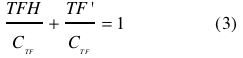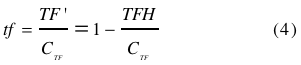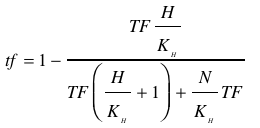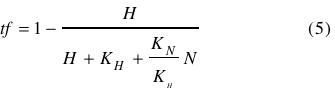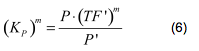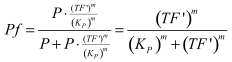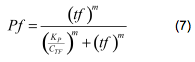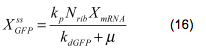Team:DTU Denmark/modelling
From 2009.igem.org
(Difference between revisions)
| (21 intermediate revisions not shown) | |||
| Line 165: | Line 165: | ||
<tr> | <tr> | ||
<td width="163px" height="100%" valign="top"> | <td width="163px" height="100%" valign="top"> | ||
| - | + | <!-- | |
| + | <font color="#990000" face="arial" size="3"> | ||
<br> | <br> | ||
Also check out our Danish website<br><br> | Also check out our Danish website<br><br> | ||
| Line 173: | Line 174: | ||
<br> | <br> | ||
</font> | </font> | ||
| + | --> | ||
</td> | </td> | ||
<td width="556px" height="100%" valign="top"> | <td width="556px" height="100%" valign="top"> | ||
<font color="#990000" face="arial" size="5"> | <font color="#990000" face="arial" size="5"> | ||
<br> | <br> | ||
| - | <b>Modelling of | + | <b>Modelling of The Redoxilator</b><br><br> |
</font> | </font> | ||
| Line 188: | Line 190: | ||
<font size="3"><p><b>Introduction</b></p></font> | <font size="3"><p><b>Introduction</b></p></font> | ||
| + | Here a model for the GFP expression under the control of “The Redoxilator” expression system is derived. The model is based on modular structured kinetics and links the level of the signal molecules NADH and NAD to the final expression of GFP through the events of <br><br> | ||
| - | + | i) transcription factor activation,<br> | |
| - | <br> | + | ii) promoter activation, <br> |
| - | <br | + | iii) synthesis and degradation of mRNA and finally, <br> |
| - | + | iv) synthesis, dilution and degradation of GFP. <br><br> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | This model formed the basis of the genetic design of the The redoxilator. Importantly it provided understanding of the influence of each of the parameters when designing the system, e.g. that in order to achieve fast response time a fast degradable GFP was needed as reporter. However, since many parameters specific for this system are not yet available, the model is not expected to make exact predictions but to ease the understanding of the design "principles". | |
