Team:Duke
From 2009.igem.org
| (117 intermediate revisions not shown) | |||
| Line 200: | Line 200: | ||
<div id="home-box"> <p> | <div id="home-box"> <p> | ||
| - | <center><font size="+ | + | <center><font size="+1"><font color="blue"><b> One-Step Construction of a Bioplastic Production |
| + | Pathway in E. <i>coli</i></b></font></font></center><br> | ||
<br> | <br> | ||
| - | <b>We, the determined and motivated <font size="+ | + | <b>We, the determined and motivated <font size="+1"><font color="blue">Duke University iGEM 2009 team</font></font> of 6 undergraduate students and 3 graduate students, have developed a practical and powerful method to deliver on the promise of sustainable <font size="+1"><font color="green">green</font></font> synthetic biology!</b><br> |
<br> | <br> | ||
| - | + | Due to the high costs and inefficiency of the gene and pathway cloning process, we invented a new procedure which <font size="+1"><font color="blue">lowers costs</font></font> and <font size="+1"><font color="blue">increases efficiency</font></font>. This method, Circular Polymerase Extension Cloning (CPEC), <font size="+1"><font color="blue">saves time</font></font> as well, since it does not involve ligation or restriction enzymes. See publication of our method <a href="http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0006441">here.</a></font> <br> | |
<br> | <br> | ||
| - | We have applied our method to the production of <font size="+ | + | We have applied our method to the production of <font size="+1"><font color="blue">biologically derived plastics</font></font>. The high costs of the current method of producing biodegradable plastics in an environmentally sound manner has hindered its widespread use. However, we have discovered a <font size="+1"><font color="blue">more efficient pathway</font></font> to produce these biodegradable plastics. </p> |
<br><br> | <br><br> | ||
| - | <center> <object width="580" height="360"><param name="movie" value="http://www.youtube.com/v/TZ_P8SIsWCQ&hl=en&fs=1&border=1"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess" value="always"></param><embed src="http://www.youtube.com/v/TZ_P8SIsWCQ&hl=en&fs=1&border=1" type="application/x-shockwave-flash" allowscriptaccess="always" allowfullscreen="true" width="580" height="360"></embed></object></center> | + | <center> <object width="580" height="360"><param name="movie" value="http://www.youtube.com/v/TZ_P8SIsWCQ&hl=en&fs=1&border=1"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess" value="always"></param><embed src="http://www.youtube.com/v/TZ_P8SIsWCQ&hl=en&fs=1&border=1" type="application/x-shockwave-flash" allowscriptaccess="always" allowfullscreen="true" width="580" height="360"></embed></object></center><br> |
| + | |||
| + | <div style="border:1.5px dashed black"> | ||
| + | <h2>Why do we deserve <b>GOLD</b>?</h2> | ||
| + | <ul> | ||
| + | <li> We had a <b>Great</b> summer working on our project</li> | ||
| + | <li> We submitted <b>2</b> biobricks: <a href="http://partsregistry.org/wiki/index.php?title=Part:BBa_K282001">BBa_K282001</a> and <a href="http://partsregistry.org/Part:BBa_K282000">BBa_K282000</a></li> | ||
| + | <li> We Created and implemented with great success a more accurate, flexible and efficient method of splicing multi-component biobricks systems into plasmid vectors. </li> | ||
| + | <li> and... </li> | ||
| + | </ul> | ||
| + | <br> | ||
| + | <center> <img src="https://static.igem.org/mediawiki/2009/a/a3/V_Duke.JPG" width="150" height="150"/> *We were also proud to help Team Valencia with their survey.</center> | ||
| + | </div> | ||
</div> | </div> | ||
| Line 214: | Line 227: | ||
<!-- Project --> | <!-- Project --> | ||
| - | <div id="project-box"> <h1> | + | <div id="project-box"> |
| - | Circular Polymerase Extension Cloning (CPEC) is | + | <h1>Abstract</h1> |
| + | A convenient ligation-free, sequence-independent one-step plasmid assembly and cloning method is developed [Quan J, Tian J (2009) Circular Polymerase Extension Cloning of Complex Gene Libraries and Pathways. PLoS ONE 4(7): e6441]. This strategy, Circular Polymerase Assembly Cloning (CPEC), relies solely on polymerase extension to assemble and clone multiple fragments into any vector. Using this method, we are able to quickly assemble a metabolic pathway consisting of multiple enzymes and regulatory elements for the production of a biocompatible as well as biodegradable plastic polymer in E. <i>coli</i>. | ||
| + | |||
| + | <html> | ||
| + | <h1> What is CPEC?</h1> | ||
| + | |||
| + | Circular Polymerase Extension Cloning (CPEC) is a much simplified | ||
sequence-independent cloning technology based entirely on the polymerase extension | sequence-independent cloning technology based entirely on the polymerase extension | ||
mechanism. This method extends overlapping regions between the insert and vector | mechanism. This method extends overlapping regions between the insert and vector | ||
| - | fragments to form a complete circular plasmid | + | fragments to form a complete circular plasmid (Figure 1a). The basic principle of this strategy is that, after denaturation, both single-stranded vector and inserts overlap with each other by end sequences and extend by using the other as the primer in a typical PCR reaction. After complete circular plasmids with one nick in each strand are formed, the relaxed plasmids are transformed to competent cells and sealed as closed plasmids. CPEC strategy is not only suitable for the cloning of an individual gene but also for gene libraries, combinatorial libraries and multi-fragment plasmid assembling. With its simplicity, cost-effectiveness and high transformation efficiency, CPEC has become the most convenient, economical and accurate cloning method at the present day, especially for cloning applications with high complexity. |
| - | + | </html> | |
| - | overlap with the | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | {| | + | {| style="background:transparent; margin:auto" |
| - | + | ||
| - | + | ||
| - | + | ||
|- | |- | ||
|| [[Image:CPEC.jpg|thumb|350px|Figure 1a. Biobrick spliced into vector using CPEC]] | || [[Image:CPEC.jpg|thumb|350px|Figure 1a. Biobrick spliced into vector using CPEC]] | ||
|| [[Image:multicomponent.jpg|thumb|350px|Figure 1b. Multicomponent Biobrick system spliced into vector simulatenously using CPEC]] | || [[Image:multicomponent.jpg|thumb|350px|Figure 1b. Multicomponent Biobrick system spliced into vector simulatenously using CPEC]] | ||
| - | |||
|} | |} | ||
| - | |||
<html> | <html> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
<br> | <br> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | < | + | <h1>One-Step Construction of a Bioplastic Production Pathway using CPEC</h1> |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | </ | + | |
| - | [[Image: | + | We applied CPEC in the construction of a multi-component |
| + | pathways in a single reaction. The proposed multi-way cloning mechanism is shown in Figure 1b. First, we constructed the biobrick parts containing genes involved in this pathway. Then we used multi-way CPEC for construction of | ||
| + | a metabolic pathway for synthesizing a biodegradable plastic, poly(3HB-co-4HB) in E. <i>coli.</i> | ||
| + | |||
| + | <h3> Biodegradable Plastic Synthesis Pathway in E. <i>coli.</i></h3> | ||
| + | </html>[[Image:Fig.2a.jpg|framed|Figure 2. PHA granule production in bacteria]] <html> | ||
