Team:IIT Madras/Experiments
From 2009.igem.org
(New page: {{:Team:IITM/main}} <html> <br><br><br><br><br><br><br><br><br><br><br> <head><style> body { background: #000; } #aContent { background-color:#fff; margin: 50px auto; padding: 10px...) |
(→Modeling) |
||
| (60 intermediate revisions not shown) | |||
| Line 2: | Line 2: | ||
<html> | <html> | ||
| - | <br><br><br><br><br><br><br><br><br><br><br> | + | <br><br><br><br><br><br><br><br><br><br><br><br> |
<head><style> | <head><style> | ||
| Line 34: | Line 34: | ||
<div id="aContent"> | <div id="aContent"> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
</html> | </html> | ||
| + | ==Experiments== | ||
| + | ===Comparing the differences in the growth rates of cells with and without plasmids in various media=== | ||
| - | + | <p CLASS="justifyalign">We wish to compare the growth rate of the cells that are transformed with a plasmid which shows a constitutive expression of a certain protein to that of growth rate of cells which are not transformed with any plasmids. To study this pattern, we need to grow the different strains of cells in various media as shown in the figure 7. | |
| - | + | ||
| - | <p CLASS="justifyalign"> | + | |
| - | + | ||
| - | + | ||
</p> | </p> | ||
| - | <html><br></html> | + | [[Image:growth curves.jpg|700px]] |
| + | <html><br> | ||
| + | <b><font color="#000">Fig 7: </font></b><i>This experiment helps in comparing the growth rates of various strains in different antibiotic media.</i> | ||
| + | </html> | ||
| - | + | <p CLASS="justifyalign">We inoculate a colony from the plate containing the required strain into its corresponding media (the media with the required antibiotic). That is, DH5a will be inoculated into LB without any antibiotic, RFP (in pSB1C3)containing cells will be inoculated into LB containing Chloramphenicol (Chl), CFP (in pSB1A2)containing cells will be inoculated into LB containing Ampicillin (Amp) and RFP-CFP co-transformed cells into LB containing both the antibiotics. These are then grown for about 4 hours or till they reach an OD600 value between 0.1 to 0.5.</p> | |
| - | + | <p CLASS="justifyalign">Then the culture of each strain is taken and inoculated into each of the 5ml broths which contains no antibiotic, Amp, Chl and Amp-Chl so that the OD value in all the freshly inoculated tubes is 0.01. Now we have an array of 16 tubes with various possible combinations: | |
| - | + | ||
| - | + | ||
| - | <p CLASS="justifyalign"> | + | |
| - | + | ||
</p> | </p> | ||
| + | * DH5a in no antibiotic, DH5a in Amp, DH5a in Chl and Dh5a in Amp-Chl | ||
| + | * RFP (pSB1C3) in no antibiotic, RFP (pSB1C3)in Amp, RFP (pSB1C3)in Chl and RFP (pSB1C3)in Amp-Chl | ||
| + | * CFP (pSB1A2) in no antibiotic, CFP (pSB1A2)in Amp, CFP (pSB1A2)in Chl and CFP (pSB1A2)in Amp-Chl | ||
| + | * RFP (1C3)-CFP (1A2) in no antibiotic, RFP (1C3)-CFP (1A2) in Amp, RFP (1C3)-CFP (1A2) in Chl and RFP (1C3)-CFP (1A2)in Amp-Chl | ||
| - | < | + | <p CLASS="justifyalign">Thus the starting point for all the samples is the same - an OD600 of 0.01. |
