Team:Waterloo/Project
From 2009.igem.org
m (→The Goal of on Chromosome Assembly of Interchangeable parts) |
(→The Goal of on Chromosome Assembly of Interchangeable Parts) |
||
| Line 39: | Line 39: | ||
=====The Goal of on Chromosome Assembly of Interchangeable Parts===== | =====The Goal of on Chromosome Assembly of Interchangeable Parts===== | ||
| - | With the design of non-cross reactive ''att'' sites it would be possible to regenerate the landing pad after a successful integration. This would be done by having a second non-cross-reactive pair of ''att'' sites downstream of our integrated BioBrick (attP2 attP2). A landing pad regeneration plasmid with attB2 sites flanking a landing pad would be introduced and a ΦC31 integrase reaction would integrate the landing pad. Another attP1 attB1 reaction could be carried out to integrate another BioBrick. The problem with an approach like this is that attL sites would build up. We decided to write software to try and solve this problem. The software is further discussed in the modelling section. | + | With the design of non-cross reactive ''att'' sites it would be possible to regenerate the landing pad after a successful integration. This would be done by having a second non-cross-reactive pair of ''att'' sites downstream of our integrated BioBrick (''attP2 attP2''). A landing pad regeneration plasmid with attB2 sites flanking a landing pad would be introduced and a ΦC31 integrase reaction would integrate the landing pad. Another ''attP1 attB1'' reaction could be carried out to integrate another BioBrick. The problem with an approach like this is that ''attL'' sites would build up. We decided to write software to try and solve this problem. The software is further discussed in the modelling section. |
== The Experiments == | == The Experiments == | ||
Revision as of 22:42, 12 October 2009
| Home | The Team | The Project | Parts Submitted to the Registry | Modeling | Notebook |
|---|
(Or you can choose different headings. But you must have a team page, a project page, and a notebook page.)
Contents |
Abstract:
Chromobricks: A Platform for Chromosome Engineering with BioBricks
The aim of our project is to develop a fully-featured platform for chromosome engineering, allowing the in vivo assembly of a synthetic chromosome from interchangeable parts, followed by selective degradation of the native chromosome. We have designed a proof-of-concept for chromosome-building that will use the site-specific integrase of phage ΦC31 to integrate a BioBrick into a defined locus of the E. coli genome. Six pairs of integrase-targeted att sites have been designed to be non-cross-reactive in order to support repeatable cassette-exchange reactions for chromosome building. We have also written software to model the integrase-mediated rearrangement of DNA molecules containing att sites, to aid the design of more elaborate chromosome-building systems. To selectively degrade the native chromosome we designed a nuclease-based, inducible genome-degradation system. In its simplest form, our system can be used to integrate biological devices into a chromosome in situations requiring stable copy number and selection-free maintenance.
Project Details
Background
Chromosome Engineering provides many benefits over plasmid based engineering, including precise copy number and stable maintenance without selection.Imprecise copy number plasmid-based systems can add unnecessary noise to synthetic biology systems, limiting the potential complexity of these systems. However, a plasmid can easily be isolated, engineered with restriction enzymes and reintroduced into a host strain. Chromosomes cannot be engineered in this way.
Traditionally, methods to engineer chromosomes in bacteria have been based on homologous recombination, which is an inefficient process. Other methods have taken advantage of recombinant lysogenic phage, such as the E. coli phage λ. These phage integrate their genome into the bacterial chromosome via a site-specific recombination. Genes that have been added to the small phage genome will be carried along. The mechanisms of phage genome integration have been studied and there are some well characterized systems. These systems can be separated from the phage itself. They can be used to engineer DNA molecules in vitro or in vivo without actually infecting the bacteria with a phage (Groth, 2004).
The phage integration system usually involves an integrase/recombinase enzyme, the phage attachment site (attP), the bacterial attachment site (attB), and for the reverse reaction an excisionase enzyme. The integrase enzyme catalyzes a stand exchange between attP and attB sites on the DNA yielding attL and attR sites which are composites of attP and attB sites. If there is excisionase and integrase present the reaction will go in the reverse direction. The well known λ integration system is the basis of Invitrogen's Gateway® technology. Gateway® technology uses the activity of the λ integrase and excisionase to carry out cassette exchange reactions, where sections of two molecules each flanked by respective att sites are exchanged. The system is used to engineer plasmids as an alternative to traditional cloning using restriction enzymes. For our project, the goal of engineering chromosomes, without conflicting with the widely adopted Gateway® system, led us to choose the integrase from the Streptomyces phage C31 (ΦC31).