| + | <p>Recently, polyhydroxyalkanoates (PHA) have received a lot of attention for their | ||
| + | potential use as bioplastic. They are naturally occurring biopolymers – under low nitrogen, | ||
| + | phosphorus, or oxygen conditions, many bacteria produce protein- and lipid-coated granules of | ||
| + | PHA for energy storage, and these granules can then in turn be harvested in large quantities as | ||
| + | bioplastics (Fig. 2). Bioplastics not only help reduce the reliance on petroleum-based plastics, but | ||
| + | also have a wide range of applications in the agricultural and medical fields because they are | ||
| + | biodegradable and biocompatible. The most common PHA is poly(3-hydroxybutyrate), or poly(3HB), which is produced by | ||
| + | the bacteria <i>Cupriavidus necator</i>. Three genes – phaA, phaB, and phaC – make up the | ||
| + | phaCAB operon that synthesizes the enzymes required for formation of poly(3HB), which is not | ||
| + | elastic enough for general plastic applications. But when phaCAB is expressed with cat2, a | ||
| + | succinate degradation gene from Clostridium kluyveri that produces the 4HB monomer, the | ||
| + | copolymer poly(3-hyroxybutyrate-co-4-hydroxybutyrate), or poly(3HB-co-4HB), is produced (Figure 3). Poly(3HB-co-4HB) is more elastic and thus has potential to be used in a variety of applications. | ||
| + | </html> | ||
<html> | <html> | ||
| + | However, bioplastic production is currently too costly to be used widely. For example, | ||
| + | poly(3HB-co-4HB) pathways that have been developed so far produce copolymers with very low | ||
| + | concentrations of 4HB, the monomer that gives the copolymer its elastic properties. This could | ||
| + | be because the enzyme that catalyzes PHA formation, PHA synthase, has higher affinity for the | ||
| + | 3HB-CoA substrate (100%) than the 4HB-CoA substrate (0.03%). </html> | ||
| + | [[Image:Bioplastic_pathway.jpg|framed|Figure 3. Pathway for poly(3HB-co-4HB) synthesis [Madison and Huisman, 1999]]] | ||
| + | <html>In addition, the 3HB | ||
| + | pathway uses acetyl-CoA, an input to the citric acid cycle, as a precursor, but cat2 uses succinyl- | ||
| + | CoA, an intermediate in the cycle. Since more processing is required to obtain the 4HB cursor, the pathway may naturally favor production of the 3HB polymer, resulting in higher 3HB composition in the final copolymer. | ||
| + | |||
<br><br> | <br><br> | ||
| - | Thus, this project aims to develop a more efficient | + | Thus, this project aims to develop a more efficient biopathway for poly(3HB-co-4HB) while increasing the 4HB monomer composition predictably. To achieve this goal, we designed vectors with various combinations of the phaCAB operon and the cat2 gene so that genetic manipulations can be made to the genes for PHA synthase and precursor processing to |
| - | biopathway for poly(3HB-co-4HB) while increasing the 4HB monomer composition predictably. | + | increase bioplastic production and quality. |
| - | + | </html> | |
| - | + | [[Image:Fig.4.jpg|framed|Figure 4. The reddish tint present on some colonies indicates the presence of PHA granules in the bacteria]] | |
| - | and cat2 | + | |
| - | + | <html> | |
| - | + | <h3>3HB genetic pathway</h3> | |
| - | + | The phaCAB operon genomic DNA from <i>Cupriavidus necator</i> was cloned into PCR Blunt II-TOPO vector as the backbone and the cloning product was transformed into bacteria cells which were then plated on kanamycin (50 μg/ml) plates. Successful insertion of phaCAB was shown by Nile Red fluorescence in colonies (Figure 4). | |
| + | |||
| + | <h3>4HB genetic pathway</h3> | ||
| + | The cat2 gene was inserted into the pSOS vector to obtain the | ||
| + | pSOS-cat2 construct. To obtain 4HB production, though, the phaC gene must also be expressed. | ||
| + | The 4HB precursor γ-butyrolactone (GBL) is required for 4HB production in this pathway, as the | ||
| + | 4HB precursor does not naturally exist in E. <i>coli.</i></html> | ||
| + | |||
| + | |||
| + | |||
| + | <html><h3>pASK vector constructs</h3> | ||
| + | The PCR-Blunt II-phaCAB and pSOS-cat2 plasmids described above were then used as | ||
| + | templates to create seven different vector constructs in pASK (Tables 1 and 2). The first | ||
| + | six vector constructs were created by PCR amplification of the insert, restriction digestion of the | ||
| + | vector and insert, ligation of the fragments, and finally transformation into E. <i>coli.</i> The seventh vector (pASKphaAB-term-pLZCat2phaC) was created using CPEC cloning and transformation into E. <i>coli</i> (Figure 5). All the constructs were sequenced to ensure that the constructs were correct. Aslo genotyping was done for the final construct 7 (Figure 6). </html> | ||
| + | |||
| + | {| class="wikitable" border="0" align="center" | ||
| + | |- | ||
| + | |[[Image:Bioplasticpathway_plasmid.jpg|thumb|380px|Figure 5. Construct 7.]] | ||
| + | |[[Image:Multi_component_gel.jpg|thumb|380px|right|Figure 6. (Upper left) Gel electrophoresis analysis of the final | ||
| + | assembly product after a 20-cycle CPEC. 5 ml of the reaction was | ||
| + | separated on a 0.8% agarose gel and visualized after ethidium bromide | ||
| + | staining. The full-length plasmid was 8360 bp. (Upper right) Gel electrophoresis | ||
| + | analysis of the multi-way CPEC reaction. 5 ml was taken out of the | ||
| + | reaction after 2, 5, and 10 cycles and separated on a 0.8% agarose gel | ||
| + | (lanes 1, 2 and 3). The starting lengths of the four fragments were 3280, | ||
| + | 2959, 2040, and 171 bp, respectively. The 171-bp band was not visible. | ||
| + | (Bottom) Restriction mapping of the isolated plasmids derived from the CPEC | ||
| + | reaction. Plasmid DNA from five independent colonies (I-V) were | ||
| + | digested with BamHI (lane 2, 8.4 kb), BamHI-XhoI (lane 3, 6.6 kb and | ||
| + | 1.8 kb), and NdeI (lane 4, 5.4 kb and 3 kb). Purified plasmids not | ||
| + | subjected to restriction digestion are shown in lane 1. The molecular | ||
| + | weight marker used in this figure was NEB 1 kb DNA ladder.]] | ||
| + | |} | ||
| + | |||
| + | <html> | ||
| + | <h3>Screening Experiments</h3> | ||
| + | Two different experiments were conducted to investigate (1) the optimal | ||
| + | anhydrotetracylcine level for inducing expression of bioplastic genes and (2) Nile Red as a | ||
| + | screening technique for polymer production. To determine the optimal anydrotetracycline levels | ||
| + | for phaCAB and cat2 gene expression, agar plates were prepared with 0, 5, 50, 150, and 200 | ||
| + | ng/ml of anyhdrotetracyline, and Construct 7 E. coli were grown on them overnight at 37°C. The | ||
| + | number of colonies on each plate was then compared the following day. To test Nile Red as a | ||
| + | screening technique to detect PHA production, agar plates were prepared with various levels of | ||
| + | Nile Red (0, 0.2, 0.5,1.0 μg/ml). Bacteria that were known to produce PHA (positive control) and | ||
| + | negative control bacteria were then plated and grown overnight at 37°C. After sitting on the | ||
| + | benchtop for a few days, they were examined under equal levels of UV lighting. | ||
| + | </html> | ||
| + | [[Image:Fig.5.jpg|framed|Figure.7. Precipitated product from pASK-phaC-no tag (2 tubes on left) and negative control | ||