| + | Every hour starting from the point of inoculation, the OD of all the 16 samples is measured. This would give a fair idea of the growth rates of various strains in different antibiotic media.</p> | ||
| - | < | + | <b><font color="#000">Experimental protocol:</font></b> |
| - | + | ||
| - | == | + | <p CLASS="justifyalign">1. A colony of DH5a is inoculated into 3ml LB without any antibiotic in a 50ml centrifuge tube. Similarly, RFP (1C3) colony is inoculated into 3ml LB with Chl, CFP (1A2) colony into 3ml LB with Amp and RFP-CFP colony into 3ml LB with Amp-Chl.</p> |
| + | <p CLASS="justifyalign">2. This inoculum is allowed to grow for about 4 hours or till the OD600 of each sample crosses 0.1.</p> | ||
| + | <p CLASS="justifyalign">3. Then we use OD 1 x Vol 1 = OD 2 x Vol 2 to measure how much to inoculte from this 4 hour culture to each of the fresh 5ml LB medium with different antibiotic combinations so that the starting OD is 0.01. In this case, OD 1 is the OD600 of the 4 hour culture, Vol 1 is the volume of this 4 hour culture that needs to be inoculted in to the fresh 5ml culture, OD 2 is 0.01 (starting OD for all the 5 ml cultures) and the Vol 2 is the final volume (5ml + vol 1).</p> | ||
| + | <p CLASS="justifyalign">4. From this freshly inoculated sample, 150ul of the culture is used to measure the OD every hour starting from the point if inoculation. The 150ul of the sample is diluted 5 times to 750ul and then the OD600 is measured.</p> | ||
| + | <p CLASS="justifyalign">5. The whole procedure is repeated to check for reproducibility.</p> | ||
| - | <p CLASS="justifyalign"> | + | <p CLASS="justifyalign">Note: After the initial 4 hour incubation (the 3ml cultures), we used this to inoculate a 5ml culture with the same antibiotic in the medium as in the 3ml culture. The OD600 of this 5ml tube after the inoculation was 0.01. This was then made to grow for 2.5 hours. This is the culture from which we inoculated the final 16 tubes which would then be used for measurements.</p> |
| - | + | Click [https://2009.igem.org/Team:IIT_Madras/Results#Comparing_the_differences_in_the_growth_rates_of_cells_with_and_without_plasmids_in_various_media here] for the results | |
| - | + | ||
| - | + | ===Modeling=== | |
| - | <html><br></ | + | <html><br> |
| + | <b><font color="#000">Basic Equations:</font></b> | ||
| + | <br> | ||
| + | <img src="https://static.igem.org/mediawiki/2009/8/89/Modeling.jpg" align="center" width="400" height="200"></a> | ||
| + | <br> | ||
| - | <p | + | <p>Where,</p> |
| - | < | + | <p>X<sup>+</sup> is the cells with the plasmid.</p> |
| + | <p>X<sup>-</sup> is the cells without the plasmid.</p> | ||
| + | <p>u<sup>+</sup> is the specific cell growth rate of cells with the plasmid.</p> | ||
| + | <p>u<sup>-</sup> is the specific cell growth rate of cells without the plasmid.</p> | ||
| + | <p>p is the probability of a cell to lose a plasmid.</p> | ||
| + | <br> | ||
| - | < | + | <p>We are trying to come up with a mathematical model for the relative populations of plasmid containing and plasmid free cells with respect to time using the growth curves that have been generated and the results from the fluorescence.</p> |
| - | + | ||
| - | + | </html> | |
