Team:Tsinghua/Background
From 2009.igem.org
Contents |
Background
Gene Therapy
Gene therapy is defined as the introduction of genes into tissues or cells via gene transfer, with the purpose of deriving a therapeutic or preventative benefit from the function of these genes. It is the insertion of genes into an individual's cells and tissues to treat a disease, such as a hereditary disease in which a deleterious mutant allele is replaced with a functional one[1]. Although the technology is still in its infancy, it is one of the most promising and active research fields in medicine[1,2]. Antisense therapy is not strictly a form of gene therapy, but is a genetically-mediated therapy and is often considered together with other methods[1].
Recently the scientific breakthroughs in the genomics field made gene therapy one of the most rapidly advancing fields of biotechnology and we can have a better understanding of the role of genes in disease with great promise for treating inherited and acquired diseases. the absence or inappropriate presence of a protein results in many human diseases. The protein could then be administered to patients in order to compensate for its absence. Nowadays gene therapy is the ultimate method of protein delivery. With the moving steps from the laboratory into the clinic, several issues have been brought in a central role of the development of this technology: gene identification, gene expression and gene delivery. More and more disease-related genes are found one by one, which still growing in a rapid speed. Broader clinical application of related genes are also being utilized to achieve immune activating agents with an acurate site and applying in immune fields. http://cmbi.bjmu.edu.cn/cmbidata/therapy/index/index.htm
Vectors in Gene Therapy
Despite substantial progress, a number of key technical issues need to be resolved before gene therapy can be safely and effectively applied in the clinic, and an ideal gene delivery vector is one of the bottlenecks in gene therapy application[3]. Generally, vectors applied in gene therapy can be classified into viral or non-viral.
Viral Methods
All viruses bind to their hosts and introduce their genetic material into the host cell as part of their replication cycle. This genetic material contains basic 'instructions' of how to produce more copies of these viruses, hijacking the body's normal production machinery to serve the needs of the virus[1].
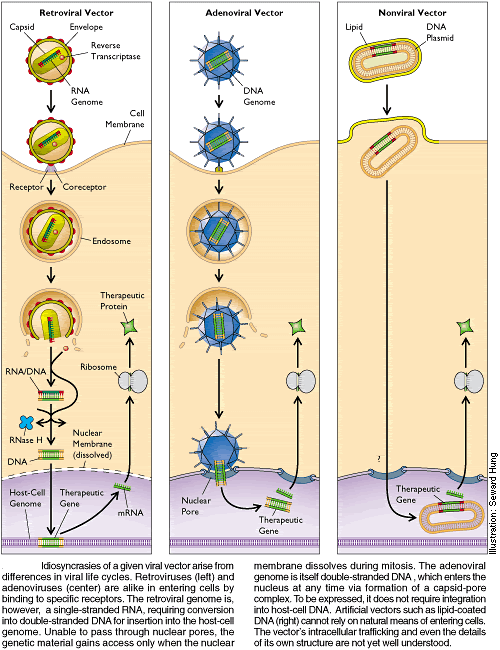
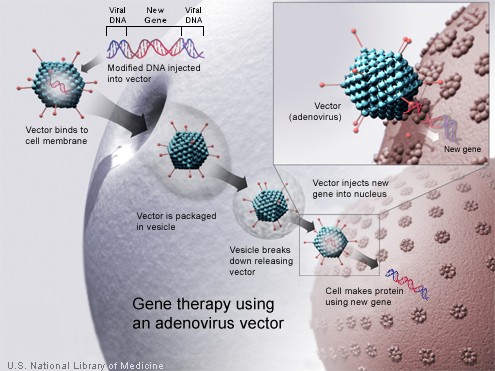
Adenovirus. Adenoviruses are viruses that carry their genetic material in the form of double-stranded DNA. They cause respiratory, intestinal, and eye infections in humans (especially the common cold). When these viruses infect a host cell, they introduce their DNA molecule into the host. The genetic material of the adenoviruses is not incorporated (transient) into the host cell's genetic material. The DNA molecule is left free in the nucleus of the host cell, and the instructions in this extra DNA molecule are transcribed just like any other gene.
Adeno-associated virus. Adeno-associated viruses, from the parvovirus family, are small viruses with a genome of single stranded DNA. The wild type AAV can insert genetic material at a specific site on chromosome 19 with near 100% certainty. But the recombinant AAV, which does not contain any viral genes and only the therapeutic gene, does not integrate into the genome. Instead the recombinant viral genome fuses at its ends via the ITR (inverted terminal repeats) recombination to form circular, episomal forms which are predicted to be the primary cause of the long term gene expression.