| + | pASK (2 tubes on right) harvesting.]] | ||
| + | |||
| + | <html> | ||
| + | |||
| + | |||
| + | <h3>Polymer harvesting</h3> | ||
| + | To obtain in vivo PHA polymer, E. coli containing the desired construct were grown in 3 | ||
| + | mL of LB broth overnight, of which 1 mL was then cultured in 50 mL of LB broth and grown at | ||
| + | 30oC until the cells reached an optical density of .7 A (600 nm). Glucose was added to a final | ||
| + | concentration of 1%, and the cells were harvested after 48 hour of incubation at 37°C. | ||
| + | To harvest, the cells were centrifuged at 3500 rpm and suspended in saline (0.0067 M | ||
| + | phosphate buffer) solution. The cells were re-centrifuged to obtain a washed pellet, freezethawed | ||
| + | in dry ice, and then freeze-dried by lyophilization. The pellet was shaken overnight at 37°C in chloroform, after which bacterial cell waste was filtered out using Whatman paper and a vacuum flask. The polymer was then precipitated from the filtered solution by adding 10 volumes of ethanol, centrifuged, and then re-dissolved in chloroform. Finally, 10 volumes of methanol was used to re-precipitate the polymer before centrifuging to obtain a purified polymer pellet. A promising sign was that there was | ||
| + | much more white powder precipitate in the experimental samples than the negative control | ||
| + | samples after harvesting (Figure 7). | ||
| + | |||
| + | <h3><sup>1</sup>H NMR Spectroscopy</h3> | ||
| + | To characterize harvested polymers, 300 MHz 1H NMR spectroscopy was used. The | ||
| + | NMR sample was prepared by re-dissolving 20 mg of each polymer sample in 600 μl of | ||
| + | deuterochloroform in an NMR tube. Attempts were made to harvest and identify the 3HB, 4HB, and poly(3HB-co-4HB) | ||
| + | polymers. The first harvest attempt was made on pUC19phaCAB and then on pASK-phaCAB-no | ||
| + | tag (Construct 1) to try and obtain 3HB polymer; the NMR results for the harvested product were | ||
| + | compared to that of a negative control (no insert) and commercial 3HB. Several harvest attempts | ||
| + | were also made on pSOS-cat2phaC bacteria (Construct 4) to try to confirm 4HB polymer | ||
| + | production. Finally, bacteria transformed with pASKphaAB-termpLZCat2phaC (Construct 7) | ||
| + | were harvested in an attempt to identify poly(3HB-co-4HB) by NMR. | ||
| + | </html> | ||
| + | |||
| + | |||
| + | |||
| + | {| class="wikitable" border="0" align="center" | ||
| + | |- | ||
| + | |[[Image:Fig.6.jpg|thumb|240px|Figure 8a. 300 MHZ <sup>1</sup>H NMR for commercial 3HB]] | ||
| + | |[[Image:Fig.7.jpg|thumb|240px|Figure 8b. 300 MHZ <sup>1</sup>H NMR for experimental 3HB]] | ||
| + | |[[Image:Fig.8.jpg|thumb|240px|right|Figure 8c. 300 MHZ <sup>1</sup>H NMR for negative control (pASK vector)]] | ||
| + | |} | ||
| + | |||
| + | {| class="wikitable" border="0" align="center" | ||
| + | |- | ||
| + | |[[Image:Fig.9.jpg|thumb|380px|Figure 8d. Comparison of expansions of the methane proton region in the <sup>1</sup>H NMR spectra –commercial 3HB (left) and experimental (right).]] | ||
| + | |[[Image:Fig.10.jpg|thumb|420px|right|Figure 8e. Comparison of expansions of the CH2 regions in the <sup>1</sup>H NMR spectra – Commercial 3HB (left) and experimental (right)]] | ||
| + | |} | ||
| + | |||
| + | <html>The <sup>1</sup>H NMR spectra confirmed production of 3HB in the experimental bacteria and | ||
| + | no 3HB production in the negative control bacteria (Fig. 8a-e). Commercial 3HB (from Sigma- | ||
| + | Aldrich) was used as a comparison control (Fig. 8a). The 3HB peaks in the experimental sample | ||
| + | matched the commercial 3HB sample, as well as 3HB spectra found in the literatures. | ||
| + | However, the NMR spectra for the experimental and control samples had extremely large peaks | ||
| + | from residual methanol and ethanol left over from the harvesting procedures, a problem | ||
| + | corrected in later NMR readings by freeze-drying at the end of the harvesting procedure. | ||
| + | |||
| + | |||
| + | Future directions | ||
would be to test the hypothesis to see if phaC can be manipulated to increase | would be to test the hypothesis to see if phaC can be manipulated to increase | ||
4HB-to-3HB composition in poly(3HB-co-4HB) and to increase efficient production | 4HB-to-3HB composition in poly(3HB-co-4HB) and to increase efficient production | ||
| Line 287: | Line 399: | ||
to accumulate larger quantities of PHA granules before dividing. Ultimately, once | to accumulate larger quantities of PHA granules before dividing. Ultimately, once | ||
an optimal biopathway is found, the goal would be to explore a model for mass | an optimal biopathway is found, the goal would be to explore a model for mass | ||
| - | production of PHA bioplastics so that novel applications of bioplastics can be | + | production of PHA bioplastics so that novel applications of bioplastics can be </html>[[Image:Table 3.jpg|framed|Table 1. Vector designs with various phaCAB and cat2 combinations]] |
| + | [[Image:Table 2.jpg|framed|Table 2. Restriction enzymes and details of the vector constructs]] | ||
| + | <html> | ||
feasible economically.</p> | feasible economically.</p> | ||
| Line 294: | Line 408: | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
<html> | <html> | ||
| + | <h1> Biobricks </h1> | ||
| + | |||
| + | In order to facilitate the completion of this project, several biobricks were created and used. The two foremost (the ones submitted) were phaAB and a double terminator. | ||
| + | |||
| + | <br> | ||
| + | <h3> phaAB </h3> | ||
| + | <br> | ||
| + | Part: <b><a href="http://partsregistry.org/wiki/index.php?title=Part:BBa_K282000"> BBa_K282000 </a></b> | ||
| + | <br> | ||
| + | Used in the creation of bioplastics. Contains both the PhaA gene and the PhaB gene. Part of the pathway PhaAB - Terminator - Cat2PhaC . | ||
| + | <br> | ||
| + | |||
| + | <br> | ||
| + | <h3> Terminator </h3> | ||
| + | <br> | ||
| + | Part: <b><a href="http://partsregistry.org/wiki/index.php?title=Part:BBa_K282001"> BBa_K282001 </a></b> | ||
| + | <br> | ||
| + | |||
| + | Based off of Part:<b>BBa_B0014</b>. Comprised of forward terminator Part:<b>BBa_B0010</b> and reverse terminator Part:<b>BBa_B0012</b>. | ||
| + | |||
| + | <br> | ||
</div> | </div> | ||
| Line 307: | Line 437: | ||
<div id="notebook-box"></html> | <div id="notebook-box"></html> | ||
| - | + | =Calendar= | |
| - | + | <html><center><iframe src="http://www.google.com/calendar/embed?showPrint=0&mode=AGENDA&height=600&wkst=1&bgcolor=%23FFFFFF&src=aoqcbepvqdo8afcgih0d5mj1cc%40group.calendar.google.com&color=%23A32929&ctz=America%2FNew_York" style=" border-width:0 " width="900" height="600" frameborder="0" scrolling="no"></iframe></center></html> | |
| - | + | ||
| - | <html>< | + | |
| - | < | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
=Protocols= | =Protocols= | ||
| Line 333: | Line 445: | ||
[[#Protocols | Back to top]] | [[#Protocols | Back to top]] | ||
| - | + | '''Materials''' | |
| - | Materials | + | * Phusion™ High-Fidelity PCR Kit (FINNZYMES, Cat. No. F-553) |
| - | + | * Thermocycler | |
| - | + | ||
| - | + | '''Preparation''' | |
| - | 5x Phusion HF Buffer | + | <pre> |
| - | 10 mM dNTPs | + | 5x Phusion HF Buffer 4 ul |
| - | Vector | + | 10 mM dNTPs 0.4 ul |
| - | Insert | + | Vector 50 ng/1kb |
| - | Phusion DNA Polymerase 0.2 ul | + | Insert x ng* |
| - | H2O | + | Phusion DNA Polymerase 0.2 ul |
