Team:KULeuven/Project Results
From 2009.igem.org
JochemDeen (Talk | contribs) (→Project Results) |
(→Project Results) |
||
| (12 intermediate revisions not shown) | |||
| Line 8: | Line 8: | ||
Our Results can be roughly divided in two sections, the biobrick assembly from starting parts in order to create our (test)-devices and secondly the characterizations of a part. | Our Results can be roughly divided in two sections, the biobrick assembly from starting parts in order to create our (test)-devices and secondly the characterizations of a part. | ||
| - | First and foremost | + | First and foremost the following biobricks were succesfully assembled and sent to the registry: |
{| border ="0" align="Justify" | {| border ="0" align="Justify" | ||
| Line 16: | Line 16: | ||
|{{kulpart|K238005}} ||Antisense key | |{{kulpart|K238005}} ||Antisense key | ||
|- align="justify" | |- align="justify" | ||
| - | |{{kulpart|K238007}} ||Sam5 + rbs | + | |{{kulpart|K238007}} ||Sam5 + rbs, the DNA for this part was not yet available in the registry, the DNA has been submitted and should now be available to everybody! |
|- align="justify" | |- align="justify" | ||
|{{kulpart|K238008}} ||virA | |{{kulpart|K238008}} ||virA | ||
| Line 24: | Line 24: | ||
|{{kulpart|K238011}} ||VirB | |{{kulpart|K238011}} ||VirB | ||
|- align="justify" | |- align="justify" | ||
| - | |{{kulpart|K238013}} ||Blue light | + | |{{kulpart|K238013}} ||Blue light promoter |
|- align="justify" | |- align="justify" | ||
|{{kulpart|K238014}} ||Ech+fcs | |{{kulpart|K238014}} ||Ech+fcs | ||
| Line 44: | Line 44: | ||
|{{kulpart|K238022}} ||Promotor + Sam8 + Sam5 + comt + terminator | |{{kulpart|K238022}} ||Promotor + Sam8 + Sam5 + comt + terminator | ||
|- align="justify" | |- align="justify" | ||
| - | |{{kulpart|K238024}} ||lock | + | |{{kulpart|K238024}} ||lock, the DNA for this part was not yet available in the registry, the DNA has been submitted and should now be available to everybody! |
|- align="justify" | |- align="justify" | ||
|{{kulpart|K238027}} ||Sam8 + RBS | |{{kulpart|K238027}} ||Sam8 + RBS | ||
| Line 53: | Line 53: | ||
|} | |} | ||
| + | '''Helping out other teams''' | ||
| - | Furthermore, the blue light | + | We helped TU Delft with their [http://spreadsheets.google.com/viewform?formkey=dElEZjI4Vk0wWmxHTWoycG03b2RrQ3c6MA.. survey on reductionism in synthetic biology]. Follow the results on their [https://2009.igem.org/Team:TUDelft wiki]. |
| + | |||
