Team:Valencia/WetLab/YeastTeam/Results
Experimental results
Our ultimate goal was to make a bio-screen made with cellular pixels (LECs). But, before to be able to build this iLCD, we had to study the behaviour of one single LEC. Therefore, we focused on the electrical excitation of our transformed yeasts.
This is the behaviour that stands as the cornerstone of our project. With a serie of voltage inputs, we acomplished a series of light emissions. No exciting light was needed, only aequorin, coelenterazine (the prosthetic group) and Ca2+.
graph continu amb dos pics
We have investigated a set of different voltages and times in order to precisely know how is the reaction of our system to electric current. Knowing this, we could control it properly.
We will show and explain here the major results, for a thorough description of the behaviour of the aequorin light emission system please refer to the Characterization page or to the Registry.
Controls
In every little step in a molecular biology laboratory, one has to bear in mind the use of negative controls for every logical step of the hypothesis.
Therefore, we have taken advantage of the different experimental designs that were available: we had several calcium channel knock outs (mid1 and ch1), as well as functional inhibitors of the calcium channels (KCl) and divalent ion quelant (EDTA).
All these designs were used in order to reject the idea of looking to an artifact.
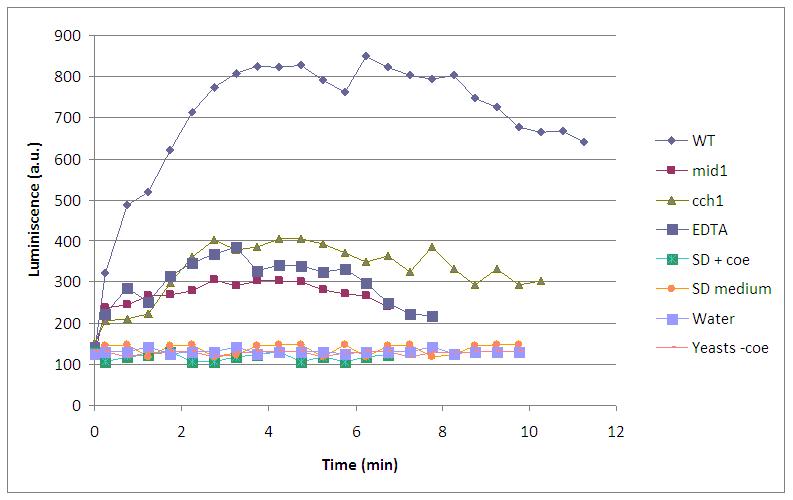