| + | ------------------------------- | ||
| + | H2O to 20 ul | ||
| + | </pre> | ||
| - | *The amount of insert is determined so that the molar ratio for vector and insert is 1 to 2. | + | <nowiki>*The amount of insert is determined so that the molar ratio for vector and insert is 1 to 2.</nowiki> |
| - | 98°C | + | '''Procedures''' |
| - | 10X | + | <pre> |
| - | 98°C | + | 98°C 30sec |
| - | Annealing** | + | 10X |
| - | 72°C | + | 98°C 10 sec |
| - | 72°C | + | Annealing** 30 sec |
| - | 4°C | + | 72°C x sec*** |
| - | + | 72°C 5min | |
| - | + | 4°C hold | |
| + | </pre> | ||
| - | + | <nowiki>** Anneal at Tm + 3°C. The Tm should be calculated with the nearest-neighbor method.</nowiki><br /> | |
| - | + | <nowiki>***The extension time is usually calculated according to the shortest piece with 15 sec /kb if the cloning is not complicated. For example, if there is only one insert and is shorter than the vector, say, 600 bp, then I will use 15 sec for extension. Refer to the published paper for detailed information.</nowiki> | |
==DNA Purification== | ==DNA Purification== | ||
| - | [[#Protocols | Back to top]]<br | + | [[#Protocols | Back to top]]<br /> |
| - | + | ||
| - | + | '''Materials :''' <html> <ul> | |
| - | Materials : <html> <ul> | + | |
<li> E.Z.N.A Gel Purification Kit (Omega Bio-Tek, Cat No. D2500-02 )</li> | <li> E.Z.N.A Gel Purification Kit (Omega Bio-Tek, Cat No. D2500-02 )</li> | ||
<li> Water bath equilibrated to 55-65C</li> | <li> Water bath equilibrated to 55-65C</li> | ||
| Line 375: | Line 488: | ||
<li>Isopropanol (for fragments < 500 bp only)</li></ul></html> | <li>Isopropanol (for fragments < 500 bp only)</li></ul></html> | ||
| - | Protocol: | + | '''Protocol:''' |
| - | + | # Perform agarose gel electrophoresis to fractionate DNA fragments. Any type or grade of agarose may be used. It is strongly recommended, however, that fresh TAE buffer or TBE buffer be used as running buffer. Do not re-use running buffer as its pH will increase and reduce yields. | |
| + | # When adequate separation of bands has occurred, carefully excise the DNA fragment of interest using a wide, clean scalpel. | ||
| + | # Determine the approximate volume of the gel slice by weighing it in a clean 1.5 ml microfuge tube. Assuming a density of 1 g/ml of gel, the volume of gel is derived as follows: A gel slice of mass 0.3 g will have a volume of 0.3 ml. Add equal volume of Binding Buffer (XP2). Incubate the mixture at 55C-60C for 7 min or until the gel has completely melted. Mix by shaking or inverting the tube every 2-3 minutes. Centrifuge the tube briefly to collect all the liquid to the bottom of the tube. | ||
| - | |||
| - | |||
| - | |||
Note: For DNA fragment less than 500bp, add 1 sample volume of isopropanol after the addition of Binding Buffer (XP2). | Note: For DNA fragment less than 500bp, add 1 sample volume of isopropanol after the addition of Binding Buffer (XP2). | ||
| - | + | # Apply up to 700 ul of the DNA/agarose solution to a HiBind® DNA spin column assembled in a clean 2 ml collection tube (provided) and centrifuge in a microcentrifuge at 8,000-10,000 x g for 1 min at room temperature. Discard the liquid. Re-use the collection tube in Steps 5-8. For volumes greater than 700 ul, load the column and centrifuge successively, 700 ul at a time. Each HiBind® spin-column has a total capacity of 25-30 ug DNA. | |
| + | # Discard liquid and add 300ul Binding Buffer. Centrifuge at 10,000 x g for 1 minutes. | ||
| + | # Add 700 ul of SPW Buffer diluted with absolute ethanol into the column and wait 2-3 minutes. Centrifuge at 10,000 x g for 1 min at room temperature to wash the sample. | ||
| + | # Discard liquid and repeat step 6 with another 700 ul SPW Buffer. | ||
| + | # Discard liquid and, re-using the collection tube, centrifuge the empty column for 1 min at maxi speed (>13,000 x g) to dry the column matrix. This drying step is critical for good DNA yields. | ||
| + | # Place column into a clean 1.5 ml microcentrifuge tube (not provided). Add 30-50 ul depending on desired concentration of final product) Elution Buffer (or sterile deionized water) directly to the center of the column matrix, then incubate for 1 minute. Centrifuge 1 min at maxi speed (>13,000 x g) to elute DNA. This represents approximately 70% of bound DNA. An optional second elution will yield any residual DNA, though at a lo | ||
| - | + | ==PCA (Polymerase Cycle Assembly)== | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | ==PCA (Polymerase Cycle Assembly== | + | |
[[#Protocols | Back to top]] <br /> | [[#Protocols | Back to top]] <br /> | ||
| - | Materials | + | '''Materials''' |
| - | + | * Phusion™ High-Fidelity PCR Kit (FINNZYMES, Cat. No. F-553) | |
| - | + | * Thermocycler | |
| - | + | '''Preparation''' | |
| - | 5x Phusion HF Buffer | + | {| |
| - | 10 mM dNTPs | + | |5x Phusion HF Buffer || 5 ul |
| - | Oligo mixture | + | |- |
| - | Phusion DNA Polymerase | + | |10 mM dNTPs || 0.5 ul |
| - | H2O | + | |- |
| + | |Oligo mixture || 125 ng /250 ng /500 ng / | ||
| + | |- | ||
| + | |Phusion DNA Polymerase || 0.25 ul | ||
| + | |- | ||
| + | | --------------------- || -------- | ||
| + | |- | ||
| + | |H2O || to 25 ul | ||
| + | |} | ||
| - | 98°C | + | '''Procedures''' |
| - | 40X | + | <pre> |
| - | 98°C | + | 98°C 30sec |
| - | 70-50°C | + | 40X |
| - | 50°C | + | 98°C 7 sec |
| - | 72°C | + | 70-50°C slow ramp, 0.1°C/sec |
| - | 72°C | + | 50°C 30 sec |
| - | 4°C | + | 72°C 15 sec /kb |
| + | 72°C 5 min | ||
| + | 4°C hold | ||
| + | </pre> | ||
| - | == | + | ==PCR Product Clean-up for DNA Sequencing== |
| - | [[#Protocols | Back to top]] | + | [[#Protocols | Back to top]] |
| - | Materials: | + | '''Materials:''' |
| - | + | * ExoSAP-IT® (usb, Cat. No. 78200) | |
| - | + | *Thermocycler | |
| - | + | ||
| - | + | '''Protocol:''' | |
| + | # Remove ExoSAP-IT from -20°C freezer and keep on ice throughout this procedure. | ||
| + | # Mix 5 μl of a post-PCR reaction product with 2 μl of ExoSAP-IT for a combined 7 μl reaction volume. | ||
| + | # Incubate at 37°C for 15 min to degrade remaining primers and nucleotides. | ||
| + | # Incubate at 80°C for 15 min to inactivate ExoSAP-IT. | ||
| + | # The PCR product is now ready for use in DNA sequencing etc. | ||
| - | + | ==PCR== | |
| + | [[#Protocols | Back to top]] | ||
| - | + | '''Materials''' | |
| + | * Phusion™ High-Fidelity PCR Kit (FINNZYMES, Cat. No. F-553) | ||
| + | * Thermocycler | ||
| - | + | '''Preperation''' | |
| + | {| | ||
| + | |- | ||
| + | |5x Phusion HF Buffer || 10 ul | ||
| + | |- | ||
| + | |10 mM dNTPs || 1 ul | ||
| + | |- | ||
| + | |DNA template || 1 pg – 10 ng | ||
| + | |- | ||
| + | |Forward primer (10 uM) || 2.5 ul | ||
| + | |- | ||
| + | |Reverse primer (10 uM) || 2.5 ul | ||
| + | |- | ||
| + | |Phusion DNA Polymerase || 0.5 ul | ||
| + | |- | ||
| + | | -------------------- || ----- | ||
| + | |- | ||
| + | |H2O || to 50 ul | ||
| + | |} | ||
| - | |||
| - | |||
| - | |||
| - | + | '''Procedure''' | |
| - | + | <pre> | |
| - | + | 98°C 30sec | |
| + | 30X | ||
| + | 98°C 10 sec | ||
| + | Annealing* 30 sec | ||
| + | 72°C 15 sec per 1 kb | ||
| + | 72°C 5min | ||
| + | 4°C hold | ||
| + | </pre> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | + | <nowiki>* Anneal at Tm + 3°C. The Tm should be calculated with the nearest-neighbor method.</nowiki> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