The ΦC31 Integrase
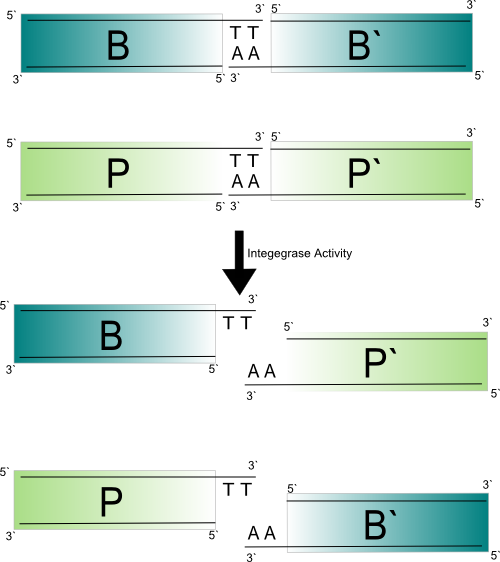
The ΦC31 integrase catalyzes a unidirectional stand exchange between a 39 bp attP site and a 34 bp attB site. The enzyme works by making a synapse between attP and attB, making double strand breaks producing a 2 bp sticky overhang, exchanging the strands, and re-ligating them in the recombinant configuration (attL and attR).
Our BBa Compatible Cassette Exchange System
Due to the lack of any BBa compatible chromosome engineering tools we decided to build a cassette exchange system based on the ΦC31 integrase. The system can be broken down into three components: the landing pad strain (LPS), the helper plasmid, and the BBa donor vector.
The protocol to integrate a BioBrick into the chromosome of the LPS involves a conjugation of a section of the donor plasmid via bi-parental mating into the LPS which is also expressing ΦC31 integrase. Then the LPS strain is selected with naladixic acid, and the loss of the landing pad markers is selected with sucrose. The colonies would then be screened for kanamycin sensitivity. The LPS contains a Landing Pad cassette in the melA locus. This cassette consists of sacB,rfp,and nac(kanamycin resistance) genes flanked by ΦC31 attP sites. After cassette exchange these markers are lost. The BioBrick will replace them.
The Goal of on Chromosome Assembly of Interchangeable Parts
With the design of non-cross reactive att sites it would be possible to regenerate the landing pad after a successful integration. This would be done by having a second non-cross-reactive pair of att sites downstream of our integrated BioBrick (attP2 attP2). A landing pad regeneration plasmid with attB2 sites flanking a landing pad would be introduced and a ΦC31 integrase reaction would integrate the landing pad. Another attP1 attB1 reaction could be carried out to integrate another BioBrick. The problem with an approach like this is that attL sites would build up. We decided to write software to try and solve this problem. The software is further discussed in the modelling section.
The Experiments
Cassette Exchange
BBa Donor Plasmid
Landing Pad Strain
Integrase expression plasmid
Non-cross-reactive att sites
We set out to design and test 6 different att site pairs that would be non-cross-reactive. This would allow for increased specificity. It would also allow for repeatable insertion of DNA pieces into the chromosome as long as a build up of attL sites was not an issue.
First we discuss the mechanism of the integrase; then we move on to the design and testing of our "orthogonal att sites".
Integrase Mechanism
The ΦC31 integrase catalyzes a unidirectional stand exchange between a 39 bp attP site and a 34 bp attB site. The enzyme works by making a synapse between attP and attB, making double strand breaks producing a 2 bp sticky overhang, exchanging the strands, and re-ligating them in the recombinant configuration (attL and attR). This reaction is not reversible with only the integrase present. The att sites can be mutated to produce different overhangs. A mutant attB with different a different overhang (A.K.A core dinucleotide)has been shown, in a related system, not to recombine with attP (Ghosh 2003). Instead of a recombination activity the integrase showed a topoisomerase activity (reducing supercoiling on a plasmid) suggesting the double stranded breaks were made but the DNA would not re-ligate in the recombinant configuration (Ghosh 2003). When indentical mutations are made to the overhangs the recombination activity is restored (Ghosh 2003). This implies that making orthogonal att sites (non cross-reactive site pairs) for the ΦC31 integrase is possible.
The sequence of the overhangs also has implications in the directionality of the recombination, which can lead to different end results of recombination (excision or inversion for example). The palindromic overhangs will lead to loss of direction specificity. Given the 16 possible core dinucleotide sequences six of them should be completely non-cross reactive while maintaining direction specificity. Integrase Mechanism:
 "
"