|
|
| (56 intermediate revisions not shown) |
| Line 1: |
Line 1: |
| - | {| style="color:#1b2c8a;background-color:#0c6;" cellpadding="3" cellspacing="1" border="1" bordercolor="#fff" width="62%" align="center" | + | __NOTOC__ |
| - | !align="center"|[[Team:Washington|Home]]
| + | {{Template:Team:Washington/Templates/Header}} |
| - | !align="center"|[[Team:Washington/Team|The Team]]
| + | |
| - | !align="center"|[[Team:Washington/Project|Project Description]]
| + | |
| - | !align="center"|[[Team:Washington/Notebook|Notebook]]
| + | |
| - | !align="center"|[[Team:Washington/UserGuide|User Guide]]
| + | |
| - | !align="center"|[[Team:Washington/Accomplishments|Goals and Accomplishments]]
| + | |
| - | |}
| + | |
| - | | + | |
| - | {|align="center"
| + | |
| - | |
| + | |
| - | ''
| + | |
| - | | + | |
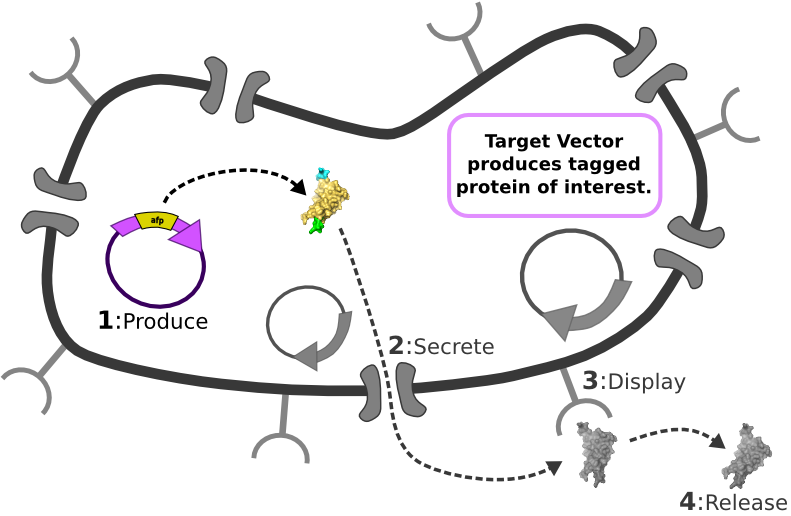
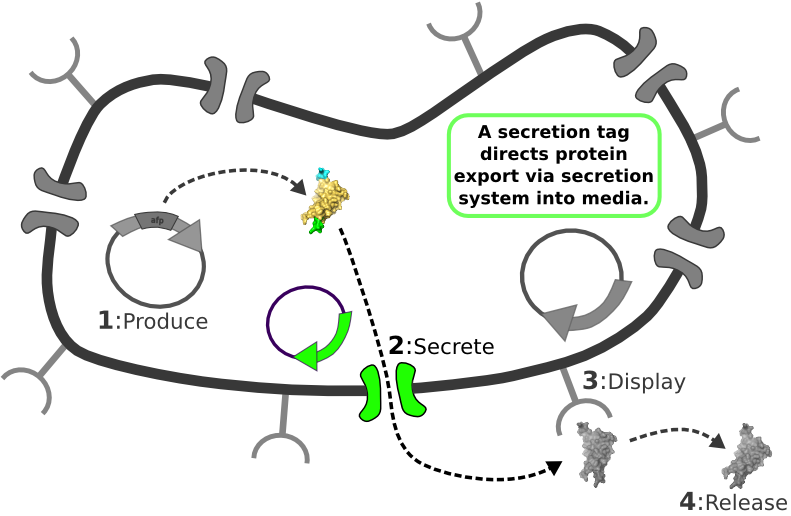
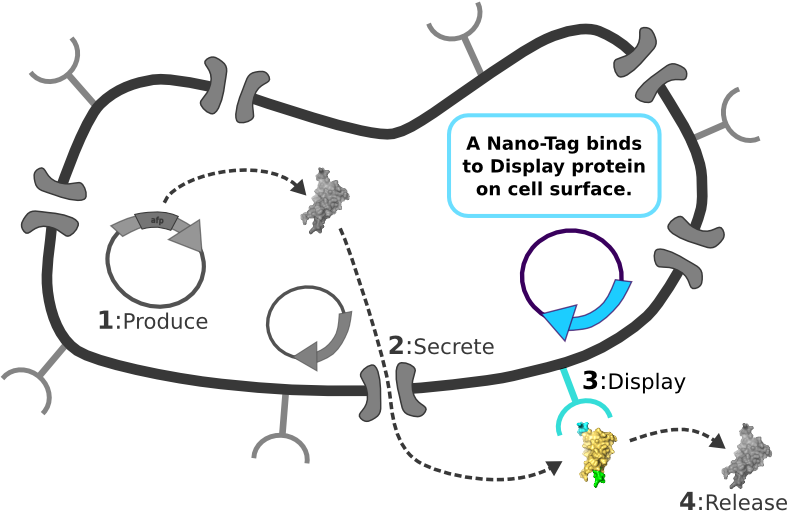
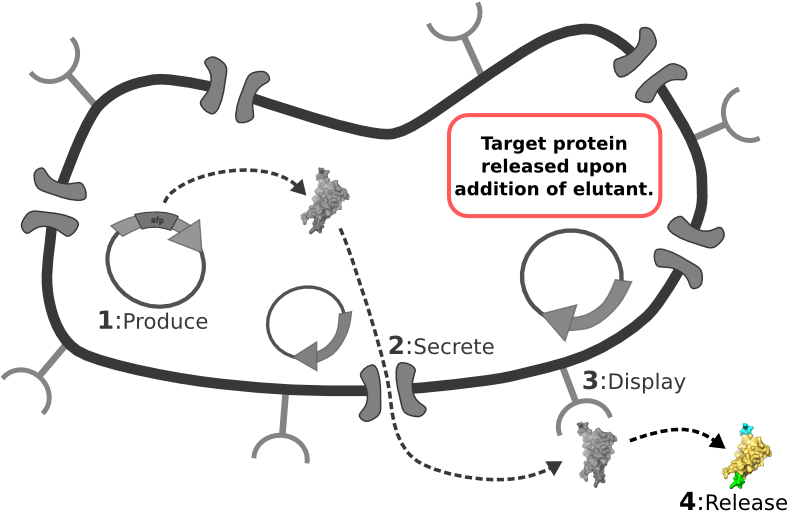
| - | [[Image:Main_graphic.png|center|800px]] [1] Target Expression Vector produces fusion protein of interest.
| + | |
| - | [2] Secretion Tag on fusion protein is recognized by secretion system and transported to extracellular space.
| + | |
| - | | + | |
| - | [3] Nano-Tag on fusion protein is recognized by protein on cell surface and binds non-covalently to cell.
| + | |
| - | | + | |
| - | [4] Competing binder for surface protein releases protein of interest into supernatant.
| + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | The use of recombinant protein production using E. coli-based expression
| + | |
| - | systems has revolutionized the fields of biotechnology and medicine.
| + | |
| - | However, the ability to utilize such proteins hinges upon their capacity to
| + | |
| - | be isolated from their expression systems. Our project aims to create an
| + | |
| - | all-in-one protein expression and purification system using BioBrick
| + | |
| - | standards to greatly simplify protein production for synthetic biologists,
| + | |
| - | reducing the time and cost involved in standard protein purification
| + | |
| - | methods. Our method uses a novel combination of two systems: secretion and
| + | |
| - | display. By fusing two tags to the protein it can be secreted into the
| + | |
| - | expression media, and subsequently directed to bind to the outside of the
| + | |
| - | cell. To collect the pure proteins, cells only need to be spun down and
| + | |
| - | then resuspended in an elution buffer, releasing the protein of interest.