==Single Colony PCR== | ==Single Colony PCR== | ||
| - | [[#Protocols | Back to top]] | + | [[#Protocols | Back to top]] |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | '''Materials''' | |
| - | 10x Standard Taq Buffer | + | * Taq DNA Polymerase with Standard Taq Buffer (NEB, Cat. No. M0273) |
| - | 10 mM dNTPs | + | * 10 mM dNTP Mix (NEB, Cat. No. N0447) |
| - | Bacteria culture* | + | * Thermocycler |
| - | Forward primer (10 uM) | + | |
| - | Reverse primer (10 uM) | + | '''Preperation''' |
| - | Taq DNA Polymerase | + | {| |
| - | H2O | + | |- |
| + | |10x Standard Taq Buffer || 2 ul | ||
| + | |- | ||
| + | |10 mM dNTPs || 0.4 ul | ||
| + | |- | ||
| + | |Bacteria culture* || 1 ul | ||
| + | |- | ||
| + | |Forward primer (10 uM) || 1 ul | ||
| + | |- | ||
| + | |Reverse primer (10 uM) || 1 ul | ||
| + | |- | ||
| + | |Taq DNA Polymerase || 0.2 ul | ||
| + | |- | ||
| + | | ----------------- || ------- | ||
| + | |- | ||
| + | |H2O || to 20 ul | ||
| + | |} | ||
| + | <nowiki>*Bacteria culture refers to E. coli cultured in LB solution overnight.</nowiki> | ||
| - | * | + | '''Procedures''' |
| + | <pre> | ||
| + | 94°C 5 min | ||
| + | 25X | ||
| + | 94°C 15 sec | ||
| + | Annealing** 30 sec | ||
| + | 72°C 1 min per kb | ||
| + | 72°C 5min | ||
| + | 4°C hold | ||
| + | </pre> | ||
| - | + | <nowiki>**Anneal at Tm which is calculated with salt-adjusted method.</nowiki> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | **Anneal at Tm which is calculated with salt-adjusted method. | + | |
==Transformation== | ==Transformation== | ||
[[#Protocols | Back to top]] <br /> | [[#Protocols | Back to top]] <br /> | ||
| - | Materials:<html><ul> | + | '''Materials:'''<html><ul> |
<li>GC5 Chemical Competent Cells (Genesee Scientific, Cat. No. 42-653)</li> | <li>GC5 Chemical Competent Cells (Genesee Scientific, Cat. No. 42-653)</li> | ||
<li>SOC Medium (Sigma, Cat. No. S1797)</li> | <li>SOC Medium (Sigma, Cat. No. S1797)</li> | ||
| Line 503: | Line 649: | ||
<li>37°C shaker</li> | <li>37°C shaker</li> | ||
<li>water bath</li></ul></html> | <li>water bath</li></ul></html> | ||
| - | Protocol: | + | |
| + | '''Protocol:''' | ||
1. Thaw 1 tube of competent cells on ice; | 1. Thaw 1 tube of competent cells on ice; | ||
| Line 520: | Line 667: | ||
8. Place the plate up-side-down in 37°C incubator for 16-18 hours (overnight). | 8. Place the plate up-side-down in 37°C incubator for 16-18 hours (overnight). | ||
| + | |||
| + | ==NMR== | ||
| + | |||
| + | # Login and start NMR program. | ||
| + | # Click Acqi. Load sample. Make sure spin is on and lock is off. | ||
| + | # Go to lock. Decrease number of sine waves to 1 to obtain step function. Make sure Zo is 1100, lockpower is below 30, lockgain is 36, lockphase is 352, and spin is 20. | ||
| + | # Go to shim. Increase lock level. | ||
| + | # Click main menu. Click set up. Click H1CDCl3. Type ‘nt=64’ (number of scans), ‘ss=2’ (dummy scans), ‘go’, ‘lb=0.2’ (line broadening). | ||
| + | # Viewing: Type ‘wft’, ‘dscale’, ‘dfp’ to display peak frequency. | ||
| + | # Zooming in: Click display. Go to interactive. Type ‘cr=8p, delta=8p’ to set width of zoom. Use cursor to set boundaries. Click to cut. | ||
| + | # Printing: Type “pL pscale(0) pltext ppf page’ to print spectra with text and peak frequencies. | ||
| + | |||
| + | = Lab Safety = | ||
| + | |||
| + | None of our project ideas raised any safety issues whether it be researcher, environmental, or public safety. Duke has an in-house review board for projects and they did not raise any flags regarding our project. The biobrick parts created also do not pose any safety issues. | ||
| + | |||
<html> | <html> | ||
| Line 525: | Line 688: | ||
| - | <div id="team-box" | + | <!-- Team Profile --> |
| - | + | ||
| + | <div id="team-box"></html> | ||
| + | |||
| + | [[Image:IMG_2374.jpg|thumb|400px|Not Including: Andrew Ang and Kevin Chien]] | ||
| + | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
<html><h2>Students</h2></html> | <html><h2>Students</h2></html> | ||
{| | {| | ||
| Line 548: | Line 702: | ||
|[[Image:chienk.jpg|200px]] | |[[Image:chienk.jpg|200px]] | ||
|'''Kevin Chien''' <br /> kevin.chien(at)duke.edu <br /> | |'''Kevin Chien''' <br /> kevin.chien(at)duke.edu <br /> | ||
| + | Kevin Chien is a freshman at duke, majoring in Biomedical Engineering. He wants to explore his options in many different careers. He occasionally plays Club Frisbee and is interested in many different areas of knowledge, ranging from Astrophysics to Public Policy (and even sometimes History). He worked on this iGEM team last year as well and enjoyed his stay in Boston(hopes to have a good one this year too!). | ||
|- | |- | ||
|[[Image:Fuy.jpg|200px]] | |[[Image:Fuy.jpg|200px]] | ||
|'''Yaoyao Fu''' <br /> yf21(at)duke.edu <br /> | |'''Yaoyao Fu''' <br /> yf21(at)duke.edu <br /> | ||
| + | Yaoyao Fu is a master's student in Biomedical Engineering. She is interested in molecular synthetic biology. Her hobbies are traveling and sports. | ||
|- | |- | ||
|[[Image:Faith.jpg|200px]] | |[[Image:Faith.jpg|200px]] | ||
| Line 565: | Line 721: | ||
|[[Image:Nicholas.jpg|200px]] | |[[Image:Nicholas.jpg|200px]] | ||
|'''Nicholas Tang''' <br /> nicholas.tang(at)duke.edu <br /> | |'''Nicholas Tang''' <br /> nicholas.tang(at)duke.edu <br /> | ||
| + | Nicholas Tang is a junior Biomedical Engineering/Electrical and Computer Engineering student. He plans to pursue his interests in computational biology and biological engineering. His hobbies include cycling, playing the violin and playing the piano. As a participant in the 2006, 2007, and 2008 Duke iGEM teams, he is excited to contribute to the progress of the synthetic biology community, and hopes to help in any way he can. | ||
|- | |- | ||
|[[Image:zhup.jpg|200px]] | |[[Image:zhup.jpg|200px]] | ||
| Line 570: | Line 727: | ||
|- | |- | ||
|} | |} | ||
| + | <html><h2>Faculty Advisors</h2></html> | ||
| + | {| | ||
| + | |[[Image:TianJ.jpg|100px]] | ||
| + | |'''Dr. Jingdong Tian''' <br /> jtian(at)duke.edu <br />Department of Biomedical Engineering & IGSP | ||
| + | |[[Image:YouL.jpg|100px]] | ||
| + | |'''Dr. Lingchong You''' <br /> you(at)duke.edu <br /> Department of Biomedical Engineering & IGSP | ||
| + | |[[Image:Yuanf.jpg|100px]] | ||
| + | |'''Dr. Fan Yuan''' <br /> fyuan(at)duke.edu <br /> Department of Biomedical Engineering | ||
| + | |} | ||
| + | {| | ||
| + | |[[Image:Quanm.jpg|100px]] | ||
| + | |'''Maggie Jiayuan Quan''' <br /> jq7(at)duke.edu <br /> Graduate Student | ||
| + | |[[Image:Rezaf.jpg|150px]] | ||
| + | |'''Faisal Reza''' <br /> faisal.reza(at)duke.edu <br /> Graduate Student | ||
| + | |} | ||
| + | |||
<html> | <html> | ||
</div> | </div> | ||
| Line 578: | Line 751: | ||
---- | ---- | ||
| - | {| class="wikitable" border="0" | + | {| class="wikitable" border="0" style="margin:auto" |
|- | |- | ||
| - | |[[Image:LORDlogo.gif| | + | |[[Image:LORDlogo.gif|150px]] |
|[[Image:Bmeheader.gif|650px]] | |[[Image:Bmeheader.gif|650px]] | ||
|[[Image:igsp.png|100px]] | |[[Image:igsp.png|100px]] | ||
Latest revision as of 04:00, 22 October 2009
| Home | Project | Notebook | Team |
We, the determined and motivated Duke University iGEM 2009 team of 6 undergraduate students and 3 graduate students, have developed a practical and powerful method to deliver on the promise of sustainable green synthetic biology!