| + | |||
| + | <br><br> | ||
| + | </html> | ||
| + | [[Image:ReporterMechanism.png|400px|thumb|center|The redox coupled system]] | ||
| + | <html> | ||
| + | |||
| + | <p align="justify"><i><b>Gene design and redox regulation</b><br> | ||
| + | NADH and NAD competes about binding to REX, however only when NADH is bound, the trancription factor is inhibited from binding to DNA. When the transcription factor is active (NADH not bound), the fused activator domain summons the RNA polymerase and the reporter gene is transcribed.</i></p><br> | ||
| + | |||
| + | |||
| + | <font size="3"><p><b>NADH and NAD binding to the transcription factor</b></p></font> | ||
Both NADH and NAD binds reversible to the same site of the transcription factor (TF): <br><br> | Both NADH and NAD binds reversible to the same site of the transcription factor (TF): <br><br> | ||
| - | Dissociation of TF and NADH | + | Dissociation of TF and NADH |
| - | <br></html>[[image:picture_2.png]]<html | + | <br></html>[[image:picture_2.png|120px]]<html><br> |
| - | at equilibrium: | + | at equilibrium: |
| - | <br></html>[[image:picture_3.png]]<html | + | <br></html>[[image:picture_3.png]]<html><br> |
| - | Dissociation of TF and NAD: | + | Dissociation of TF and NAD: |
| - | <br></html>[[image: | + | <br></html>[[image:picture_46.png|120px]]<html><br><br> |
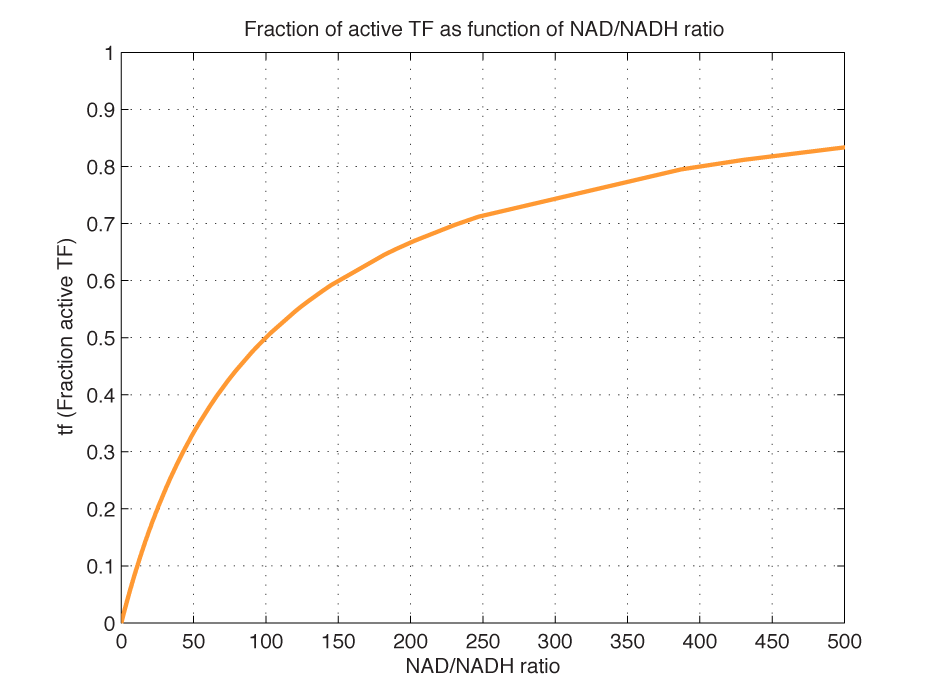
| - | Dissociation of TF and NAD: | + | Dissociation constant of TF and NAD: |
<br></html>[[image:picture_5.png]]<html><br><br> | <br></html>[[image:picture_5.png]]<html><br><br> | ||
| - | Balance of | + | Balance of NAD and NADH. Constant total level of NADH + NAD in the cell are assumed: |
<br></html>[[image:picture_6.png]]<html><br><br> | <br></html>[[image:picture_6.png]]<html><br><br> | ||
| Line 229: | Line 230: | ||
<br></html>[[image:picture_8.png]]<html><br><br> | <br></html>[[image:picture_8.png]]<html><br><br> | ||
| - | + | NADH inhibits DNA binding activity of TF and thus transcription activity. Active TF is designated TF’. | |
| - | <br></html>[[image: | + | <br></html>[[image:picture_38.png]]<html><br><br> |
Leading to the conserved moiety: | Leading to the conserved moiety: | ||
| - | <br></html>[[image: | + | <br></html>[[image:picture_40.png]]<html><br><br> |
Using (3), the fraction of TF’ (<i>tf</i>) can be written as | Using (3), the fraction of TF’ (<i>tf</i>) can be written as | ||