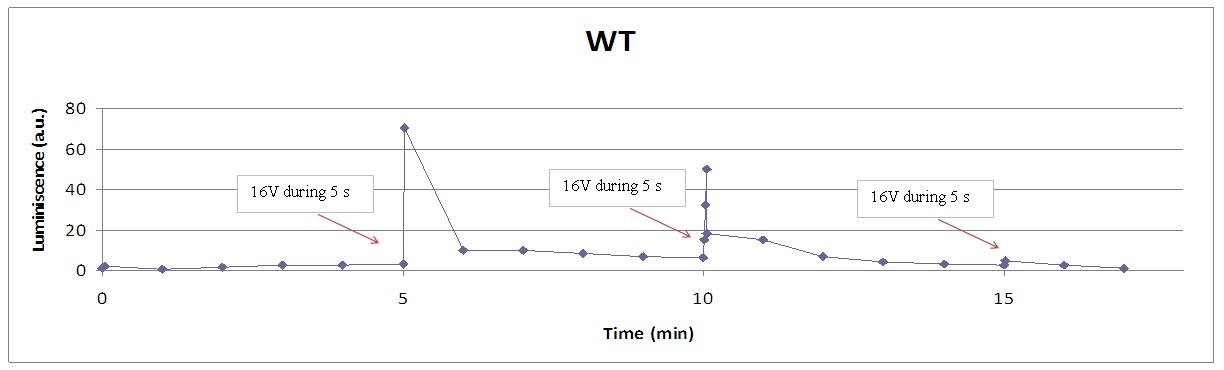
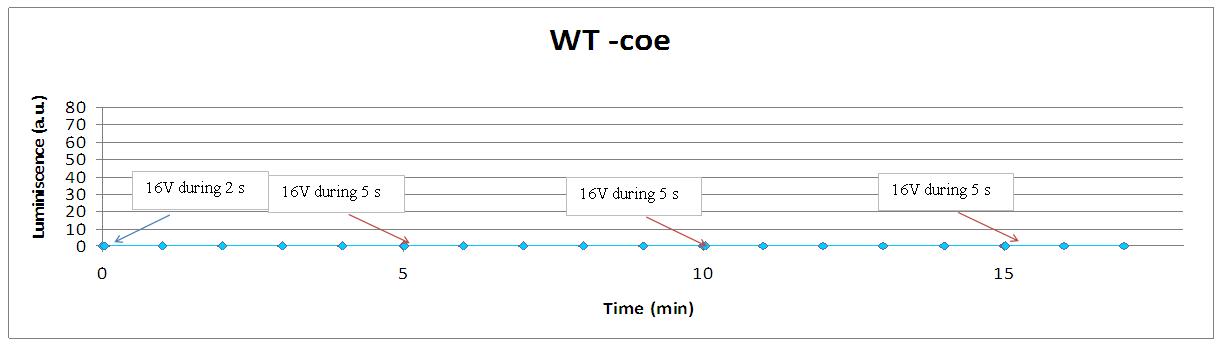
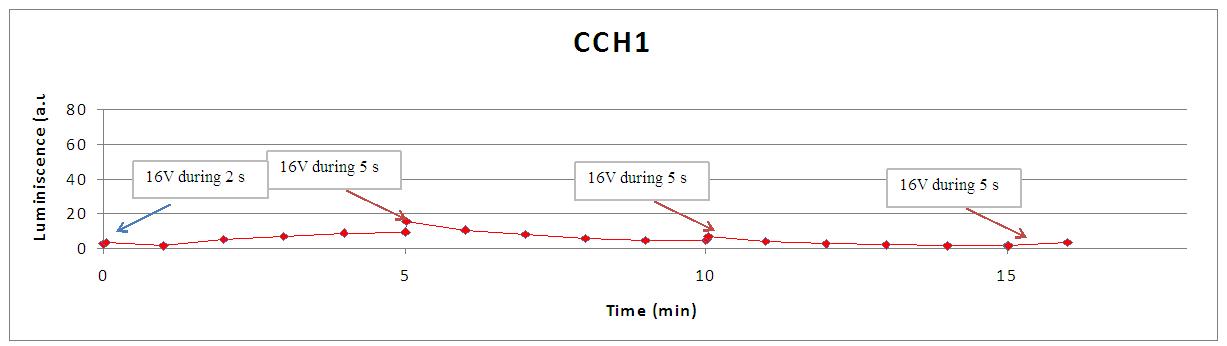
This graph shows the behaviour of a set of designs after the supply of a 4V shock.
- WT is our wild type luminiscent cell, with coelenterazine and fully working calcium channels
- mid1 is a knock out for a calcium channel. Light is not observed because Ca2+ can’t enter into the cell and bind to the aequorin-coelenterazine complex.
- cch1 is another knock out mutant for a calcium channel, so the absence of light can be explainned in the same way.
- EDTA is a divalent ion quelant, so Ca2+ is quenched and not useful for the light emission, although every compound necessary for the reaction is present.
- SD +coe is growth media with coelenterazine, just to be sure that without cells we had no light.
- SD is plain growth media.
- yeast -coe is our wild type luminiscent cell, without coelenterazine, the prosthetic group that is needed for the light emission.
Three kinds of behaviour stem out of this experiment: no light at all, some light emission and full light emission.
- no light at all is the case of SD +coe,SD and yeast -coe where there is a lack of at least one of the compnents of the LEC.
- some light emission is the case of mid1, cch1 and EDTA where all the LEC components are present, but free calcium in the cell is somehow constrained.
- full light emission is the case of WT where all the components of the LEC are presents and calcium channels are fully open.