| + | |
| - | Our research exhibits the utility of synthetic biology for developing new
| + | |
| - | techniques that improve upon established practices.''
| + | |
| - | |-
| + | |
| - | |}
| + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | <!-- *** What falls between these lines is the Alert Box! You can remove it from your pages once you have read and understood the alert *** -->
| + | |
| - | | + | |
| | <html> | | <html> |
| - | <div id="box" style="width: 700px; margin-left: 137px; padding: 5px; border: 3px solid #000; background-color: #fe2b33;"> | + | <script type="text/javascript"> |
| - | <div id="template" style="text-align: center; font-weight: bold; font-size: large; color: #f6f6f6; padding: 5px;">
| + | $(function() { |
| - | This is a template page. READ THESE INSTRUCTIONS.
| + | var mainimg = document.getElementById("mainimg"); |
| - | </div> | + | var default_src = mainimg.src; |
| - | <div id="instructions" style="text-align: center; font-weight: normal; font-size: small; color: #f6f6f6; padding: 5px;"> | + | var restore_mainimg = function() { |
| - | You are provided with this team page template with which to start the iGEM season. You may choose to personalize it to fit your team but keep the same "look." Or you may choose to take your team wiki to a different level and design your own wiki. You can find some examples <a href="https://2009.igem.org/Help:Template/Examples">HERE</a>.
| + | mainimg.src = default_src; |
| - | </div> | + | }; |
| - | <div id="warning" style="text-align: center; font-weight: bold; font-size: small; color: #f6f6f6; padding: 5px;"> | + | $(document.getElementById("area_target")).hover(function() { |
| - | You <strong>MUST</strong> have a team description page, a project abstract, a complete project description, and a lab notebook. PLEASE keep all of your pages within your teams namespace.
| + | mainimg.src = "/wiki/images/1/1d/Main_graphic2_target.png"; |
| - | </div> | + | }, restore_mainimg); |
| | + | $(document.getElementById("area_secretion")).hover(function() { |
| | + | mainimg.src = "/wiki/images/2/23/Main_graphic2_secretion.png"; |
| | + | }, restore_mainimg); |
| | + | $(document.getElementById("area_display")).hover(function() { |
| | + | mainimg.src = "/wiki/images/a/aa/Main_graphic2_display.png"; |
| | + | }, restore_mainimg); |
| | + | $(document.getElementById("area_release")).hover(function() { |
| | + | mainimg.src = "/wiki/images/e/ed/Main_graphic2_release.png"; |
| | + | }, restore_mainimg); |
| | + | }); |
| | + | </script> |
| | + | <img id="mainimg" src="/wiki/images/4/41/Main_graphic.png" alt="IPP System Overview" usemap="#cellmap" /> |
| | + | <map name="cellmap"> |
| | + | <area id="area_target" shape="poly" coords="76,282,167,343,395,184,144,102" href="/Team:Washington/Project/Target" alt="Target System" /> |
| | + | <area id="area_secretion" shape="poly" coords="485,202,392,182,208,312,322,505,485,505" href="/Team:Washington/Project/Secretion" alt="Secretion System" /> |
| | + | <area id="area_display" shape="rect" coords="485,202,660,505" href="/Team:Washington/Project/Display" alt="Display System" /> |
| | + | <area id="area_release" shape="rect" coords="660,344,781,503" href="/Team:Washington/Project/Release" alt="Release System" /> |
| | + | </map> |
| | + | <div style="display:none"> |
| | + | <img src="/wiki/images/1/1d/Main_graphic2_target.png" alt="target" /><br /> |
| | + | <img src="/wiki/images/2/23/Main_graphic2_secretion.png" alt="secretion" /><br /> |
| | + | <img src="/wiki/images/a/aa/Main_graphic2_display.png" alt="display" /><br /> |
| | + | <img src="/wiki/images/e/ed/Main_graphic2_release.png" alt="release" /> |
| | </div> | | </div> |
| | </html> | | </html> |
| | + | <br /> |
| | | | |
| - | <!-- *** End of the alert box *** -->
| + | === The Idealized Protein Purification System: Improving the lives of molecular biologists === |
| - | | + | |
| - | | + | |
| - | ==Display System== | + | |
| - | | + | |
| - | ===Background===
| + | |
| - | This display system was biobricked by Harvard in 2006. They submitted four biobricks which where all variations on the same theme. All four are fusion proteins which have a LPP signal peptide, either one or five trans-membrane ompA, and either monomeric or dimeric streptavidin. All the variaitons and the resulting biobricks are shown below.