| - | <br></html>[[image: | + | <br></html>[[image:picture_41.png]]<html><br><br> |
With (1) and (2) in (4) the <i> tf </i> becomes | With (1) and (2) in (4) the <i> tf </i> becomes | ||
| - | <br></html>[[image: | + | <br></html>[[image:picture_42.png]]<html><br><br> |
Leading to | Leading to | ||
| - | <br></html>[[image: | + | <br></html>[[image:picture_43.png]]<html><br><br> |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| + | Importantly figure 1 shows how the transcription factor is affected by the cofactor pool of NAD(H). The competing binding of NADH and NAD makes the system sense the redox poise rather than the concentration of each of the cofactors [2]. <br> | ||
| + | </html>[[Image:TFratio.png|350px|thumb|center]]<html> | ||
| + | <i><b>Figure 1 - Transcription factor activity with increasing NAD/NADH ratio is hyperbolic.</b> <br> At NAD/NADH = 0.1, <i>tf</i> = 0.5, i.e. half of the transcription factors are active. (For this plot the following parameters were used: assuming K_H = 0,02 μM and K_N = 2 μM since REX NADH affinity > REX NAD affinity [2]. </html>[[image:picture_7.png]]<html> = 2mM, estimated TF concentration expressed from a constitutive TEF promoter.)<br><br></i> | ||
<font size="3"><p><b>Promoter binding of active transcription factor</b></p></font> | <font size="3"><p><b>Promoter binding of active transcription factor</b></p></font> | ||
| - | When the active TF (TF') is bound to the promoter P, P becomes active (P’) and transcription is initiated. The number of | + | When the active TF (TF') is bound to the promoter (P), P becomes active (P’) and transcription is initiated. The number of transcription factors needed for promoter activation is designated m: |
| - | <br></html>[[image: | + | <br></html>[[image:picture_47.png]]<html><br><br> |
Here it assumed that binding of TF and TFN is equal. <br> | Here it assumed that binding of TF and TFN is equal. <br> | ||
| Line 267: | Line 260: | ||
<br></html>[[image:picture_15.png]]<html><br><br> | <br></html>[[image:picture_15.png]]<html><br><br> | ||
| - | The number of promoters is constant, | + | The number of promoters is constant, making the sum of fractions equal to |
<br></html>[[image:picture_16.png]]<html><br><br> | <br></html>[[image:picture_16.png]]<html><br><br> | ||
| Line 278: | Line 271: | ||