Voltage
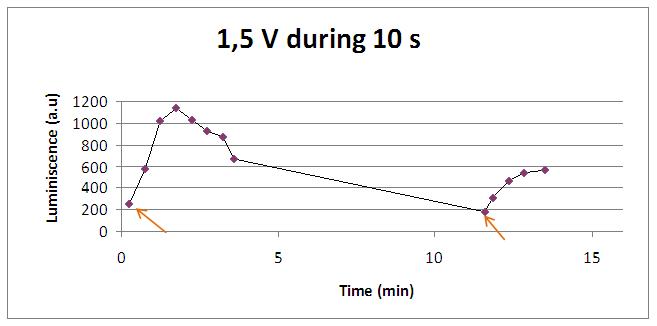
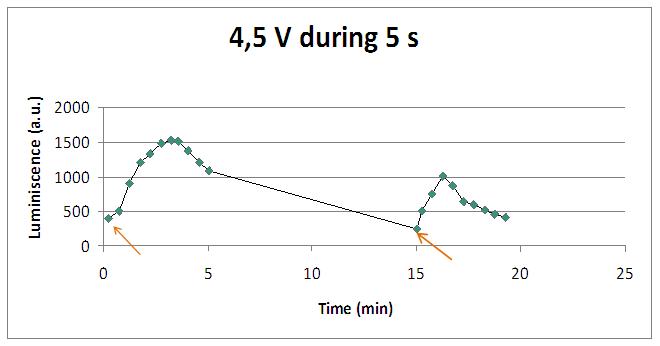
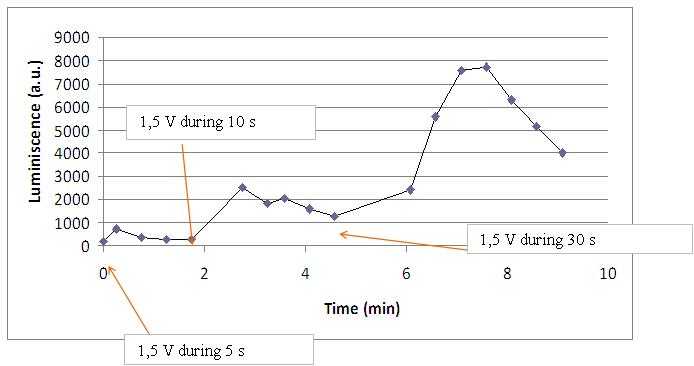
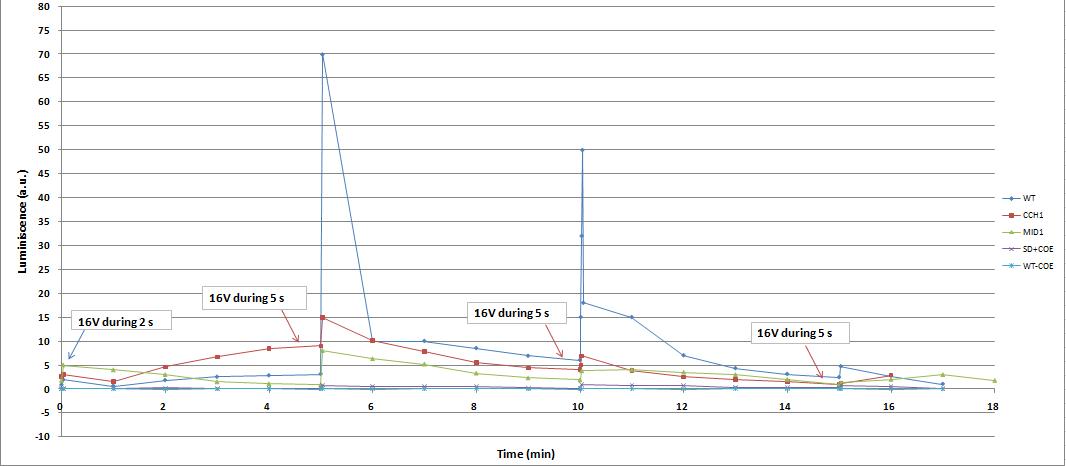
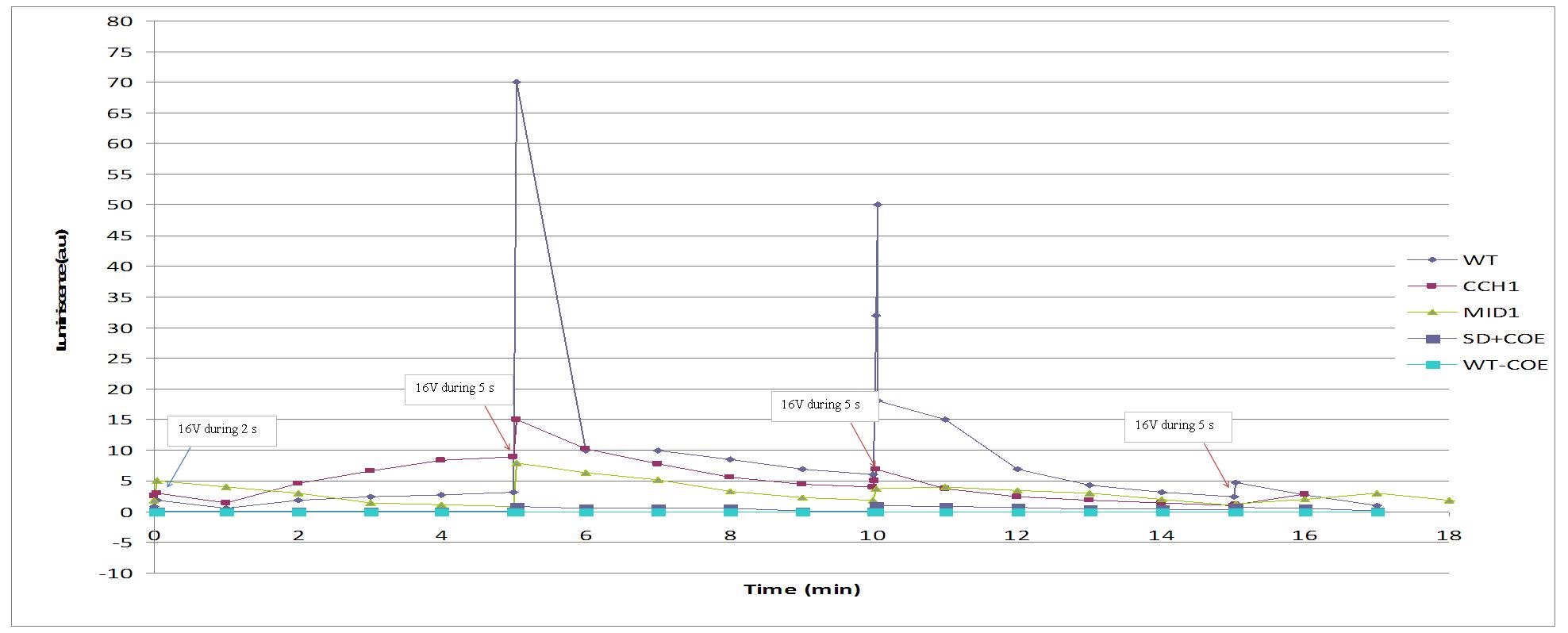
Varying voltage and exciting time, we can have a variety of responses (arrows indicate electric current supply):
Supply time
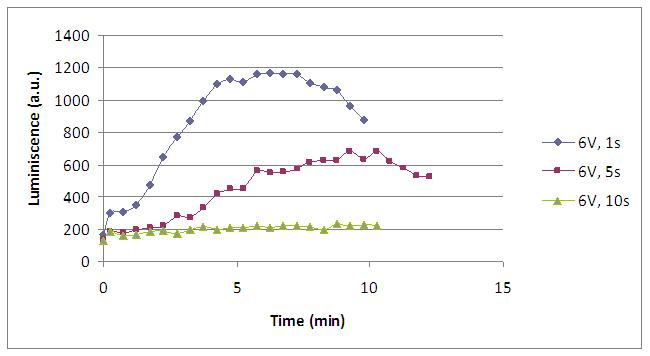
Varying the electric current time supply we can study the influence of this variable in our system. Here we see that if we apply too much time the system is overdosed...
Repetition
Our idea was to have a screen, that is to have several images sequentially, in order tos ee animated pictures. So we studied the different voltage times in a sequence of power applications to one LEC.
Refreshing rate
Finally, we were looking for the shortest refreshing rate as possible, so to have something close to a real screen.