| + | |
| - | | + | |
| - | {|
| + | |
| - | ! Bio Brick
| + | |
| - | ! OmpA trans-membrane domains
| + | |
| - | ! Type of Streptavadin
| + | |
| - | |-
| + | |
| - | |J36848
| + | |
| - | |1
| + | |
| - | |monomeric
| + | |
| - | |-
| + | |
| - | |J36849
| + | |
| - | |1
| + | |
| - | |dimeric
| + | |
| - | |-
| + | |
| - | |J36850
| + | |
| - | |5
| + | |
| - | |monomeric
| + | |
| - | |-
| + | |
| - | |J36851
| + | |
| - | |5
| + | |
| - | |dimeric
| + | |
| - | |-
| + | |
| - | |}
| + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | A basic graphic of the structure of this display system is shown below:
| + | |
| - | | + | |
| - | [[Image:Display image.jpg]]
| + | |
| - | | + | |
| - | They used a LPP signal peptide (green) to cause the fusion protein to migrate to the outer surface. Though ompA is normally a membrane bound protein, often it is only expressed in the periplasm, the LPP signal ensures it is exported all the way to the cell membrane. This LPP-ompA system is a well characterized protein for displaying enzymes on the surface of the cell. This system has been used to display the following proteins on the surface of the cell:
| + | |
| - | | + | |
| - | # Chitin binding domains
| + | |
| - | # ...
| + | |
| - | # ...
| + | |
| - | # ...
| + | |
| - | | + | |
| - | Thus we considered it only logical to use this system to display sreptavadin on the surface of the cells. We choose streptavadin because of its low affintiy for the nano tag on our target fusion protein, which can be displaced by the small molecule biotin. Streptavadin is known to have a high affinity for binding the small molecule biotin. The picture below shows the biotin streptavadin complex.
| + | |
| - | | + | |
| - | [[Image:Biotin-strept.jpg]]
| + | |
| - | | + | |
| - | In order to characterize how these parts worked, we ran the follwoing tests:
| + | |
| - | # Western against the his tags on all of these proteins
| + | |
| - | # Micrscope Assay
| + | |
| - | # Flow Cytometry Assay
| + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | === Western === | + | |
| - | The goal of this experiment was to make sure that the proteins were being expressed in our cell lines, and also to make sure that they were the correct length. Here is our procedure for the preparation of the western.
| + | |
| - | | + | |
| - | ==== Procedure ====
| + | |
| - | # Set up overnights of parts 48-51
| + | |
| - | # Dilute 1 ul overnight into 1ml
| + | |
| - | # Add 1 mm IPTG and let grow for four hours
| + | |
| - | # After cells have all grown up uniformly start boiling water for the boil step
| + | |
| - | # Add 100uL of overnight to a 1.5mL tube
| + | |
| - | # Pellet by spinning at max speed for 30 secs in the microcentrifuge
| + | |
| - | # discard supernatent
| + | |
| - | # Pull an aliquot of 5x sample loading buffer out of the freezer and thaw
| + | |
| - | # Add 20uL BME to aliquot
| + | |
| - | # Resuspend samples in 50uL sample loading buffer (pipette up/down)
| + | |
| - | # Boil samples for 10 minutes
| + | |
| - | # While boiling, prepare 500mL 1x SDS buffer:
| + | |
| - | ## 50mL 10x buffer to 450mL water
| + | |
| - | ## Take a gel out of the fridge and and put it in the gel box (keep the gel container for staining!!!)