<br></html>[[image:picture_18.png]]<html><br><br> | <br></html>[[image:picture_18.png]]<html><br><br> | ||
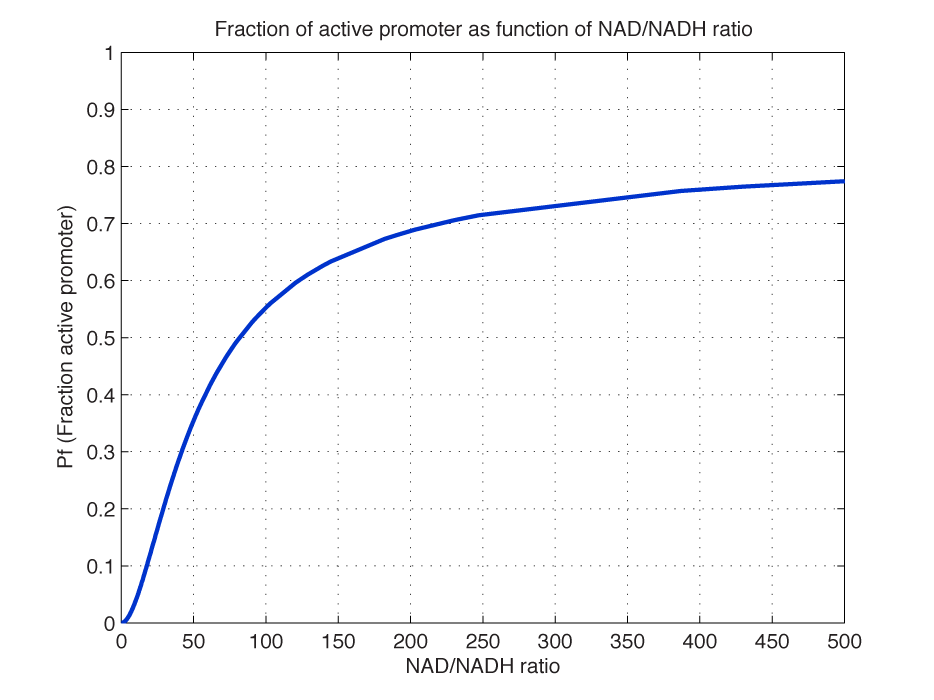
| - | Now introducing </html>[[image:picture_37.png]]<html> we can obtain a function of the fraction of active promoter sites as a function of | + | Now introducing </html>[[image:picture_37.png]]<html> we can obtain a function of the fraction of active promoter sites as a function of NAD(H) level: |
<br></html>[[image:picture_19.png]]<html><br><br> | <br></html>[[image:picture_19.png]]<html><br><br> | ||
| - | + | For simplicity the expression of (5) has not been inserted. Figure 2 shows how the redox poise modulates transcription from the promoter P, which is controlled by The Redoxilator.<br> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | <br> | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| + | <br></html>[[image:Pfratio.png|350px|thumb|center]]<html> | ||
| + | <i><b>Figure 2 - Fraction of active promoters with increasing NAD/NADH ratio.</b><br> The curve is sigmoidal and has almost switch like behavior. From NAD/NADH = 0 to 0.01 the difference in active promoter is almost 60 %.(Assumed value of K_P = 1 mM, and m = 2). | ||
| + | <br></i><br> | ||
<font size="3"><p><b>mRNA production</b></p></font> | <font size="3"><p><b>mRNA production</b></p></font> | ||
| Line 303: | Line 291: | ||
The term </html>[[image:picture_22.png]]<html> represents the dilution due to growth. However mRNA is highly unstable and is thus degraded within tens of seconds to order few minutes, making <i> k_dm</i> very small compared to growth rate. This makes </html>[[image:picture_24.png]]<html>, simplifying the mRNA balance to: | The term </html>[[image:picture_22.png]]<html> represents the dilution due to growth. However mRNA is highly unstable and is thus degraded within tens of seconds to order few minutes, making <i> k_dm</i> very small compared to growth rate. This makes </html>[[image:picture_24.png]]<html>, simplifying the mRNA balance to: | ||
| - | <br></html>[[image:picture_25.png]]<html><br><br> | + | <br></html>[[image:picture_25.png]]<html> (9)<br><br> |
<b>Production rate of mRNA </b> </html>[[image:picture_26.png]]<html> <br> | <b>Production rate of mRNA </b> </html>[[image:picture_26.png]]<html> <br> | ||
