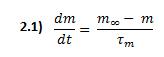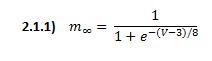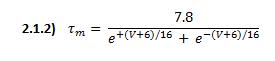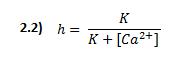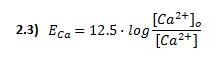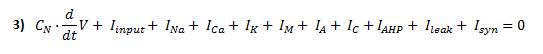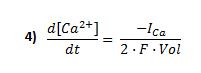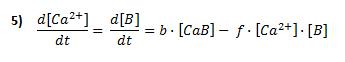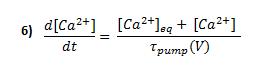Team:Valencia/OurModel
From 2009.igem.org
Our Model
- Modelling Ionic Current Flow through VDCCs
If we assume that the whole calcium currents occur through this calcium channels and that the instantaneous current-voltage relation is linear, we can describe the ionic current ICa by the Ohm's law:
Where g is the conductance associated with the channel, V is the transmembrane potential and ECa is the Nerst potential, related to the different ionic concentration inside and outside the cell.
Considering that these channels are only permeable to calcium and have two states -open or closed-, the total conductance associated with the population of VDCCs can be expressed as the maximal conductance ![]() times the fraction of all channels that are open. This fraction is determined by hypothetical activation and inactivation variables m and h, which depend on voltage and time:
times the fraction of all channels that are open. This fraction is determined by hypothetical activation and inactivation variables m and h, which depend on voltage and time:
![]() is the steady-state value of m and
is the steady-state value of m and ![]() is the time constant. They are defined functions of voltage:
is the time constant. They are defined functions of voltage:
K is the halfway inactivation concentration and [Ca2+]o is the constant extracellular calcium concentration.
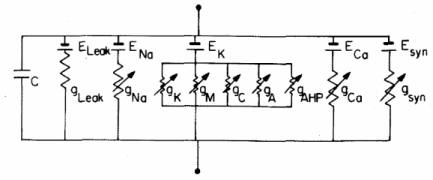
Now, we have to model how transmembrane potential changes in time. To do this, we can consider the following membrane-equivalent electrial circuit, where all ionic currents involved in initiation and propagation of the action potential are represented:
We can know the transmembrane potential at any time after applying an electrical input by solving this equation:
However, we have assumed that our stimulus triggers the excitatory post-synaptic potential (EPSP), so it's not necessary to solve the previous equation. But modelling the calcium influx is only the first step...
- Modelling Free Intracellular Calcium Concentration
The change in free intracellular calcium concentration is mostly due to the influx of calcium ions described above, but there are several factors which also contribute. For instance, we have considered calcium buffers and calcium remove by membrane pumps.
Calcium current
The relation between the calcium inward current ICa and the change in intracellular calcium concentration is given by:
F is the Faraday's constant, [Ca2+] is the calcium concentration just below the plasma membrane and Vol is the cell volume considered.
At this point we have taken into account the presence of calcium buffers such as calmodulin, calcineurin, calbindin, and other ones in the cell. To make the model easier, we have assumed that calcium binds to a single binding site on a single buffer as it is expressed here:
f and b are the forward and backward rates of the binding reaction:
Once the buffering system has reduced the amount of free intracellular calcium, the remaining calcium ions must be removed from the cell in order to maintain calcium homeostasis. We have described the behaviour of calcium pumps by the following first-order equation:
Where [Ca2+]eq is the equilibrium concentration of the pump, [Ca2+] is the calcium concentration in the shell just below the membrane and tpump is the pump's time constant, which depends on voltage:
We have neglected the intracellular diffusion of calcium due to the different concentrations between the inner perimembranal area and deeper areas of the citoplasm, we have considered that the calcium release from intracellular organelles (for instance, endoplasmic reticulum and mitochondria) may reduce these concentration differences. Thus, we assume the calcium concentration just below the plasma membrane as whole intracellular calcium.
 "
"