoter, which means we can use L-tryptophan to induce expression of RelB-antitoxin. The Lamda CI promoter can also be induced by high temperature.
This part can be also called: Hypnos’ Mercy. The growth of E.coli can be restored by coexpression of the cognate relB antitoxin, which can form a heterotetrameric (relB-relE)2 structure when binding with relE. Thus,this structure is too large to fit into the A site, so the toxic relE can be neutralized. Overexpression of relB restored protein synthesis and colony formation.
Part III: tmRNA-rescue sytem
The tmRNA plays a very important role in rescuing system.We construct tmRNA under the control of PBAD, which can be induced by L-arabinose. SmpB is a small protein, as well as the coeffector of tmRNA, assisting tmRNA to bind stalled ribosomes. Therefore, a constitutive promoter(PsmpB) is designed to express SmpB.
The tmRNA system orchestrates three key biological functions: (1) recognition and rescue of ribosomes stalled on aberrant mRNAs, (2) disposal of the causative defective mRNAs, and (3)addition of a degradation tag to ribosome-associated protein fragments for directed proteolysis.
Next, go to Modelling.
 "
"