| + | |
| - | ## Pour the mixed buffer solution into the half of the gel box that the gel is in
| + | |
| - | ## Remove the gel comb
| + | |
| - | ## Fill the little container on the top of the gel until it's about 0.5 cm from the top with buffer
| + | |
| - | ## Remove any bubbles in the wells
| + | |
| - | # Spin down samples for a few seconds
| + | |
| - | # Vortex samples
| + | |
| - | # Load 3uL into each well
| + | |
| - | # Load 10uL of ladder in appropriate wells
| + | |
| - | # Run at 180V until the dye is about to fall off the gel
| + | |
| - | | + | |
| - | ==== Results ====
| + | |
| - | | + | |
| - | [[Image:Western.jpg]]
| + | |
| - | ''I know this is not annotated, but it will be on the real site''
| + | |
| - | | + | |
| - | The expected values of each of these proteins are shown in the table below:
| + | |
| - | {|
| + | |
| - | ! Bio Brick
| + | |
| - | ! Length (in kD)
| + | |
| - | |-
| + | |
| - | |J36848
| + | |
| - | |21478.7
| + | |
| - | |-
| + | |
| - | |J36849
| + | |
| - | |34600.9
| + | |
| - | |-
| + | |
| - | |J36850
| + | |
| - | |31215.4
| + | |
| - | |-
| + | |
| - | |J36851
| + | |
| - | |44972.3
| + | |
| - | |-
| + | |
| - | |}
| + | |
| - | | + | |
| - | From this data one can see that all of the bands from our proteins came out to be in the right locations. Thus our protein was being expressed.
| + | |
| - | | + | |
| - | | + | |
| - | === Microscope Assay ===
| + | |
| - | The goal of this experiment was to see if we could confirm the presence of streptavadin on the outside of our cell. We wanted to do this using a biotyntalated flourophore. We expected that this flourophore would bind to the streptavadin on the outside of the cell in such a high level that we would be able to detect it under a high powered florescence microscope. As a positive control we used streptavadin coated beads which were roughly the same size (with respect to volume) as our cells (except spherical).
| + | |
| - | | + | |
| - | ==== Procedure ====
| + | |
| - | # Set up overnights of parts 48-51. Let grow overnight.
| + | |
| - | # Dilute 1 ul overnight into 1ml
| + | |
| - | # Add 1 mm IPTG and let grow for four hours
| + | |
| - | # After cells have grown up, place in flourophore (1 um) and allow time to bind (1 hour)
| + | |
| - | # Also place 1 ul beads in 1 ml along with 1 ul flourophore.
| + | |
| - | # Allow beads to bind to flourophore, then spin beads down, remove supernatent and replace with 1 ml water.
| + | |
| - | # Next place the cells and the beads under the microscope
| + | |
| - | | + | |
| - | ==== Data ====
| + | |
| - | [[Image:Hists.png | 800px]]
| + | |
| - | | + | |
| - | From this data we can see that while the positive control did have a visible increase in florescence around the outside, the cells did not show any such increase.
| + | |
| - | | + | |
| - | | + | |
| - | === Flow Cytometry ===
| + | |
| - | The goal of this experiment was to see how the biotin bonded to streptavadin over a whole population of cells. Where as the Microscope experiment looked at a few localized cells, we set the cytometer to look 50,000 cells or beads and read the resulting florescence.
| + | |
| - | | + | |
| - | ==== Procedure ====
| + | |
| - | # Set up overnights of parts 48-51. Let grow overnight.
| + | |
| - | # Dilute 1 ul overnight into 1ml
| + | |
| - | # Add 1 mm IPTG and let grow for four hours
| + | |
| - | # After cells have grown up, place in flourophore (1 um) and allow time to bind (1 hour)
| + | |
| - | # Also place 1 ul beads in 1 ml along with 1 ul flourophore.