| - | It is assumed that when RNA polymerase is efficiently bound to the promoter, the promoter is active and mRNA is transcribed at constant maximum rate </html>[[image:picture_27.png]]<html>. The binding of RNA polymerase is dependent on the presence of transcription factor and determined by signal strength ( | + | It is assumed that when RNA polymerase is efficiently bound to the promoter, the promoter is active and mRNA is transcribed at constant maximum rate </html>[[image:picture_27.png]]<html>. The binding of RNA polymerase is dependent on the presence of transcription factor and determined by signal strength (NAD/NADH) as well as Kp. In the overall production rate of mRNA, of course also the number of promoters (and genes) Np is influencing: |
<br></html>[[image:picture_28.png]]<html><br><br> | <br></html>[[image:picture_28.png]]<html><br><br> | ||
| Line 317: | Line 305: | ||
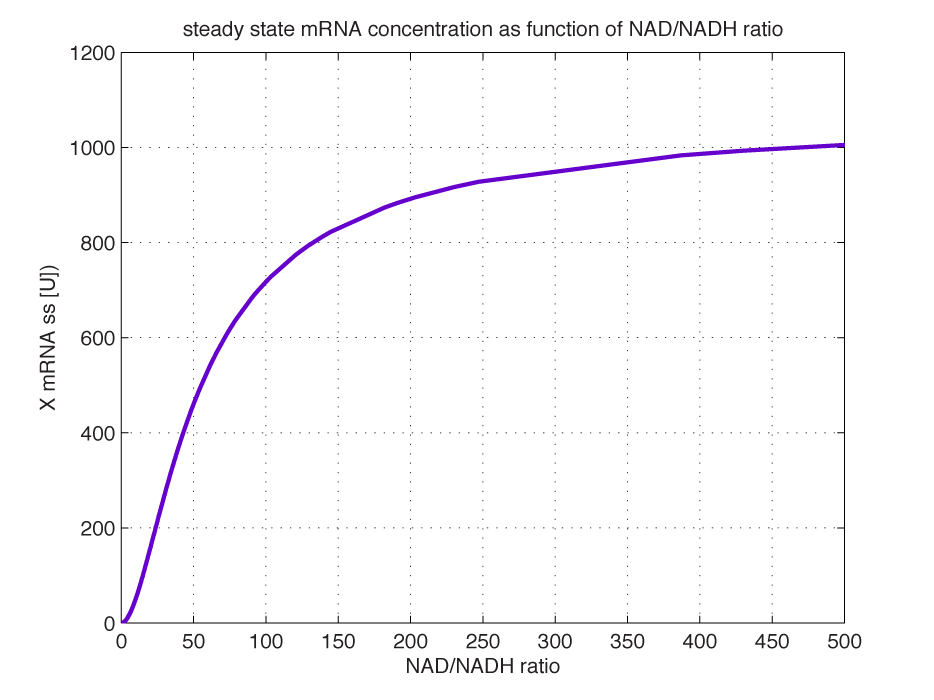
At steady state (11) is equal to zero since no mRNA accumulates. Here the mRNA concentration is: | At steady state (11) is equal to zero since no mRNA accumulates. Here the mRNA concentration is: | ||
| - | <br></html>[[image:picture_29.png]]<html | + | <br></html>[[image:picture_29.png]]<html><br> |
| - | <br></html>[[image: | + | <br></html>[[image:MRNAratio.png|350px|thumb|center]]<html> |
| - | <b>Figure 3 | + | <i><b>Figure 3 - mRNA levels at steady state with increasing NAD/NADH ratios.</b> <br>Dilution is neglected since degradation rate of mRNA is much larger. (Assumed parameters: kdm = 8.3 h-1 (half life = 5 min), Np = 60 (2 micron plasmid is used). and </html>[[image:picture_27.png]]<html> = 180 U/h (assuming 75 bases per second, and 1500 bases in gene).) |
| - | <br> | + | <br></i><br> |
| - | |||
| Line 332: | Line 319: | ||
Finally, the mRNA is used as template for the process of translation to protein, here GFP. The GFP protein balance: | Finally, the mRNA is used as template for the process of translation to protein, here GFP. The GFP protein balance: | ||
| - | <br></html>[[image: | + | <br></html>[[image:picture_30.png]]<html><br><br> |
| - | As with mRNA it is assumed that GFP is degraded by first order rate. The dilution cannot be neglected since protein degradation is much slower than mRNA degradation. Furthermore it is assumed that GFP is not transported out of the cell. | + | As with mRNA, it is assumed that GFP is degraded by first order rate. The dilution cannot be neglected since protein degradation is much slower than mRNA degradation. Furthermore it is assumed that GFP is not transported out of the cell. |