| + | We completed the [https://2009.igem.org/Team:Valencia/Human survey from the University of Valencia]. | ||
| + | <html><a href="https://2009.igem.org/Team:Valencia/Human" target="_blank"> <img src="https://static.igem.org/mediawiki/2009/3/38/V_KULeuven.JPG" width="110" height="110"></a></html> | ||
| + | |||
| + | We sent in some parts that were designed by the Berkeley 2006 team (lock) and the Edinburgh 2007 team (vanillin synthesis, thanks to Chris French!). See above for parts! | ||
| + | |||
| + | '''Experimental results''' | ||
| + | |||
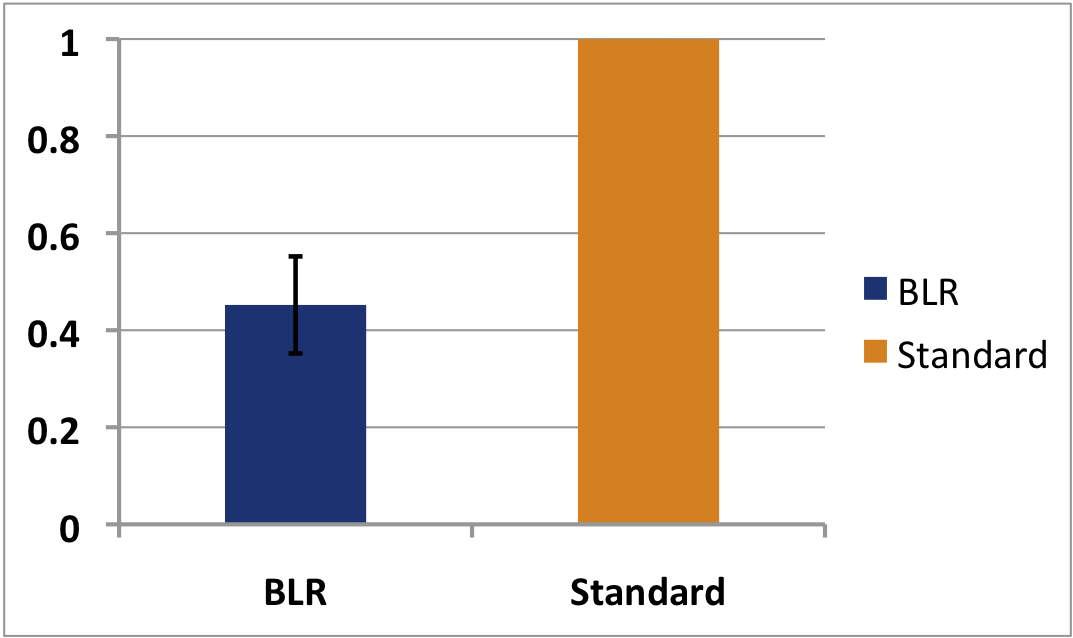
| + | Furthermore, the blue light promotor was extensively characterized. The strength of the promoter was compared to the standard iGEM promoter, {{kulpart|I20260}}, using the protocol from the iGEM measurement kit. | ||
{| | {| | ||
| - | | [[Image:BLPresult1.png|thumb|380px|Comparison of the blue light promoter (blue) to a standard promoter strength {{kulpart|I20260}} (orange) in RPU. The blue light promoter shows | + | | [[Image:BLPresult1.png|thumb|380px|Comparison of the blue light promoter (blue) to a standard promoter strength {{kulpart|I20260}} (orange) in RPU. The blue light promoter shows ca. half the strength of the standard promoter, meaning it can be classified as a weak promoter]] |
|} | |} | ||
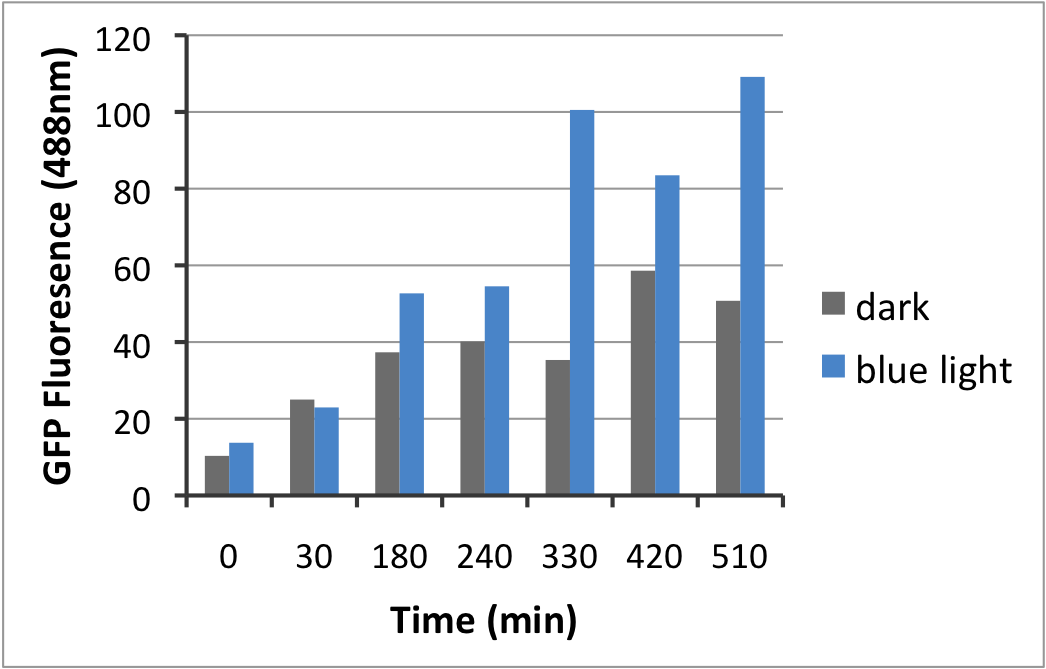
| - | + | Next, the effect of blue light on this promoter was examined. As shown on the graph below, blue light irradiation has a significant effect on the strength of the promoter. However, aside from light, temperature also has an important effect on promotor strenght (possibly even bigger then light). The influence of temperature will be on the amount of repressor available compared to the plasmid copies. For example, cells grown at 37°C will have too little repressor, resulting in constitutive activity. At 16°C, there will be more repressor and more receptor, however the low temperature will put the receptor in its active state causing it to inhibit the repressor from doing its job. When grown at 25°C the ratio between repressor and receptor is better, without light irradiance the receptor/represssor are in the ground state resulting in a good repression of the promoter, when exposing the cells to light the repressor is removed and the promotor is active. | |
| - | + | ||
{| | {| | ||
| Line 67: | Line 76: | ||
|} | |} | ||
| - | + | '''Experimental set-up''' | |