Due to the high costs and inefficiency of the gene and pathway cloning process, we invented a new procedure which lowers costs and increases efficiency. This method, Circular Polymerase Extension Cloning (CPEC), saves time as well, since it does not involve ligation or restriction enzymes. See publication of our method here.
We have applied our method to the production of biologically derived plastics. The high costs of the current method of producing biodegradable plastics in an environmentally sound manner has hindered its widespread use. However, we have discovered a more efficient pathway to produce these biodegradable plastics.
Why do we deserve GOLD?
- We had a Great summer working on our project
- We submitted 2 biobricks: BBa_K282001 and BBa_K282000
- We Created and implemented with great success a more accurate, flexible and efficient method of splicing multi-component biobricks systems into plasmid vectors.
- and...
 *We were also proud to help Team Valencia with their survey.
*We were also proud to help Team Valencia with their survey.Abstract
A convenient ligation-free, sequence-independent one-step plasmid assembly and cloning method is developed [Quan J, Tian J (2009) Circular Polymerase Extension Cloning of Complex Gene Libraries and Pathways. PLoS ONE 4(7): e6441]. This strategy, Circular Polymerase Assembly Cloning (CPEC), relies solely on polymerase extension to assemble and clone multiple fragments into any vector. Using this method, we are able to quickly assemble a metabolic pathway consisting of multiple enzymes and regulatory elements for the production of a biocompatible as well as biodegradable plastic polymer in E. coli.What is CPEC?
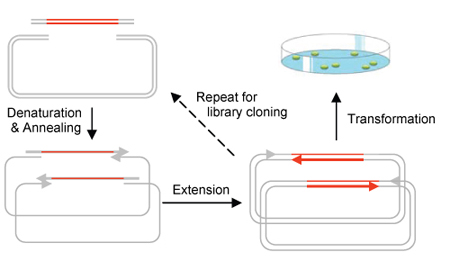
Circular Polymerase Extension Cloning (CPEC) is a much simplified sequence-independent cloning technology based entirely on the polymerase extension mechanism. This method extends overlapping regions between the insert and vector fragments to form a complete circular plasmid (Figure 1a). The basic principle of this strategy is that, after denaturation, both single-stranded vector and inserts overlap with each other by end sequences and extend by using the other as the primer in a typical PCR reaction. After complete circular plasmids with one nick in each strand are formed, the relaxed plasmids are transformed to competent cells and sealed as closed plasmids. CPEC strategy is not only suitable for the cloning of an individual gene but also for gene libraries, combinatorial libraries and multi-fragment plasmid assembling. With its simplicity, cost-effectiveness and high transformation efficiency, CPEC has become the most convenient, economical and accurate cloning method at the present day, especially for cloning applications with high complexity.
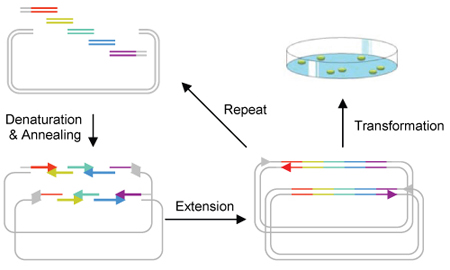
One-Step Construction of a Bioplastic Production Pathway using CPEC
We applied CPEC in the construction of a multi-component pathways in a single reaction. The proposed multi-way cloning mechanism is shown in Figure 1b. First, we constructed the biobrick parts containing genes involved in this pathway. Then we used multi-way CPEC for construction of a metabolic pathway for synthesizing a biodegradable plastic, poly(3HB-co-4HB) in E. coli.Biodegradable Plastic Synthesis Pathway in E. coli.
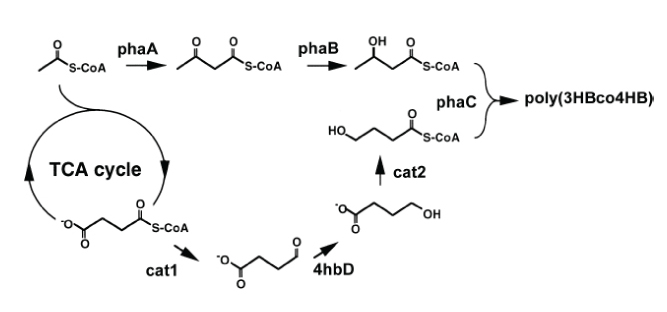
Recently, polyhydroxyalkanoates (PHA) have received a lot of attention for their potential use as bioplastic. They are naturally occurring biopolymers – under low nitrogen, phosphorus, or oxygen conditions, many bacteria produce protein- and lipid-coated granules of PHA for energy storage, and these granules can then in turn be harvested in large quantities as bioplastics (Fig. 2). Bioplastics not only help reduce the reliance on petroleum-based plastics, but also have a wide range of applications in the agricultural and medical fields because they are biodegradable and biocompatible. The most common PHA is poly(3-hydroxybutyrate), or poly(3HB), which is produced by the bacteria Cupriavidus necator. Three genes – phaA, phaB, and phaC – make up the phaCAB operon that synthesizes the enzymes required for formation of poly(3HB), which is not elastic enough for general plastic applications. But when phaCAB is expressed with cat2, a succinate degradation gene from Clostridium kluyveri that produces the 4HB monomer, the copolymer poly(3-hyroxybutyrate-co-4-hydroxybutyrate), or poly(3HB-co-4HB), is produced (Figure 3). Poly(3HB-co-4HB) is more elastic and thus has potential to be used in a variety of applications.
However, bioplastic production is currently too costly to be used widely. For example, poly(3HB-co-4HB) pathways that have been developed so far produce copolymers with very low concentrations of 4HB, the monomer that gives the copolymer its elastic properties. This could be because the enzyme that catalyzes PHA formation, PHA synthase, has higher affinity for the 3HB-CoA substrate (100%) than the 4HB-CoA substrate (0.03%).
In addition, the 3HB
pathway uses acetyl-CoA, an input to the citric acid cycle, as a precursor, but cat2 uses succinyl-
CoA, an intermediate in the cycle. Since more processing is required to obtain the 4HB cursor, the pathway may naturally favor production of the 3HB polymer, resulting in higher 3HB composition in the final copolymer.
Thus, this project aims to develop a more efficient biopathway for poly(3HB-co-4HB) while increasing the 4HB monomer composition predictably. To achieve this goal, we designed vectors with various combinations of the phaCAB operon and the cat2 gene so that genetic manipulations can be made to the genes for PHA synthase and precursor processing to
increase bioplastic production and quality.
