Team:Uppsala-Sweden/Butanol
From 2009.igem.org
Karl.brune (Talk | contribs) (→The Pathway) |
Karl.brune (Talk | contribs) (→The Pathway) |
||
| Line 9: | Line 9: | ||
==The Pathway== | ==The Pathway== | ||
| - | The original pathway to obtain branched-chain higher alcohols was proposed and succesfully established in <i>E. coli</i> by Shota Atsumi, Taizo Hanai and James C. Liao from the University of Californa, Los Angeles [https://2009.igem.org/Team:Uppsala-Sweden/Butanol#ref1 [1]]. As we expect a broad range of higher alcohols, we will exemplarily demonstrate the biochemical mechanism at the pathway of isobutanol, the product we expect to be the most occuring one. | + | The original pathway to obtain branched-chain higher alcohols was proposed and succesfully established in <i>E. coli</i> by Shota Atsumi, Taizo Hanai and James C. Liao from the University of Californa, Los Angeles [https://2009.igem.org/Team:Uppsala-Sweden/Butanol#ref1 <nowiki>[1]</nowiki>]. As we expect a broad range of higher alcohols, we will exemplarily demonstrate the biochemical mechanism at the pathway of isobutanol, the product we expect to be the most occuring one. |
| - | [[Image:butanolpath_horizontal.png]] | + | <center>[[Image:butanolpath_horizontal.png]]</center> |
| - | The precursor for | + | The precursor for isobutanol production is pyruvate, a central metabolite which is usually derived by the glycolisis. Two pyruvate molecules are converted first to 2-acetolactate and then subsequently to 2,3-dihydroxy-isovalerate. A further dehydration reaction results in our first molecule of interest, 2-ketoisovalerate, which is a precursor for the synthesis of the aminoacids valine, alanine and leucine.[https://2009.igem.org/Team:Uppsala-Sweden/Butanol#ref2 <nowiki>[2]</nowiki>]. |
| - | So far only host enzymes were used and now | + | So far, only host enzymes were used and now we introduce our first BioBrick <partinfo>K273006</partinfo> for the Butanol Approach, the codon-optimized version of the alpha-ketoisovalerate decarboxylase (kivd) from <i>Lactococcus lactis ssp. lactis</i>[https://2009.igem.org/Team:Uppsala-Sweden/Butanol#ref3 <nowiki>[3]</nowiki>]. |
| + | |||
| + | This particular enzyme performs a decarboylation reaction, thus transforms 2-ketoisovalerate to an aldehyde, isobutanal[https://2009.igem.org/Team:Uppsala-Sweden/Butanol#ref4 <nowiki>[4]</nowiki>]. | ||
| + | |||
| + | Subsequently isobutanal is oxidized to isobutanol by the the BioBrick <partinfo>K273005</partinfo>, the alcohol dehydrogenase 2 (adh2) from <i>S. cerevisiae</i>. | ||
==The Construct== | ==The Construct== | ||
Revision as of 15:38, 19 October 2009

Contents |
The Butanol Approach
Our goal is to achieve photosynthetic production of butanol and its derivates with the cyanobacterium Synechocystis sp. PCC 6803 as the final host organism.
Background
Cyanobacteria have the capability to harvest the energy from the sun and convert it into other forms of energy. The natural way for these organisms is to store it as sugars or other carbohydrates in a way similar to plants. By introducing a casette of genes for isobutanol production, we would like to derive a cyanobacteria that produces butanol and its derivates.
The Pathway
The original pathway to obtain branched-chain higher alcohols was proposed and succesfully established in E. coli by Shota Atsumi, Taizo Hanai and James C. Liao from the University of Californa, Los Angeles [1]. As we expect a broad range of higher alcohols, we will exemplarily demonstrate the biochemical mechanism at the pathway of isobutanol, the product we expect to be the most occuring one.
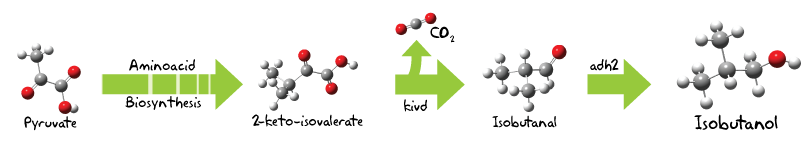
The precursor for isobutanol production is pyruvate, a central metabolite which is usually derived by the glycolisis. Two pyruvate molecules are converted first to 2-acetolactate and then subsequently to 2,3-dihydroxy-isovalerate. A further dehydration reaction results in our first molecule of interest, 2-ketoisovalerate, which is a precursor for the synthesis of the aminoacids valine, alanine and leucine.[2].
So far, only host enzymes were used and now we introduce our first BioBrick for the Butanol Approach, the codon-optimized version of the alpha-ketoisovalerate decarboxylase (kivd) from Lactococcus lactis ssp. lactis[3].
This particular enzyme performs a decarboylation reaction, thus transforms 2-ketoisovalerate to an aldehyde, isobutanal[4].
Subsequently isobutanal is oxidized to isobutanol by the the BioBrick , the alcohol dehydrogenase 2 (adh2) from S. cerevisiae.
The Construct
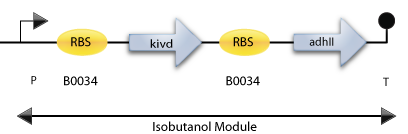
Here is the preliminary construct for Synechocystis 6803.
 "
"