| - | + | ===Fluorescence Imaging of cells to check for directed plasmid loss=== | |
| - | |||
| - | |||
| - | + | This experiment is designed to study the plasmid loss when cells (transformed with a plasmid) are grown in media without the required selection pressures. Here, we perform 4 different experiments to study how | |
| - | + | # Cells transformed with RFP (pSB1C3) lose the plasmid in the absence of Chloramphenicol in the medium | |
| - | + | # Cells transformed with CFP (pSB1A2) lose the plasmid in the absence of Ampicillin in the medium. | |
| - | + | # Cell cotransformed with RFP (pSB1C3) and CFP (pSB1A2) lose the RFP (pSB1C3) plasmid when grown in absence of Chloramphenicol and in the presence of Ampicillin in the medium. | |
| - | + | # Cell cotransformed with RFP (pSB1C3) and CFP (pSB1A2) lose the CFP (pSB1A2) plasmid when grown in absence of Ampicillin and in the presence of Chloramphenicol in the medium. | |
| - | + | ||
| - | + | ||
<html><br></html> | <html><br></html> | ||
| - | + | [[Image:flourescenceimagingexp.jpg|700px]] | |
| - | + | ||
| - | + | ||
<html><br></html> | <html><br></html> | ||
| - | + | <html><b><font color="#000">Fig 6.3: </font></b><i>Experimental procedure for measuring the rate of plasmid loss by fluorescent imaging</i> | |
| - | + | ||
| - | + | ||
| - | < | + | |
| - | + | ||
| - | + | ||
| - | + | ||
<html><br></html> | <html><br></html> | ||
| - | |||
| - | + | We pick up colonies of each strain and inoculate it first in a medium with the appropriate antibiotic. Then use this to inoculate in specific growth media as show in the figure 6.3. | |
| - | + | # RFP (pSB1C3) containing cells will be grown in medium containing no antibiotic and in medium containing Chloramphenicol | |
| + | # CFP (pSB1A2) containing cells will be grown in medium containing no antibiotic and in medium containing Ampicillin. | ||
| + | # Co-transformed cell will be grown in medium containing no antibiotic, medium containing only Ampicillin, medium containing only Chloramphenicol, medium containing both Ampicillin and Chloramphenicol. | ||
| - | |||
| - | |||
| - | + | The expected behavior of the system is (listed in the same order as the above 3 conditions): | |
| - | + | # RFP (pSB1C3)containing Cells grown in medium with no Chloramphenicol would eventually lose the plasmid and hence show no Red fluorescence when imaged under the microscope and those which are grown in the chloramphenicol containing medium would all be fluorescing red. | |
| + | # CFP (pSB1A2)containing cells grown in medium with no Ampicillin would eventually lose the plasmid and hence show no Cyan fluorescence and those which are grown in the Ampicillin containing medium would all be fluorescing Cyan. | ||
| + | # Cotransformed cells grown in medium with no antibiotic would eventually lose both the plasmids and hence show no red or Cyan fluorescence, those which are grown in the Ampicillin containing medium would all be fluorescing Cyan, those which are grown in the Chloramphenicol containing medium would all be fluorescing Red and those which are grown in medium containing both the Antibiotics would all be fluorescing both Cyan and Red. | ||