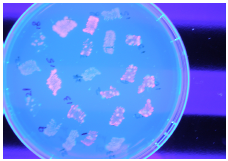
3HB genetic pathway
The phaCAB operon genomic DNA from Cupriavidus necator was cloned into PCR Blunt II-TOPO vector as the backbone and the cloning product was transformed into bacteria cells which were then plated on kanamycin (50 μg/ml) plates. Successful insertion of phaCAB was shown by Nile Red fluorescence in colonies (Figure 4).4HB genetic pathway
The cat2 gene was inserted into the pSOS vector to obtain the pSOS-cat2 construct. To obtain 4HB production, though, the phaC gene must also be expressed. The 4HB precursor γ-butyrolactone (GBL) is required for 4HB production in this pathway, as the 4HB precursor does not naturally exist in E. coli.
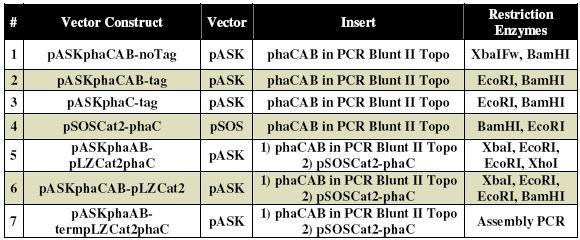
pASK vector constructs
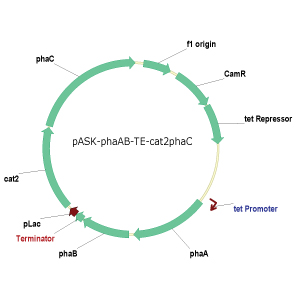
The PCR-Blunt II-phaCAB and pSOS-cat2 plasmids described above were then used as templates to create seven different vector constructs in pASK (Tables 1 and 2). The first six vector constructs were created by PCR amplification of the insert, restriction digestion of the vector and insert, ligation of the fragments, and finally transformation into E. coli. The seventh vector (pASKphaAB-term-pLZCat2phaC) was created using CPEC cloning and transformation into E. coli (Figure 5). All the constructs were sequenced to ensure that the constructs were correct. Aslo genotyping was done for the final construct 7 (Figure 6).
Screening Experiments
Two different experiments were conducted to investigate (1) the optimal anhydrotetracylcine level for inducing expression of bioplastic genes and (2) Nile Red as a screening technique for polymer production. To determine the optimal anydrotetracycline levels for phaCAB and cat2 gene expression, agar plates were prepared with 0, 5, 50, 150, and 200 ng/ml of anyhdrotetracyline, and Construct 7 E. coli were grown on them overnight at 37°C. The number of colonies on each plate was then compared the following day. To test Nile Red as a screening technique to detect PHA production, agar plates were prepared with various levels of Nile Red (0, 0.2, 0.5,1.0 μg/ml). Bacteria that were known to produce PHA (positive control) and negative control bacteria were then plated and grown overnight at 37°C. After sitting on the benchtop for a few days, they were examined under equal levels of UV lighting.
Polymer harvesting
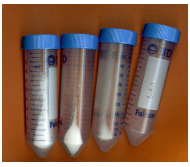
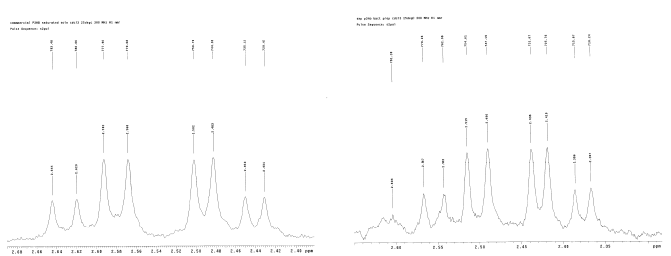
To obtain in vivo PHA polymer, E. coli containing the desired construct were grown in 3 mL of LB broth overnight, of which 1 mL was then cultured in 50 mL of LB broth and grown at 30oC until the cells reached an optical density of .7 A (600 nm). Glucose was added to a final concentration of 1%, and the cells were harvested after 48 hour of incubation at 37°C. To harvest, the cells were centrifuged at 3500 rpm and suspended in saline (0.0067 M phosphate buffer) solution. The cells were re-centrifuged to obtain a washed pellet, freezethawed in dry ice, and then freeze-dried by lyophilization. The pellet was shaken overnight at 37°C in chloroform, after which bacterial cell waste was filtered out using Whatman paper and a vacuum flask. The polymer was then precipitated from the filtered solution by adding 10 volumes of ethanol, centrifuged, and then re-dissolved in chloroform. Finally, 10 volumes of methanol was used to re-precipitate the polymer before centrifuging to obtain a purified polymer pellet. A promising sign was that there was much more white powder precipitate in the experimental samples than the negative control samples after harvesting (Figure 7).1H NMR Spectroscopy
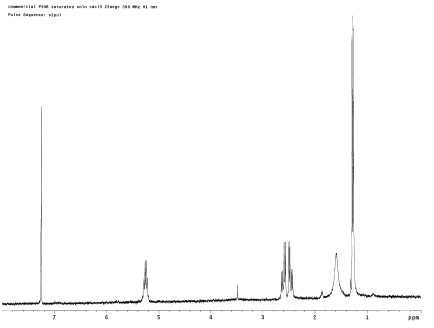
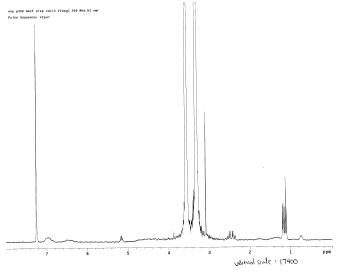
To characterize harvested polymers, 300 MHz 1H NMR spectroscopy was used. The NMR sample was prepared by re-dissolving 20 mg of each polymer sample in 600 μl of deuterochloroform in an NMR tube. Attempts were made to harvest and identify the 3HB, 4HB, and poly(3HB-co-4HB) polymers. The first harvest attempt was made on pUC19phaCAB and then on pASK-phaCAB-no tag (Construct 1) to try and obtain 3HB polymer; the NMR results for the harvested product were compared to that of a negative control (no insert) and commercial 3HB. Several harvest attempts were also made on pSOS-cat2phaC bacteria (Construct 4) to try to confirm 4HB polymer production. Finally, bacteria transformed with pASKphaAB-termpLZCat2phaC (Construct 7) were harvested in an attempt to identify poly(3HB-co-4HB) by NMR.
feasible economically.
Biobricks
In order to facilitate the completion of this project, several biobricks were created and used. The two foremost (the ones submitted) were phaAB and a double terminator.phaAB
Part: BBa_K282000
Used in the creation of bioplastics. Contains both the PhaA gene and the PhaB gene. Part of the pathway PhaAB - Terminator - Cat2PhaC .
Terminator
Part: BBa_K282001
Based off of Part:BBa_B0014. Comprised of forward terminator Part:BBa_B0010 and reverse terminator Part:BBa_B0012.
Contents |
Calendar
Protocols
CPEC Cloning
Materials
- Phusion™ High-Fidelity PCR Kit (FINNZYMES, Cat. No. F-553)
- Thermocycler
Preparation
5x Phusion HF Buffer 4 ul 10 mM dNTPs 0.4 ul Vector 50 ng/1kb Insert x ng* Phusion DNA Polymerase 0.2 ul ------------------------------- H2O to 20 ul
*The amount of insert is determined so that the molar ratio for vector and insert is 1 to 2.
Procedures
98°C 30sec
10X
98°C 10 sec
Annealing** 30 sec
72°C x sec***
72°C 5min
4°C hold
** Anneal at Tm + 3°C. The Tm should be calculated with the nearest-neighbor method.
***The extension time is usually calculated according to the shortest piece with 15 sec /kb if the cloning is not complicated. For example, if there is only one insert and is shorter than the vector, say, 600 bp, then I will use 15 sec for extension. Refer to the published paper for detailed information.