<br><br> | <br><br> | ||
| - | The protein is synthesized by ribosomes. The number of ribosomes bound to one mRNA (Np) and the protein production rate of the ribosome (kp) should be taken into account. The number of ribosomes in the cell, ATP and available charged amino acids are assumed to be non-limiting. Taking this into account the protein synthesis rate can be described as | + | The protein is synthesized by ribosomes. The number of ribosomes bound to one mRNA (Np) and the protein production rate of the ribosome (kp) should be taken into account. The number of ribosomes in the cell, ATP and available charged amino acids are assumed to be non-limiting. Taking this into account, the protein synthesis rate can be described as |
<br></html>[[image:picture_31.png]]<html><br><br> | <br></html>[[image:picture_31.png]]<html><br><br> | ||
| Line 346: | Line 333: | ||
<br></html>[[image:picture_32.png]]<html><br><br> | <br></html>[[image:picture_32.png]]<html><br><br> | ||
| - | + | This final relation relates the GFP balance directly to the levels of NADH and NAD through mRNA level, promoter activity and transcription factor activity. <br><br> | |
<b>Steady state concentration of GFP</b><br> | <b>Steady state concentration of GFP</b><br> | ||
| Line 353: | Line 340: | ||
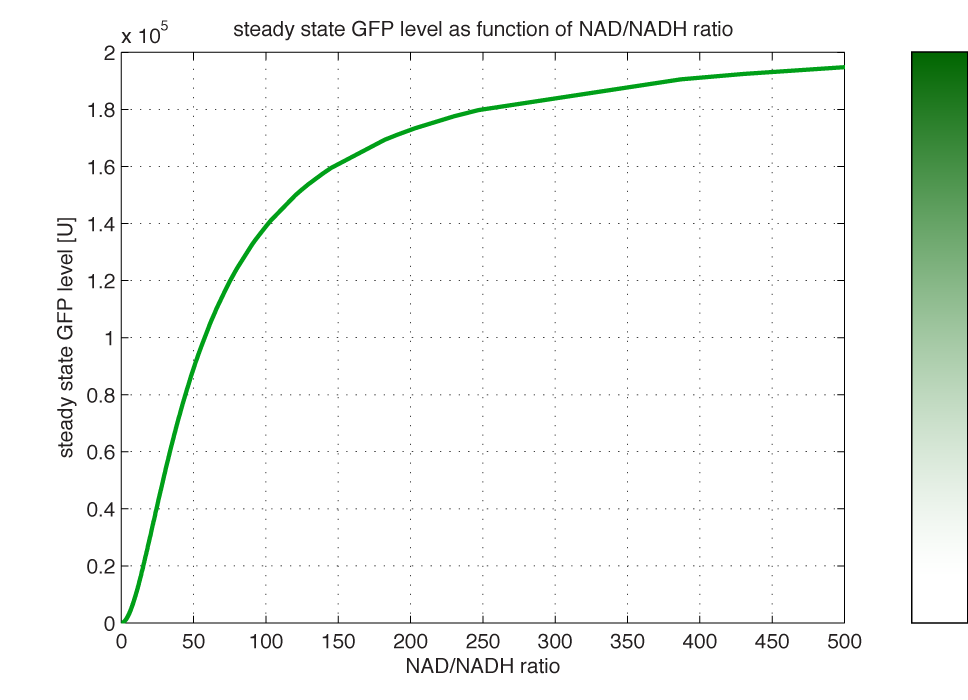
<br></html>[[image:picture_33.png]]<html><br><br> | <br></html>[[image:picture_33.png]]<html><br><br> | ||
| - | <br></html>[[image: | + | This relation opens certain possibilities: By knowing the NAD/NADH ratio of the cell, one can predict the GFP expression using this model. Or opposite and even more relevant, knowing (by measurement) the GFP level of the cell, one can point out the redox state. In this way The redoxilator acts as an <i>in vivo</i> redox sensor. Figure 4 shows how the steady state concentration of GFP changes at different redox poises. |
| - | <b>Figure 4 | + | <br> |
| - | Assumed parameters Nrib = 1. kp = 288 U/h | + | <br></html>[[image:GFPssratio.png|350px|thumb|center]]<html> |
| + | <i><b>Figure 4 - Steady state GFP levels as a direct function of NAD/NADH signal levels.</b><br> of a cell in steady state. (Assumed parameters Nrib = 1. kp = 288 U/h, assuming 40 amino acids per second. k_dGFP = 1.386 h-1 equal to a half time of 30 min [3]. Growth rate = 0.1 h-1).</i> <br><br> | ||