| - | |||
| - | |||
| - | |||
| - | |||
<html><br></html> | <html><br></html> | ||
| - | + | <html><b><font color="#000">Protocol: </font></b> | |
| - | + | <html><br></html><html><br></html> | |
| + | 1. Inoculate 3ml LB containing Ampicillin with a colony of cells transformed with CFP (pSB1A2)- broth 1, inoculate 3ml LB containing Chloramphenicol with a colony of cells transformed with RFP (pSB1C3) - broth 2, inoculate 3ml LB containing Ampicillin and Chloramphenicol with a colony of cells co-transformed with CFP (pSB1A2)and RFP (pSB1C3) - broth 3. | ||
<html><br></html> | <html><br></html> | ||
| - | + | 2. Culture is grown for 4 hours or till the OD600 crosses 0.1. | |
| - | + | ||
| - | + | ||
| - | + | ||
<html><br></html> | <html><br></html> | ||
| - | Then we | + | 3. Then we use OD 1 x Vol 1 = OD 2 x Vol 2 to measure how much to inoculate from this 4 hour culture to each of the fresh 5ml LB medium with different antibiotic combinations so that the starting OD is 0.01. In this case, OD 1 is the OD600 of the 4 hour culture, Vol 1 is the volume of this 4 hour culture that needs to be inoculated in to the fresh 5ml culture, OD 2 is 0.01 (starting OD for all the 5 ml cultures) and the Vol 2 is the final volume (5ml + vol 1). |
| - | + | ||
<html><br></html> | <html><br></html> | ||
| - | |||
| - | |||
| - | |||
<html><br></html> | <html><br></html> | ||
| - | + | Required inoculations are: | |
| - | + | a. Broth 1 into tube with no antibiotic in the LB - tube 1 | |
| - | + | ||
| - | + | ||
<html><br></html> | <html><br></html> | ||
| + | b. Broth 1 into tube with Ampicillin in the LB - tube 2 | ||
<html><br></html> | <html><br></html> | ||
| - | + | c. Broth 2 into tube with no antibioti in the LB - tube 3 | |
| - | + | ||
| - | + | ||
<html><br></html> | <html><br></html> | ||
| - | + | d. Broth 2 into tube with Chloramphenicol in the LB - tube 4 | |
| - | + | ||
| - | + | ||
<html><br></html> | <html><br></html> | ||
| - | + | e. Broth 3 into tube no antibiotic in the LB - tube 5 | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
<html><br></html> | <html><br></html> | ||
| - | + | f. Broth 3 into tube with Ampicillin in the LB - tube 6 | |
| - | + | ||
| - | + | ||
<html><br></html> | <html><br></html> | ||
| - | + | g. Broth 3 into tube with Chloramphenicol in the LB - tube 7 | |
| - | + | ||
<html><br></html> | <html><br></html> | ||
| - | + | h. Broth 3 into tube with both the antibiotics in the LB - tube 8 | |
| - | + | ||
| - | + | ||
<html><br></html> | <html><br></html> | ||
| - | |||
| - | |||
| - | |||
<html><br></html> | <html><br></html> | ||
| - | + | 4. This culture is allowed to grow for 2.5 hours. | |
| - | + | ||
| - | + | ||
<html><br></html> | <html><br></html> | ||
| + | 5. Measure the OD at this stage and also prepare a slide for fluorescence imaging from each of the 8 tubes. | ||
<html><br></html> | <html><br></html> | ||