DNA Purification
Materials :
- E.Z.N.A Gel Purification Kit (Omega Bio-Tek, Cat No. D2500-02 )
- Water bath equilibrated to 55-65C
- Microcentrifuge capable of at least 10,000 x g
- Nuclease-free 1.5 ml centrifuge bottles
- Absolute (95%-100%) ethanol
- Protective eye-wear
- Isopropanol (for fragments < 500 bp only)
Protocol:
- Perform agarose gel electrophoresis to fractionate DNA fragments. Any type or grade of agarose may be used. It is strongly recommended, however, that fresh TAE buffer or TBE buffer be used as running buffer. Do not re-use running buffer as its pH will increase and reduce yields.
- When adequate separation of bands has occurred, carefully excise the DNA fragment of interest using a wide, clean scalpel.
- Determine the approximate volume of the gel slice by weighing it in a clean 1.5 ml microfuge tube. Assuming a density of 1 g/ml of gel, the volume of gel is derived as follows: A gel slice of mass 0.3 g will have a volume of 0.3 ml. Add equal volume of Binding Buffer (XP2). Incubate the mixture at 55C-60C for 7 min or until the gel has completely melted. Mix by shaking or inverting the tube every 2-3 minutes. Centrifuge the tube briefly to collect all the liquid to the bottom of the tube.
Note: For DNA fragment less than 500bp, add 1 sample volume of isopropanol after the addition of Binding Buffer (XP2).
- Apply up to 700 ul of the DNA/agarose solution to a HiBind® DNA spin column assembled in a clean 2 ml collection tube (provided) and centrifuge in a microcentrifuge at 8,000-10,000 x g for 1 min at room temperature. Discard the liquid. Re-use the collection tube in Steps 5-8. For volumes greater than 700 ul, load the column and centrifuge successively, 700 ul at a time. Each HiBind® spin-column has a total capacity of 25-30 ug DNA.
- Discard liquid and add 300ul Binding Buffer. Centrifuge at 10,000 x g for 1 minutes.
- Add 700 ul of SPW Buffer diluted with absolute ethanol into the column and wait 2-3 minutes. Centrifuge at 10,000 x g for 1 min at room temperature to wash the sample.
- Discard liquid and repeat step 6 with another 700 ul SPW Buffer.
- Discard liquid and, re-using the collection tube, centrifuge the empty column for 1 min at maxi speed (>13,000 x g) to dry the column matrix. This drying step is critical for good DNA yields.
- Place column into a clean 1.5 ml microcentrifuge tube (not provided). Add 30-50 ul depending on desired concentration of final product) Elution Buffer (or sterile deionized water) directly to the center of the column matrix, then incubate for 1 minute. Centrifuge 1 min at maxi speed (>13,000 x g) to elute DNA. This represents approximately 70% of bound DNA. An optional second elution will yield any residual DNA, though at a lo
PCA (Polymerase Cycle Assembly)
Materials
- Phusion™ High-Fidelity PCR Kit (FINNZYMES, Cat. No. F-553)
- Thermocycler
Preparation
| 5x Phusion HF Buffer | 5 ul |
| 10 mM dNTPs | 0.5 ul |
| Oligo mixture | 125 ng /250 ng /500 ng / |
| Phusion DNA Polymerase | 0.25 ul |
| --------------------- | -------- |
| H2O | to 25 ul |
Procedures
98°C 30sec
40X
98°C 7 sec
70-50°C slow ramp, 0.1°C/sec
50°C 30 sec
72°C 15 sec /kb
72°C 5 min
4°C hold
PCR Product Clean-up for DNA Sequencing
Materials:
- ExoSAP-IT® (usb, Cat. No. 78200)
- Thermocycler
Protocol:
- Remove ExoSAP-IT from -20°C freezer and keep on ice throughout this procedure.
- Mix 5 μl of a post-PCR reaction product with 2 μl of ExoSAP-IT for a combined 7 μl reaction volume.
- Incubate at 37°C for 15 min to degrade remaining primers and nucleotides.
- Incubate at 80°C for 15 min to inactivate ExoSAP-IT.
- The PCR product is now ready for use in DNA sequencing etc.
PCR
Materials
- Phusion™ High-Fidelity PCR Kit (FINNZYMES, Cat. No. F-553)
- Thermocycler
Preperation
| 5x Phusion HF Buffer | 10 ul |
| 10 mM dNTPs | 1 ul |
| DNA template | 1 pg – 10 ng |
| Forward primer (10 uM) | 2.5 ul |
| Reverse primer (10 uM) | 2.5 ul |
| Phusion DNA Polymerase | 0.5 ul |
| -------------------- | ----- |
| H2O | to 50 ul |
Procedure
98°C 30sec
30X
98°C 10 sec
Annealing* 30 sec
72°C 15 sec per 1 kb
72°C 5min
4°C hold
* Anneal at Tm + 3°C. The Tm should be calculated with the nearest-neighbor method.
Single Colony PCR
Materials
- Taq DNA Polymerase with Standard Taq Buffer (NEB, Cat. No. M0273)
- 10 mM dNTP Mix (NEB, Cat. No. N0447)
- Thermocycler
Preperation
| 10x Standard Taq Buffer | 2 ul |
| 10 mM dNTPs | 0.4 ul |
| Bacteria culture* | 1 ul |
| Forward primer (10 uM) | 1 ul |
| Reverse primer (10 uM) | 1 ul |
| Taq DNA Polymerase | 0.2 ul |
| ----------------- | ------- |
| H2O | to 20 ul |
*Bacteria culture refers to E. coli cultured in LB solution overnight.
Procedures
94°C 5 min
25X
94°C 15 sec
Annealing** 30 sec
72°C 1 min per kb
72°C 5min
4°C hold
**Anneal at Tm which is calculated with salt-adjusted method.
Transformation
Materials:
- GC5 Chemical Competent Cells (Genesee Scientific, Cat. No. 42-653)
- SOC Medium (Sigma, Cat. No. S1797)
- LB Agar (Sigma, Cat. No. L3027)
- Petri Dishes (VWR, Cat. No. SC25373-187)
- Cell Spreader (VWR, Cat. No. 89042-018)
- 37°C incubator
- 37°C shaker
- water bath
Protocol:
1. Thaw 1 tube of competent cells on ice;
2. Add 3 ul of cloning product or 1-50 ng of plasmid into competent cells while stirring gently;
3. Keep the tube covered by ice for 30min;
4. Heat-shock the competent cells in water bath for 45 sec at 42°C;
5. Put the tube on ice for 2 minutes;
6. Add 450 ul of SOC medium and then put it in a 37°C shaker for 1 hour;
7. Dilute and spread an appropriate amount on an LB agar plate with the appropriate antibiotics;
8. Place the plate up-side-down in 37°C incubator for 16-18 hours (overnight).
NMR
- Login and start NMR program.
- Click Acqi. Load sample. Make sure spin is on and lock is off.
- Go to lock. Decrease number of sine waves to 1 to obtain step function. Make sure Zo is 1100, lockpower is below 30, lockgain is 36, lockphase is 352, and spin is 20.
- Go to shim. Increase lock level.
- Click main menu. Click set up. Click H1CDCl3. Type ‘nt=64’ (number of scans), ‘ss=2’ (dummy scans), ‘go’, ‘lb=0.2’ (line broadening).
- Viewing: Type ‘wft’, ‘dscale’, ‘dfp’ to display peak frequency.
- Zooming in: Click display. Go to interactive. Type ‘cr=8p, delta=8p’ to set width of zoom. Use cursor to set boundaries. Click to cut.
- Printing: Type “pL pscale(0) pltext ppf page’ to print spectra with text and peak frequencies.
Lab Safety
None of our project ideas raised any safety issues whether it be researcher, environmental, or public safety. Duke has an in-house review board for projects and they did not raise any flags regarding our project. The biobrick parts created also do not pose any safety issues.
Students
Faculty Advisors

| Maggie Jiayuan Quan jq7(at)duke.edu Graduate Student | 
| Faisal Reza faisal.reza(at)duke.edu Graduate Student |
Sponsors

| 
| 
|
 "
"