| + | So far a model has been derived showing how steady state GFP levels changes with different values of NAD/NADH poise. This is applicable when the NAD/NADH ratios are constant. However, when NAD/NADH changes in time, GFP also changes in time <i>dynamically</i>. In order to sense changes in NAD/NADH ratios it is important that the reporter signal (GFP level) changes relatively fast. The following paragraph deals with the dynamic state. | ||
| + | <br><br> | ||
<font size="3"><p><b>The dynamic state</b></p></font> | <font size="3"><p><b>The dynamic state</b></p></font> | ||
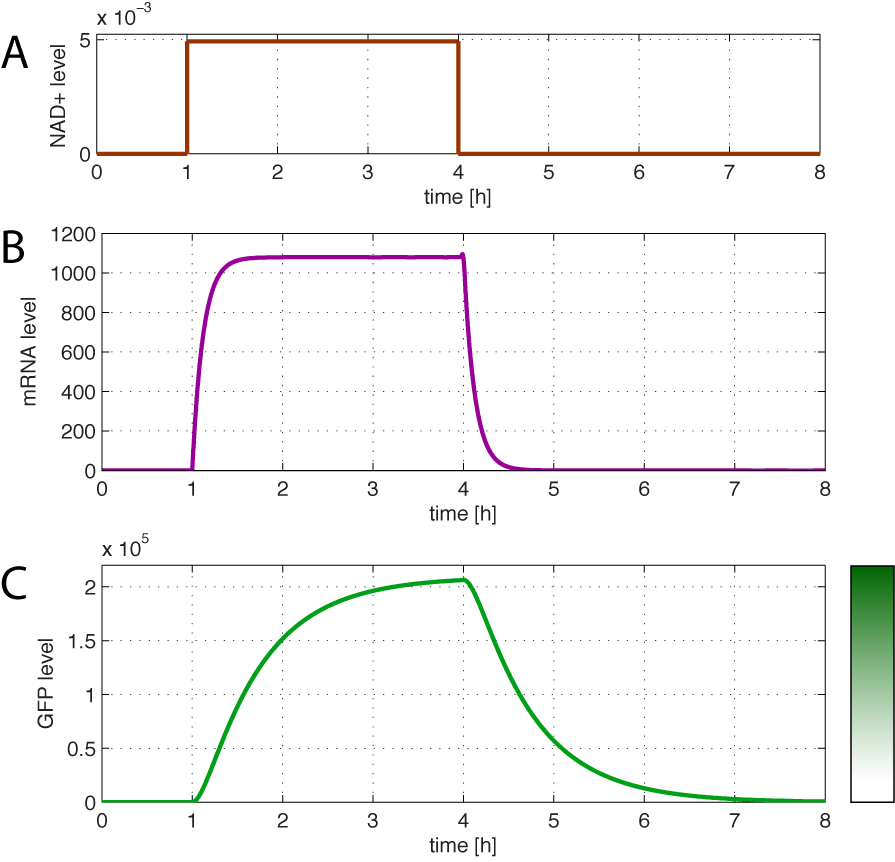
| - | When the cells are exposed to a certain | + | When the cells are exposed to a certain NAD/NADH level mRNA and in turn GFP levels will change in time according to their balances. In figure 5 the NAD+ level is changed from 0 M to 5mM equal to total amount of NAD(H). |
| - | + | <br></html>[[image:Response.png|400px|thumb|center]]<html> | |
| + | <i><b>Figure 5 - Dynamics of GFP expression when NAD+ level is changed</b><br> | ||
| + | <b>A:</b> At t = 1, the NAD+ level is suddenly shifted from 0 M to the total concentration of NAD(H). At t = 4, the NAD is shifted to 0 M. <b>B:</b> dynamic mRNA level. A fast response in mRNA levels is observed in both production and degradation. <b>C:</b> dynamic GFP levels. The response is much slower than mRNA. After app. 1/2 hour from signal burst, the GFP level has reached half of the maximum level. Degradation and dilution is relatively fast for a protein with a response time of approximately 40 min. | ||
| + | </i> <br><br> | ||
| - | + | Figure 5 illustrates the importance of using a fast degradable GFP when using the redoxilator system as a sensor. If the cells that are subject for an <i>in vivo</i> analysis of redox poise using The redoxilator suddenly shift to a new state, the sensor will respond fast (in about 40 minutes with the assumed parameters). | |
| - | + | <br><br> | |
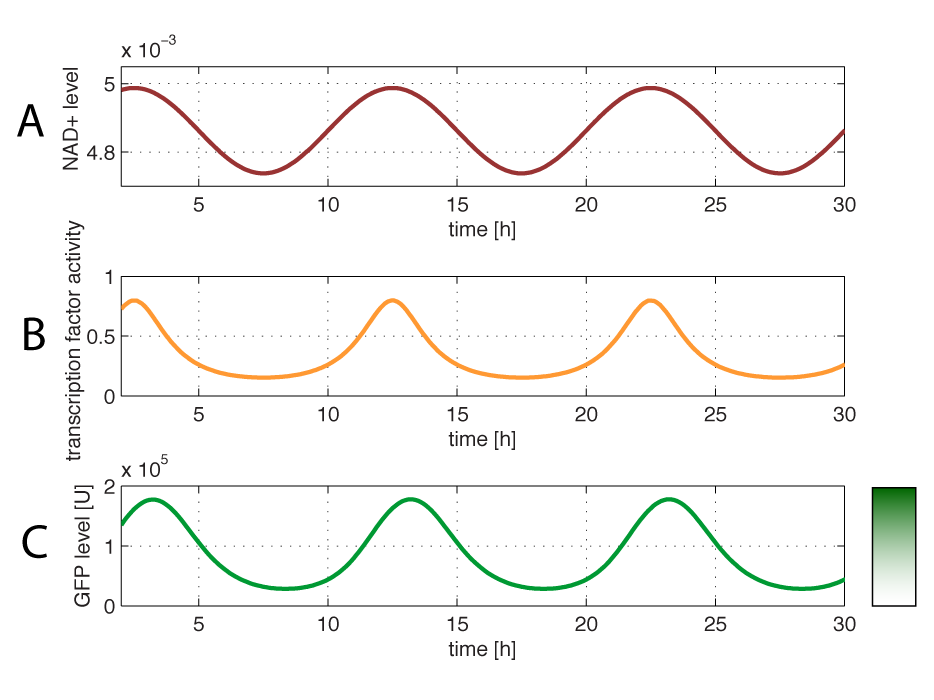
| - | + | If the NAD/NADH level changes in time, all values depending on NAD(H) concentrations will also be time depending. This will result in periodic burst in the production of the protein under control of the redox system: | |