| - | + | Liquid cultures were ented (and re-ented in the evening) and grown on two different temperatures: 37° and 25°C. Note: enting of the cultures needs to be done in a dark room (red light)and it might be best to use medium that was put at 4°C overnight. | |
| - | + | From each of these growth conditions some of the cultures were put in 16° and the other at 25°C. Each of these groups were then again divided in a group that was exposed to blue light and a group that was not exposed to light. | |
| - | + | We decided upon using FACS because then the fluorescence of single cells is measured. Since each time the fluorescence of 10,000 individual cells is measured, FACS allows more elaborate and accurate statistical analysis. Our samples were analyzed by flow cytometry with a Becton Dickinson FACSCalibur and CELLQuestTM acquisition and analysis software with gates set to forward and side scatters characteristic of the bacteria (Gate G2=R2). | |
| - | + | Measurements were done at following hours: 0h;0,5h;1,5h;3h;4,5h;6h;8h. | |
| - | + | The construct used for our test was a ligation of the blue light promotor in front of GFP using three different vectors (pSB3K3, pBR322 and pSB1A2). (An example of a sample name is: 37/16/NB/LigA, grown at 37°, put in 16°, not lit and in vector pSB1A2. ) | |
| - | + | It's important to know that the regulation of this promotor is very sensitive and that all factors need to be calculated into the experimental setup. For instance, since temperature has an equally high effect on the promotor as blue light has, cultures grown at 37°C will have a different GFP signal compared to cells grown at 25°C. To learn more about the mechanism, we refere to the following articles: | |
| - | + | *Tschowri, N., S. Busse, and R. Hengge. 2009. The BLUF-EAL protein YcgF acts as a direct anti-repressor in a blue-light response of Escherichia coli. Genes Dev. 23:522–534. | |
| - | + | *Gomelsky M., Klug G. BLUF: A novel FAD-binding domain involved in sensory transduction in microorganisms. Trends Biochem. Sci. 2002;27:497–500 | |
| + | *Nakasone Y., Ono T.A., Ishii A., Masuda S., Terazima M. Transient dimerization and conformational change of a BLUF protein: YcgF. J. Am. Chem. Soc. 2007;129:7028–7035 | ||
Latest revision as of 20:10, 21 October 2009
Project Results
Our Results can be roughly divided in two sections, the biobrick assembly from starting parts in order to create our (test)-devices and secondly the characterizations of a part.