| + | 6. Then we use OD 1 x Vol 1 = OD 2 x Vol 2 to measure how much to inoculate from this 2.5 hour culture to each of the fresh 5ml LB medium with the same antibiotic combinations so that the starting OD is 0.01. In this case, OD 1 is the OD600 of the 2.5 hour culture, Vol 1 is the volume of this 2.5 hour culture that needs to be inoculated in to the fresh 5ml culture, OD 2 is 0.01 (starting OD for all the 5 ml cultures) and the Vol 2 is the final volume (5ml + vol 1). i.e. The culture from spent tube 1 is used to inoculte a fresh tube 1. Similarly follow the same for all the other 7 tubes. | ||
| + | 7. Let this new culture grow for 2.5 hours and repeat from step 5 for about 5 times. | ||
<html><br></html> | <html><br></html> | ||
| - | + | Click [https://2009.igem.org/Team:IIT_Madras/Results#Fluorescent_Imaging here] to see the results of the fluorescence imaging. | |
| - | + | {{:Team:IITM/footer}} | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
Latest revision as of 00:18, 22 October 2009
Contents |
Experiments
Comparing the differences in the growth rates of cells with and without plasmids in various media
We wish to compare the growth rate of the cells that are transformed with a plasmid which shows a constitutive expression of a certain protein to that of growth rate of cells which are not transformed with any plasmids. To study this pattern, we need to grow the different strains of cells in various media as shown in the figure 7.
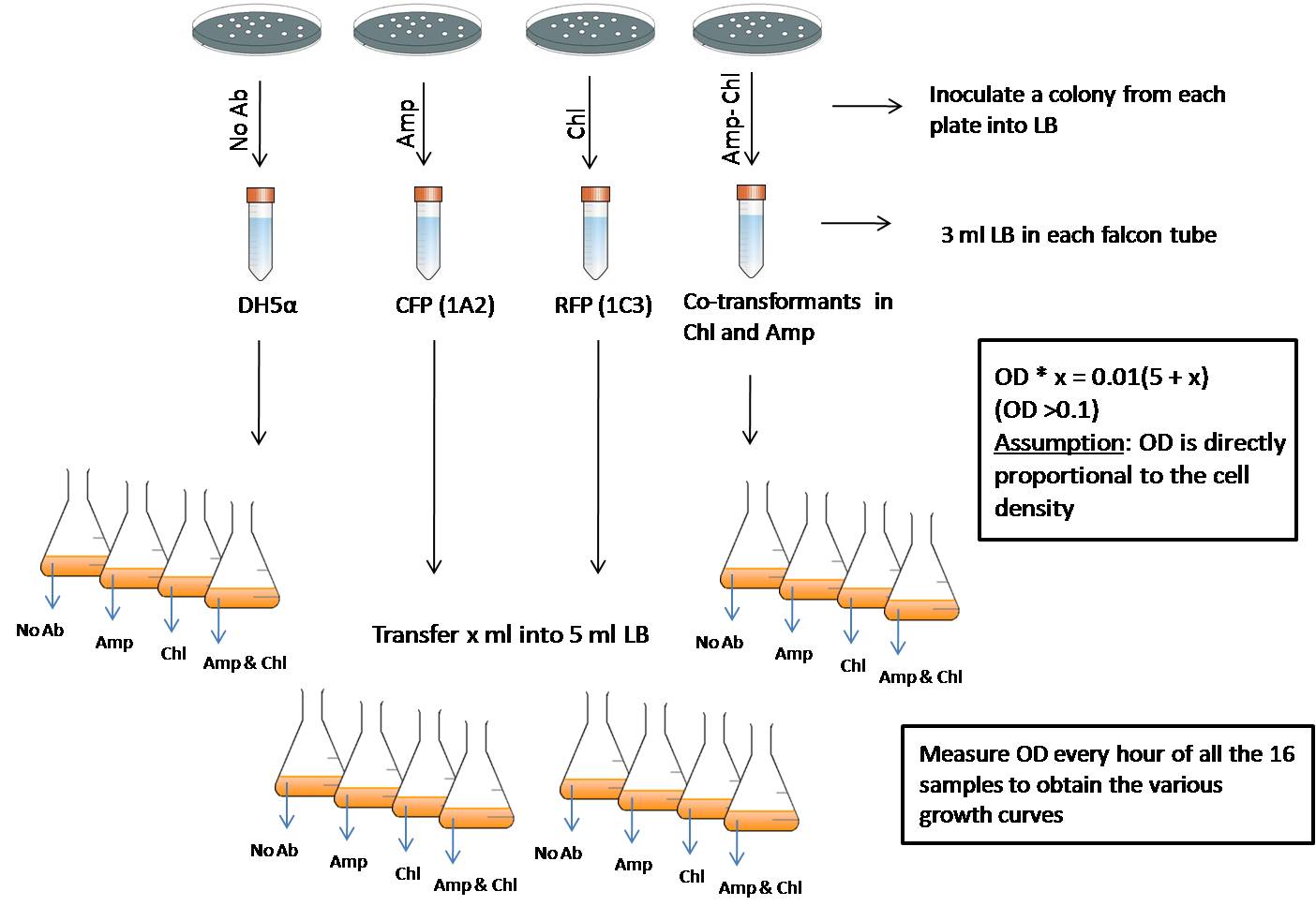
Fig 7: This experiment helps in comparing the growth rates of various strains in different antibiotic media.
We inoculate a colony from the plate containing the required strain into its corresponding media (the media with the required antibiotic). That is, DH5a will be inoculated into LB without any antibiotic, RFP (in pSB1C3)containing cells will be inoculated into LB containing Chloramphenicol (Chl), CFP (in pSB1A2)containing cells will be inoculated into LB containing Ampicillin (Amp) and RFP-CFP co-transformed cells into LB containing both the antibiotics. These are then grown for about 4 hours or till they reach an OD600 value between 0.1 to 0.5.
Then the culture of each strain is taken and inoculated into each of the 5ml broths which contains no antibiotic, Amp, Chl and Amp-Chl so that the OD value in all the freshly inoculated tubes is 0.01. Now we have an array of 16 tubes with various possible combinations:
- DH5a in no antibiotic, DH5a in Amp, DH5a in Chl and Dh5a in Amp-Chl
- RFP (pSB1C3) in no antibiotic, RFP (pSB1C3)in Amp, RFP (pSB1C3)in Chl and RFP (pSB1C3)in Amp-Chl
- CFP (pSB1A2) in no antibiotic, CFP (pSB1A2)in Amp, CFP (pSB1A2)in Chl and CFP (pSB1A2)in Amp-Chl
- RFP (1C3)-CFP (1A2) in no antibiotic, RFP (1C3)-CFP (1A2) in Amp, RFP (1C3)-CFP (1A2) in Chl and RFP (1C3)-CFP (1A2)in Amp-Chl
Thus the starting point for all the samples is the same - an OD600 of 0.01. Every hour starting from the point of inoculation, the OD of all the 16 samples is measured. This would give a fair idea of the growth rates of various strains in different antibiotic media.
Experimental protocol:
1. A colony of DH5a is inoculated into 3ml LB without any antibiotic in a 50ml centrifuge tube. Similarly, RFP (1C3) colony is inoculated into 3ml LB with Chl, CFP (1A2) colony into 3ml LB with Amp and RFP-CFP colony into 3ml LB with Amp-Chl.