Retroviruses. The genetic material in retroviruses is in the form of RNA molecules, while the genetic material of their hosts is in the form of DNA. When a retrovirus infects a host cell, it will introduce its RNA together with some enzymes, namely reverse transcriptase and integrase, into the cell. This RNA molecule from the retrovirus must produce a DNA copy from its RNA molecule before it can be integrated into the genetic material of the host cell.
Envelope protein pseudotyping of viral vectors The viral vectors described above have natural host cell populations that they infect most efficiently. Retroviruses have limited natural host cell ranges, and although adenovirus and adeno-associated virus are able to infect a relatively broader range of cells efficiently, some cell types are refractory to infection by these viruses as well. Attachment to and entry into a susceptible cell is mediated by the protein envelope on the surface of a virus[1].
Non-Viral Methods
Non-viral methods present certain advantages over viral methods, with simple large scale production and low host immunogenicity being just two. Previously, low levels of transfection and expression of the gene held non-viral methods at a disadvantage; however, recent advances in vector technology have yielded molecules and techniques with transfection efficiencies similar to those of viruses[1].
Naked DNA. This is the simplest method of non-viral transfection. Clinical trials carried out of intramuscular injection of a naked DNA plasmid have occurred with some success; however, the expression has been very low in comparison to other methods of transfection. In addition to trials with plasmids, there have been trials with naked PCR product, which have had similar or greater success. This success, however, does not compare to that of the other methods, leading to research into more efficient methods for delivery of the naked DNA such as electroporation, sonoporation, and the use of a "gene gun", which shoots DNA coated gold particles into the cell using high pressure gas.
Oligonucleotides. The use of synthetic oligonucleotides in gene therapy is to inactivate the genes involved in the disease process. There are several methods by which this is achieved. One strategy uses antisense specific to the target gene to disrupt the transcription of the faulty gene. Another uses small molecules of RNA called siRNA to signal the cell to cleave specific unique sequences in the mRNA transcript of the faulty gene, disrupting translation of the faulty mRNA, and therefore expression of the gene.
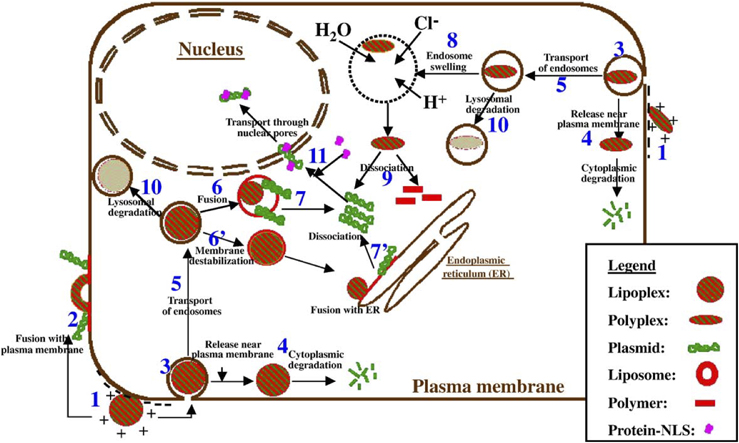
Lipoplexes and polyplexes. To improve the delivery of the new DNA into the cell, the DNA must be protected from damage and its entry into the cell must be facilitated. To this end new molecules, lipoplexes and polyplexes, have been created that have the ability to protect the DNA from undesirable degradation during the transfection process.
Hybrid methods Due to every method of gene transfer having shortcomings, there have been some hybrid methods developed that combine two or more techniques. Virosomes are one example; they combine liposomes with an inactivated HIV or influenza virus. This has been shown to have more efficient gene transfer in respiratory epithelial cells than either viral or liposomal methods alone. Other methods involve mixing other viral vectors with cationic lipids or hybridising viruses.
Dendrimers A dendrimer is a highly branched macromolecule with a spherical shape. The surface of the particle may be functionalized in many ways and many of the properties of the resulting construct are determined by its surface. In particular it is possible to construct a cationic dendrimer, i.e. one with a positive surface charge. When in the presence of genetic material such as DNA or RNA, charge complimentarity leads to a temporary association of the nucleic acid with the cationic dendrimer. On reaching its destination the dendrimer-nucleic acid complex is then taken into the cell via endocytosis.