First and foremost the following biobricks were succesfully assembled and sent to the registry:
| Partnumber | Description |
|---|---|
| Antisense key | |
| Sam5 + rbs, the DNA for this part was not yet available in the registry, the DNA has been submitted and should now be available to everybody! | |
| virA | |
| VirG | |
| VirB | |
| Blue light promoter | |
| Ech+fcs | |
| Blp + GFP | |
| Blp + RFP | |
| Ech + Fcs + terminator | |
| Promotor+ Ech + FCS + terminator | |
| Sam8 +Sam8 | |
| Sam8 + Sam5 + promoter | |
| Sam8+ Sam5 + promoter + comt | |
| Promotor + Sam8 + Sam5 + comt + terminator | |
| lock, the DNA for this part was not yet available in the registry, the DNA has been submitted and should now be available to everybody! | |
| Sam8 + RBS | |
| Ech + rbs | |
| Fcs + rbs |
Helping out other teams
We helped TU Delft with their [http://spreadsheets.google.com/viewform?formkey=dElEZjI4Vk0wWmxHTWoycG03b2RrQ3c6MA.. survey on reductionism in synthetic biology]. Follow the results on their wiki.
We completed the survey from the University of Valencia.

We sent in some parts that were designed by the Berkeley 2006 team (lock) and the Edinburgh 2007 team (vanillin synthesis, thanks to Chris French!). See above for parts!
Experimental results
Furthermore, the blue light promotor was extensively characterized. The strength of the promoter was compared to the standard iGEM promoter, , using the protocol from the iGEM measurement kit.
Next, the effect of blue light on this promoter was examined. As shown on the graph below, blue light irradiation has a significant effect on the strength of the promoter. However, aside from light, temperature also has an important effect on promotor strenght (possibly even bigger then light). The influence of temperature will be on the amount of repressor available compared to the plasmid copies. For example, cells grown at 37°C will have too little repressor, resulting in constitutive activity. At 16°C, there will be more repressor and more receptor, however the low temperature will put the receptor in its active state causing it to inhibit the repressor from doing its job. When grown at 25°C the ratio between repressor and receptor is better, without light irradiance the receptor/represssor are in the ground state resulting in a good repression of the promoter, when exposing the cells to light the repressor is removed and the promotor is active.
Experimental set-up
Liquid cultures were ented (and re-ented in the evening) and grown on two different temperatures: 37° and 25°C. Note: enting of the cultures needs to be done in a dark room (red light)and it might be best to use medium that was put at 4°C overnight. From each of these growth conditions some of the cultures were put in 16° and the other at 25°C. Each of these groups were then again divided in a group that was exposed to blue light and a group that was not exposed to light. We decided upon using FACS because then the fluorescence of single cells is measured. Since each time the fluorescence of 10,000 individual cells is measured, FACS allows more elaborate and accurate statistical analysis. Our samples were analyzed by flow cytometry with a Becton Dickinson FACSCalibur and CELLQuestTM acquisition and analysis software with gates set to forward and side scatters characteristic of the bacteria (Gate G2=R2). Measurements were done at following hours: 0h;0,5h;1,5h;3h;4,5h;6h;8h. The construct used for our test was a ligation of the blue light promotor in front of GFP using three different vectors (pSB3K3, pBR322 and pSB1A2). (An example of a sample name is: 37/16/NB/LigA, grown at 37°, put in 16°, not lit and in vector pSB1A2. ) It's important to know that the regulation of this promotor is very sensitive and that all factors need to be calculated into the experimental setup. For instance, since temperature has an equally high effect on the promotor as blue light has, cultures grown at 37°C will have a different GFP signal compared to cells grown at 25°C. To learn more about the mechanism, we refere to the following articles:
- Tschowri, N., S. Busse, and R. Hengge. 2009. The BLUF-EAL protein YcgF acts as a direct anti-repressor in a blue-light response of Escherichia coli. Genes Dev. 23:522–534.
- Gomelsky M., Klug G. BLUF: A novel FAD-binding domain involved in sensory transduction in microorganisms. Trends Biochem. Sci. 2002;27:497–500
- Nakasone Y., Ono T.A., Ishii A., Masuda S., Terazima M. Transient dimerization and conformational change of a BLUF protein: YcgF. J. Am. Chem. Soc. 2007;129:7028–7035
 "
"