2. This inoculum is allowed to grow for about 4 hours or till the OD600 of each sample crosses 0.1.
3. Then we use OD 1 x Vol 1 = OD 2 x Vol 2 to measure how much to inoculte from this 4 hour culture to each of the fresh 5ml LB medium with different antibiotic combinations so that the starting OD is 0.01. In this case, OD 1 is the OD600 of the 4 hour culture, Vol 1 is the volume of this 4 hour culture that needs to be inoculted in to the fresh 5ml culture, OD 2 is 0.01 (starting OD for all the 5 ml cultures) and the Vol 2 is the final volume (5ml + vol 1).
4. From this freshly inoculated sample, 150ul of the culture is used to measure the OD every hour starting from the point if inoculation. The 150ul of the sample is diluted 5 times to 750ul and then the OD600 is measured.
5. The whole procedure is repeated to check for reproducibility.
Note: After the initial 4 hour incubation (the 3ml cultures), we used this to inoculate a 5ml culture with the same antibiotic in the medium as in the 3ml culture. The OD600 of this 5ml tube after the inoculation was 0.01. This was then made to grow for 2.5 hours. This is the culture from which we inoculated the final 16 tubes which would then be used for measurements.
Click here for the results
Modeling
Basic Equations:

Where,
X+ is the cells with the plasmid.
X- is the cells without the plasmid.
u+ is the specific cell growth rate of cells with the plasmid.
u- is the specific cell growth rate of cells without the plasmid.
p is the probability of a cell to lose a plasmid.
We are trying to come up with a mathematical model for the relative populations of plasmid containing and plasmid free cells with respect to time using the growth curves that have been generated and the results from the fluorescence.
Fluorescence Imaging of cells to check for directed plasmid loss
This experiment is designed to study the plasmid loss when cells (transformed with a plasmid) are grown in media without the required selection pressures. Here, we perform 4 different experiments to study how
- Cells transformed with RFP (pSB1C3) lose the plasmid in the absence of Chloramphenicol in the medium
- Cells transformed with CFP (pSB1A2) lose the plasmid in the absence of Ampicillin in the medium.
- Cell cotransformed with RFP (pSB1C3) and CFP (pSB1A2) lose the RFP (pSB1C3) plasmid when grown in absence of Chloramphenicol and in the presence of Ampicillin in the medium.
- Cell cotransformed with RFP (pSB1C3) and CFP (pSB1A2) lose the CFP (pSB1A2) plasmid when grown in absence of Ampicillin and in the presence of Chloramphenicol in the medium.
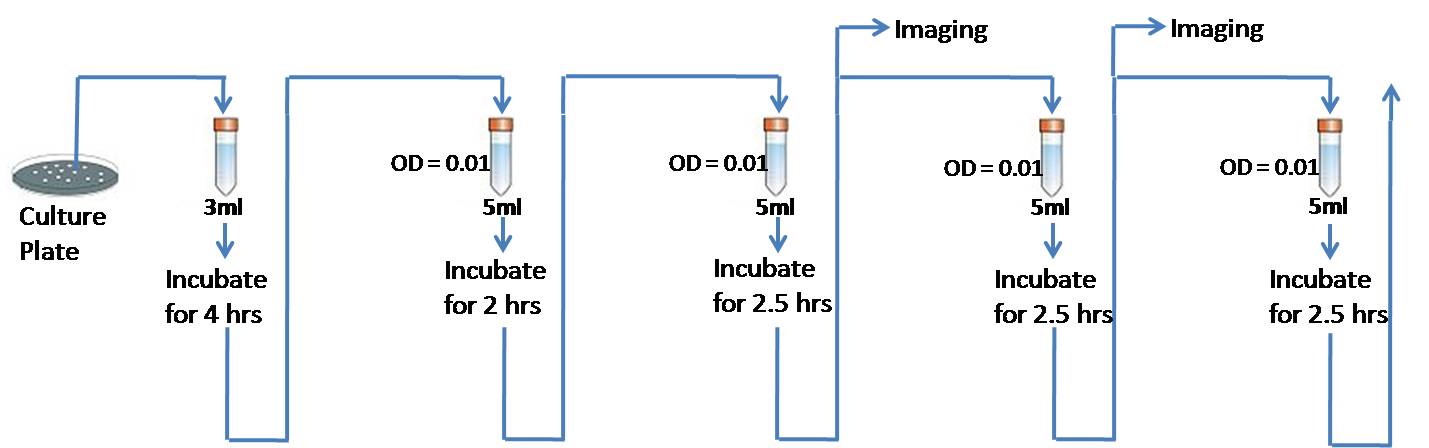
Fig 6.3: Experimental procedure for measuring the rate of plasmid loss by fluorescent imaging
We pick up colonies of each strain and inoculate it first in a medium with the appropriate antibiotic. Then use this to inoculate in specific growth media as show in the figure 6.3.