| + | |
| - | # Allow beads to bind to flourophore, then spin beads down, remove supernatent and replace with 1 ml water.
| + | |
| - | # Read the samples through the flow cytometer
| + | |
| - | | + | |
| | | | |
| - | ==== Data ====
| + | Recombinant, purified protein production is a decades-old technology that has revolutionized research in biotechnology and medicine. However, the traditional method of purified protein production is a time-consuming and laborious procedure requiring expensive and specialized equipment. Our project, the Idealized Protein Purification (IPP) system, is an all-in-one protein expression and purification platform built on BioBrick standards that will reduce costs, save time, and simplify procedures associated with recombinant protein production. The key to our IPP system is a novel combination of three subsystems: expression, secretion and display. We use ''E. coli'' bacteria that we have genetically modified to be a chassis for expressing your favorite protein, secreting it to the media, then binding and displaying the protein on the cell surface. At this point, collecting your favorite ''purified protein'' is as simple as pelleting and re-suspending a sufficient quantity of bacterial cells in an elution buffer. The speed and simplicity of our IPP system exhibits the utility of synthetic biology for developing new techniques that improve upon established practices. |
| | | | |
| - | [[Image:2009-08-07_1uL_Beads_%2B_Fluoro_%26_Control.png]] | + | <div style="text-align:right">'''Continue to [https://2009.igem.org/Team:Washington/Project Project Description >]'''</div> |
| - | [[Image:2009-08-07_51s.png]]
| + | |
| | | | |
| - | Again this data showed us that we did not have any appreciable binding occurring with our display system to biotin at the global level
| + | {{Template:Team:Washington/Templates/Footer}} |
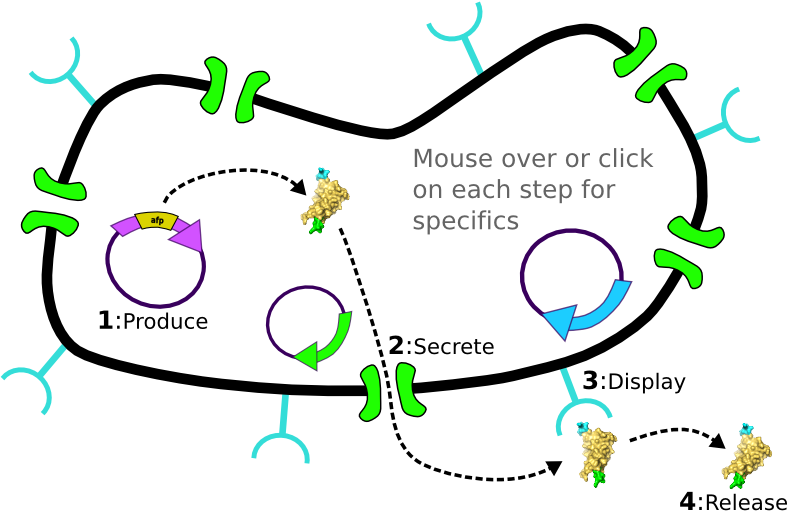
The Idealized Protein Purification System: Improving the lives of molecular biologists
Recombinant, purified protein production is a decades-old technology that has revolutionized research in biotechnology and medicine. However, the traditional method of purified protein production is a time-consuming and laborious procedure requiring expensive and specialized equipment. Our project, the Idealized Protein Purification (IPP) system, is an all-in-one protein expression and purification platform built on BioBrick standards that will reduce costs, save time, and simplify procedures associated with recombinant protein production. The key to our IPP system is a novel combination of three subsystems: expression, secretion and display. We use E. coli bacteria that we have genetically modified to be a chassis for expressing your favorite protein, secreting it to the media, then binding and displaying the protein on the cell surface. At this point, collecting your favorite purified protein is as simple as pelleting and re-suspending a sufficient quantity of bacterial cells in an elution buffer. The speed and simplicity of our IPP system exhibits the utility of synthetic biology for developing new techniques that improve upon established practices.

 "
"