Team:UCSF/NavigationPart2
From 2009.igem.org
| (One intermediate revision not shown) | |||
| Line 23: | Line 23: | ||
<p> </p> | <p> </p> | ||
<h3>Results</h3> | <h3>Results</h3> | ||
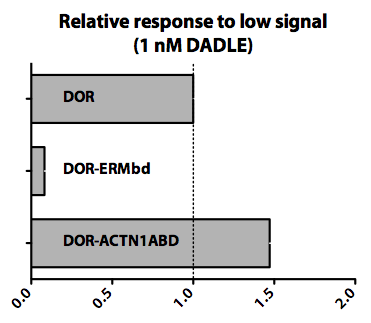
| - | <p>We found that virtually any protein domain/module known to alter the recycling of delta Opioid receptor (DOR) affected cellular migration to a low concentration of ligand (1 nM DADLE). Below, we show two examples of such domains. The actin binding domain from alpha-Actinin-1 (ACTN1ABD) appears to potentiate cellular migration when compared to the wild-type receptor. On the other hand, cellular response is inhibited at this concentration when DOR is | + | <p>We found that virtually any protein domain/module known to alter the recycling of delta Opioid receptor (DOR) affected cellular migration to a low concentration of ligand (1 nM DADLE). Below, we show two examples of such domains. The actin binding domain from alpha-Actinin-1 (ACTN1ABD), when fused to DOR (<strong><a href="http://partsregistry.org/Part:BBa_K209486">BBa_K209486: DOR-Actinin1ABD</a></strong>) appears to potentiate cellular migration when compared to the wild-type receptor. On the other hand, cellular response is inhibited at this concentration when DOR is fused to a domain of EBP50 (ERMbd) that binds to ERM (ezrin/radixin/moesin) family of proteins. </p> |
<p> </p> | <p> </p> | ||
</body> | </body> | ||
| Line 54: | Line 54: | ||
</html> | </html> | ||
| - | =Methods= | + | ==Methods== |
'''Navigation:''' | '''Navigation:''' | ||
| Line 62: | Line 62: | ||
*[[Media:ezt.pdf|HL-60: Time-lapse Microscopy Protocol]] - here is how we film HL-60 cells | *[[Media:ezt.pdf|HL-60: Time-lapse Microscopy Protocol]] - here is how we film HL-60 cells | ||
| - | =Selected Reading= | + | |
| + | ==Selected Reading== | ||
'''Navigation (New Sensors):''' | '''Navigation (New Sensors):''' | ||
Latest revision as of 19:59, 21 October 2009
Engineering NAVIGATION: Tuning Engineered Cells Sensitivity towards New Signals
Motivation: Why is this useful?
In our experiments, we could control exactly how much ligand was presented to our cells. In "real-life," however, we would want our cellular robots to be able to respond to a variety of signal strengths: from very low to very high. To accomplish this, we would want to be able to control the sensitivity of our receptors, or how the receptor's output changes when the measured quantity of ligand changes.
Approach
We felt that one key determinant of sensitivity would be the number of receptors present at the plasma membrane of the cell. Therefore, we measured the migration response of a receptor (delta Opioid receptor) whose recycling behavior could be engineered by fusing different recycling interaction modules to the C terminus of the GPCR. We tested a number of such receptor-module fusions for migration response and compared them to receptor alone. The primary assay here was again the Boyden chamber (transwell) assay.
Results
We found that virtually any protein domain/module known to alter the recycling of delta Opioid receptor (DOR) affected cellular migration to a low concentration of ligand (1 nM DADLE). Below, we show two examples of such domains. The actin binding domain from alpha-Actinin-1 (ACTN1ABD), when fused to DOR (BBa_K209486: DOR-Actinin1ABD) appears to potentiate cellular migration when compared to the wild-type receptor. On the other hand, cellular response is inhibited at this concentration when DOR is fused to a domain of EBP50 (ERMbd) that binds to ERM (ezrin/radixin/moesin) family of proteins.
Summary and Outlook
We have shown that we can tune the sensitivity of a receptor both up and down by fusing it to different recycling modules. Next, we would like to measure recycling directly, and determine whether changes in recycling are necessary for this difference in chemotactic response. All experiments in HL-60 cells would benefit the generation of stable cell lines, which would allow us to more precisely quantify these new behaviors.
Methods
Navigation:
- HL-60: Transfection Protocol - here is how we get our constructs into HL-60 cells
- HL-60: Boyden Chamber (Transwell) Protocol - here is how we assay chemotaxis in HL-60 cells
- HL-60: Time-lapse Microscopy Protocol - here is how we film HL-60 cells
Selected Reading
Navigation (New Sensors):
Receptors induce chemotaxis by releasing the betagamma subunit of Gi, not by activating Gq or Gs. Neptune ER, Bourne HR. Proc Natl Acad Sci U S A. 1997 Dec 23;94(26):14489-94.
Delineation of muscarinic receptor domains conferring selectivity of coupling to guanine nucleotide-binding proteins and second messengers. Wess J, Bonner TI, Dörje F, Brann MR. Mol Pharmacol. 1990 Oct;38(4):517-23.
Navigation (Sensor Sensitivity):
Actin-binding protein alpha-actinin-1 interacts with the metabotropic glutamate receptor type 5b and modulates the cell surface expression and function of the receptor. Cabello N, Remelli R, Canela L, Soriguera A, Mallol J, Canela EI, Robbins MJ, Lluis C, Franco R, McIlhinney RA, Ciruela F. J Biol Chem. 2007 Apr 20;282(16):12143-53. Epub 2007 Feb 20.
Engineered protein connectivity to actin mimics PDZ-dependent recycling of G protein-coupled receptors but not its regulation by Hrs. Lauffer BE, Chen S, Melero C, Kortemme T, von Zastrow M, Vargas GA. J Biol Chem. 2009 Jan 23;284(4):2448-58. Epub 2008 Nov 10.
| Home | The Team | The Project | Parts Submitted to the Registry | Our summer experience | Notebook | Human Practices |
|---|
 "
"