- RFP (pSB1C3) containing cells will be grown in medium containing no antibiotic and in medium containing Chloramphenicol
- CFP (pSB1A2) containing cells will be grown in medium containing no antibiotic and in medium containing Ampicillin.
- Co-transformed cell will be grown in medium containing no antibiotic, medium containing only Ampicillin, medium containing only Chloramphenicol, medium containing both Ampicillin and Chloramphenicol.
The expected behavior of the system is (listed in the same order as the above 3 conditions):
- RFP (pSB1C3)containing Cells grown in medium with no Chloramphenicol would eventually lose the plasmid and hence show no Red fluorescence when imaged under the microscope and those which are grown in the chloramphenicol containing medium would all be fluorescing red.
- CFP (pSB1A2)containing cells grown in medium with no Ampicillin would eventually lose the plasmid and hence show no Cyan fluorescence and those which are grown in the Ampicillin containing medium would all be fluorescing Cyan.
- Cotransformed cells grown in medium with no antibiotic would eventually lose both the plasmids and hence show no red or Cyan fluorescence, those which are grown in the Ampicillin containing medium would all be fluorescing Cyan, those which are grown in the Chloramphenicol containing medium would all be fluorescing Red and those which are grown in medium containing both the Antibiotics would all be fluorescing both Cyan and Red.
Protocol:
1. Inoculate 3ml LB containing Ampicillin with a colony of cells transformed with CFP (pSB1A2)- broth 1, inoculate 3ml LB containing Chloramphenicol with a colony of cells transformed with RFP (pSB1C3) - broth 2, inoculate 3ml LB containing Ampicillin and Chloramphenicol with a colony of cells co-transformed with CFP (pSB1A2)and RFP (pSB1C3) - broth 3.
2. Culture is grown for 4 hours or till the OD600 crosses 0.1.
3. Then we use OD 1 x Vol 1 = OD 2 x Vol 2 to measure how much to inoculate from this 4 hour culture to each of the fresh 5ml LB medium with different antibiotic combinations so that the starting OD is 0.01. In this case, OD 1 is the OD600 of the 4 hour culture, Vol 1 is the volume of this 4 hour culture that needs to be inoculated in to the fresh 5ml culture, OD 2 is 0.01 (starting OD for all the 5 ml cultures) and the Vol 2 is the final volume (5ml + vol 1).
Required inoculations are:
a. Broth 1 into tube with no antibiotic in the LB - tube 1
b. Broth 1 into tube with Ampicillin in the LB - tube 2
c. Broth 2 into tube with no antibioti in the LB - tube 3
d. Broth 2 into tube with Chloramphenicol in the LB - tube 4
e. Broth 3 into tube no antibiotic in the LB - tube 5
f. Broth 3 into tube with Ampicillin in the LB - tube 6
g. Broth 3 into tube with Chloramphenicol in the LB - tube 7
h. Broth 3 into tube with both the antibiotics in the LB - tube 8
4. This culture is allowed to grow for 2.5 hours.
5. Measure the OD at this stage and also prepare a slide for fluorescence imaging from each of the 8 tubes.
6. Then we use OD 1 x Vol 1 = OD 2 x Vol 2 to measure how much to inoculate from this 2.5 hour culture to each of the fresh 5ml LB medium with the same antibiotic combinations so that the starting OD is 0.01. In this case, OD 1 is the OD600 of the 2.5 hour culture, Vol 1 is the volume of this 2.5 hour culture that needs to be inoculated in to the fresh 5ml culture, OD 2 is 0.01 (starting OD for all the 5 ml cultures) and the Vol 2 is the final volume (5ml + vol 1). i.e. The culture from spent tube 1 is used to inoculte a fresh tube 1. Similarly follow the same for all the other 7 tubes.
7. Let this new culture grow for 2.5 hours and repeat from step 5 for about 5 times.
Click here to see the results of the fluorescence imaging.
 "
"