| - | [ | + | <br></html>[[image:Dynamic2.png|500px|thumb|center]]<html> |
| + | <i><b>Figure 6 - Dynamics of GFP expression when NAD+ level is changed</b> <br> | ||
| + | <b>A:</b>Simulated change in NAD+/NADH level.<b>B:</b> Since NAD+/NADH ratio changes periodically, also transcription factor activity changes.<b>C:</b> The changes in NAD+ affects the dynamic GFP levels. | ||
| + | </i> | ||
| + | <br><br> | ||
| + | Figure 6 shows how the protein under control of the redoxilator is produced in bursts. Due to the fast degradation of GFP in this case, GFP levels will change in response to the redox level with a fast response time. | ||
| + | <br><br> | ||
| + | |||
| + | <b>References</b> <br> | ||
| + | [1] Heijnen J.J 2009. Modular structured kinetic models: protein production from genes.<br> | ||
| + | [2] Brekasis, D., Paget, M.S., 2003. A novel sensor of NADH/NAD+ redox poise in Streptomyces coelicolor A3(2). EMBO J. 22, 4856–4865.<br> | ||
| + | [3] Mateus and Avery, 2000. Destabilized green fluorescent protein for monitoring dynamic changes in yeast gene expression with flow cytometry. Yeast 2000; 16: 1313±1323. <br> | ||
| Line 387: | Line 391: | ||
<!-- INSERT GREY BOX TEXT HERE! (formatting: <b>bold</> <i>italic> <h4>header</h4>) --> | <!-- INSERT GREY BOX TEXT HERE! (formatting: <b>bold</> <i>italic> <h4>header</h4>) --> | ||
| - | + | <b>Abbreviations used in the model</b> | |
| - | + | <br><br> | |
| - | + | <i>TF</i> = the transcription factor based on the REX protein <br><br> | |
| + | <i>TF'</i> = active transcription factor <br><br> | ||
| + | <i>N</i> = concentration of NAD <br><br> | ||
| + | <i>H</i> = concentration of NADH <br><br> | ||
| + | <i>TFH</i> = concentration of the transcription factor bound to NADH. <br><br> | ||
| + | <i>TFN</i> = concentration of the transcription factor bound to NAD. <br><br> | ||
| + | <i>P</i> = Promoter upstream of GFP <br><br> | ||
| + | <i>P'</i> = activated/transcribed promoter <br><br> | ||
| + | <i>tf</i> = fraction of active transcription factor <br><br> | ||
| + | <i>Pf</i> = fraction of active promoter<br><br> | ||
| + | <br> | ||
</font> | </font> | ||
Latest revision as of 01:12, 22 October 2009

| Home | The Team | The Project | Parts submitted | Modelling | Notebook |
| Comments or questions to the team? Please Email us -- Comments of questions to webmaster? Please Email us |
 "
"