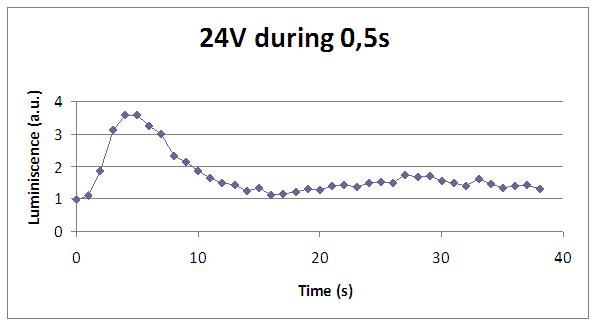
We have accomplished a refreshing rate of 12 seconds with a supply of 24V in 0,5 seconds.
Electrical input
When the experiments with an alkali input showed us that yeasts were able to produce light because of their transformation, we tried our ambitious goal: stimulate calcium channels with an electrical input.
We use two luminometers, one luminometer discontinuous and the other is continuous. Each luminometer uses different units, depends on the manufacturer. For this reasons we can't compare directly the results obtained with one luminometer with the results of the other. According this, when we only compare results in the same graph if they were obtained with the same luminometer. However, an increase (or not) in the luminosity, means the same at two luminometers and the experiments are complementary and reaffirms our conclusions.