Frontiers in Gene Therapy Research
Induced Pluripotent Stem Cell (iPS) and Gene Delivery System
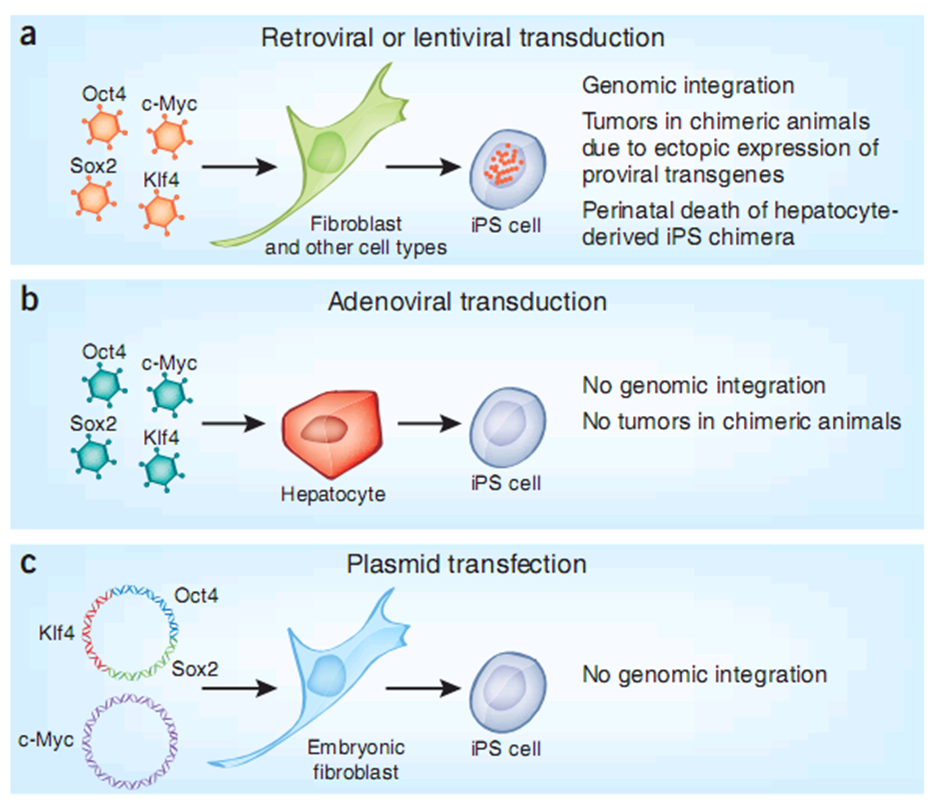
In 2006, Shinya Yamanaka et al showed that mouse embryonic and adult fbroblasts acquire properties similar to those of embryonic stem (ES) cells after retrovirally introducing genes encoding four transcription factors, namely Oct3/4, Sox2, Klf4, and c-Myc (known as iPS). In 2007, mouse and human iPS cells were generated[4].
In 2008, Qiao Zhou et al identify a specific combination of three transcription factors (Ngn3, Pdx1 and Mafa) that reprograms differentiated pancreatic exocrine cells in adult mice into cells that closely resemble beta-cells. These reprogrammed cells express genes essential for b-cell function and can ameliorate hyperglycaemia by remodelling local vasculature and secreting insulin. This approach implies a promising application of reprogramming on diabetes therapy[5].
The booming research on iPS and the subsequent in vivo reprogramming implicate an enormous future. In addition to the obstacles of transformation risks and low efficiency, specifically delivering certain transcription factors into the targeted cells is another problem before the medical and pharmaceutical applications of reprogramming.
Therapeutic microRNA Delivery
Therapeutic strategies based on modulation of microRNA (miRNA) activity hold great promise due to the ability of these small RNAs to potently influence cellular behavior[6].
Cancer Gene Therapy
As a primary threat of human health, cancer causes about 13% of all human deaths[7]. According to the American Cancer Society, 7.6 million people died from cancer in the world during 2007[8]. Current treatments often have far reaching negative side effects[9]. The systemic toxicity of chemotherapy regimens still often result in acute and delayed nausea, mouth ulcerations and mild cognitive impairments[10]. Squamous cell carcinoma of the head and neck afflicts approximately 500,000 patients per year. The first-line treatment, chemotherapy, induces a response in only 30–40% of patients, and tumors frequently recur. Alternative cancer therapy approaches have been based on oncolytic viruses, which selectively attack tumor, but not normal, cells. One of these, ONYX-015, was initially tested in a Phase I clinical trial in April 1996. In this issue, Khuri et al. report that ONYX-015, when combined with chemotherapy, promotes tumor regression in patients with recurrent squamous cell cancer of the head and neck[11].
References
[1] http://en.wikipedia.org/wiki/Gene_therapy
[2] SM Selkirk. Gene therapy in clinical medicine. Postgraduate Medical Journal.2004;80:560-570; doi:10.1136/pgmj.2003.017764
[3] RC Mulligan. The basic science of gene therapy. Science, 1993, Vol 260, Issue 5110, 926-932.
[4] Shinya Yamanaka. A Fresh Look at iPS Cells. Cell (2009), 137:13-17.
[5]Qiao Zhou, Juliana Brown, Andrew Kanarek, Jayaraj Rajagopa and Douglas A. Melton. In vivo reprogramming of adult pancreatic exocrine cells to beta-cells. Nature (2008), 455:627-633.
[6] Janaiah Kota, Raghu R. Chivukula, Kathryn A. O’Donnell, Erik A. Wentzel, Chrystal L. Montgomery, Hun-Way Hwang, Tsung-Cheng Chang, Perumal Vivekanandan, Michael Torbenson, K. Reed Clark, Jerry R. Mendell,and Joshua T. Mendell. Therapeutic microRNA Delivery Suppresses Tumorigenesis in a Murine Liver Cancer Model. Cell. 2009, 137. 1005–1017.
[7] WHO (February 2006). "Cancer". World Health Organization. http://www.who.int/mediacentre/factsheets/fs297/en/. Retrieved on 2007-06-25.
[8] American Cancer Society (December 2007). "Report sees 7.6 million global 2007 cancer deaths". Reuters. http://www.reuters.com/article/healthNews/idUSN1633064920071217. Retrieved on 2008-08-07.
[9] Peter Sinnaeve, Olivier Varenne, Désiré Collen, and Stefan Janssens. Gene therapy in the cardiovascular system: an update. Cardiovasc Res, Dec 1999; 44: 498 - 506.
[10] Chemotherapy and you: A guide to self-help during cancer treatment. National Institutes of Health Web site. Available at: http://www.cancer.gov/PDF/b21d0a74-b477-41ec-bdc0-a60bbe527786/chemoandyou.pdf. Accessed July 31, 2006.
[11] Gene therapy scores against cancer. NATURE MEDICINE, VOLUME 6, NUMBER 8, AUGUST 2000
Diagram Ref
[1]http://images.google.cn/imglanding?imgurl=http://www.acceleratingfuture.com/michael/blog/images/Gene_therapy.jpg&imgrefurl=http://www.acceleratingfuture.com/michael/blog/%3Fp%3D455&usg=__jvPXcCSXLCqT6xsyFwq5NQfjJE0%3D&h=371&w=495&sz=56&hl=zh-CN&um=1&tbnid=FfNmLhJhG4h7NM:&tbnh=97&tbnw=130&prev=/images%3Fq%3Dgene%2Btherapy%26hl%3Dzh-CN%26sa%3DN%26um%3D1%26newwindow%3D1&q=gene+therapy&sa=N&um=1&newwindow=1&start=0#start=0
[2]http://cmbi.bjmu.edu.cn/cmbidata/therapy/index/index.htm
[3]http://images.google.cn/imglanding?imgurl=http://www.nature.com/mt/journal/v11/n3/images/mt200545f1.jpg&imgrefurl=http://www.nature.com/mt/journal/v11/n3/fig_tab/mt200545f1.html&usg=__16PMODxAS0ot0uFh5qHjoNgcvmE%3D&h=443&w=739&sz=215&hl=zh-CN&um=1&tbnid=b9vKbApcf9TRPM:&tbnh=85&tbnw=141&prev=/images%3Fq%3DLipoplexes%26hl%3Dzh-CN%26um%3D1%26newwindow%3D1&q=Lipoplexes&um=1&newwindow=1&start=0#start=0
[4]william e Lowry & Kathrin Plath. The many ways to make an iPS cell. Nature, 26:1246-1248.
 "
"