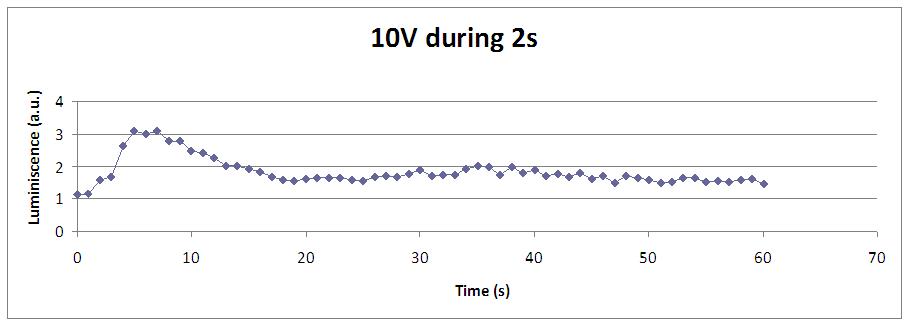
We reproduced the mentionated Viladevall et al's protocol, incubating the transformed yeasts with coelenterazine, but changing the KOH by electricity. Surprisingly, we found that light was also produced in a very similar way. We tried with different times and voltages in order to find the optim conditions for a big peak of light. Some of our graphics are theese:
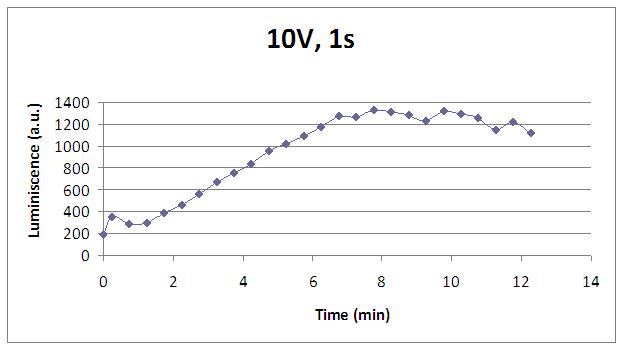
We realised that the time of exposure to the electrical stimulus was crucial, even more that the aplied voltage. That means, if we increased the voltage at very short times, cells could produce a more abrupt peak of light. But if we increased the time of exposure to the electricity, we observe a less defined response, with more flattened peaks.
That’s probably because a big exposure time of electrical input damages and killes the yeasts, making them to release their components to the medium, including the aequorin-coelenterazine-Ca2+ complex, so the emission of light is more uniform in time, instead of the production of the flash produced by the Calcium enetering in the cell.
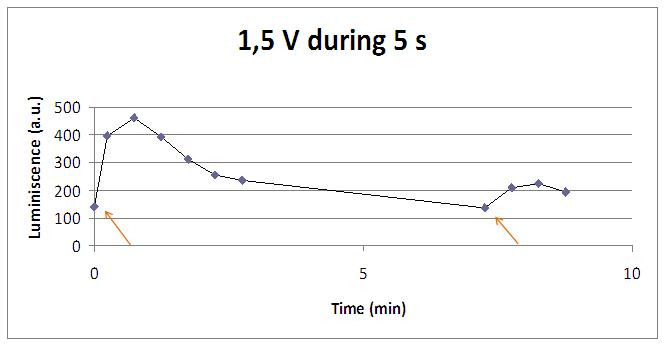
In the case of very little voltages (like 1,5V) this observation is not carried out by our yeasts. The reason must be that the electrical input is too low, so yeasts don’t die so easily as with more elevated voltage, and a better response is produce with a more prolongated electrical shock.
This graph clearly show us that using a same voltage, we obtain a better response with the shortest time of the electrical input.
Our controls discard the idea of an artifact. For example, light could be made by a spark produced during the discharge. It was not very probable, because the peak observed was produce near 400 seconds after the stimulus. But, another time, when cells without coelenterazine or mutants are used, we see no light.
Studying the repetibility of the process, this is a little different from the chemical stimulus, but the system has a similar behaviour, and we can stimulate several times the same sample getting a response. However, every next shock produces a fewer peak of light. We hace two hypothesis: one of them is that a part of our yeasts die meanwhile the electrical stimulus. The other one is that coelenterazine is not reusable, so a proportion of it runs down in every emission of light.
 "
"