Team:Virginia/Approach
From 2009.igem.org
| (One intermediate revision not shown) | |||
| Line 29: | Line 29: | ||
Part: [http://partsregistry.org/Part:BBa_K231001 BBa_K231001] | Part: [http://partsregistry.org/Part:BBa_K231001 BBa_K231001] | ||
This enzyme catalyzes the conversion of glutathione (GSH) to phytochelatin (PC). Its precursor GSH exists endogenously in ''E. coli'' making integration of PCS function relatively simple. The gene was codon and synthesized in an IPTG inducable pJexpress vector from DNA2.0. This construct was then expressed in JW3469. Induced samples displayed significantly faster growth in the presence of arsenic compared to uninduced samples. (See [[Team:Virginia/Data|Results]]). | This enzyme catalyzes the conversion of glutathione (GSH) to phytochelatin (PC). Its precursor GSH exists endogenously in ''E. coli'' making integration of PCS function relatively simple. The gene was codon and synthesized in an IPTG inducable pJexpress vector from DNA2.0. This construct was then expressed in JW3469. Induced samples displayed significantly faster growth in the presence of arsenic compared to uninduced samples. (See [[Team:Virginia/Data|Results]]). | ||
| + | |||
| + | ==Arsenic binding peptide== | ||
| + | [[Image:Ecoli_mod1.png|thumb|400px|The synthetic metallothionein ABP binds As(III) much like PC]] | ||
| + | Part: [http://partsregistry.org/Part:BBa_K231000 BBa_K231000] | ||
| + | This synthetic peptide has the same properties as the natural PC which give it arsenic binding function. However we eliminated the need for the metabolically stressful synthesis and operation of an additional protein. The end product is relatively simple therefore it can be directly synthesized from amino acid precursors. ABP takes advantage of rational peptide engineering to optimize its binding properties. | ||
| + | |||
| + | ABP was synthesized in an inducible expression vector much like PCS and expressed in KO cells. (See [[Team:Virginia/Data|Results]]). | ||
| + | |||
| + | |||
| + | |||
| + | |||
Latest revision as of 03:51, 22 October 2009
edit
- Remove the arsenite efflux mechanism in E. coli
- Create an arsenic sequestration mechanism
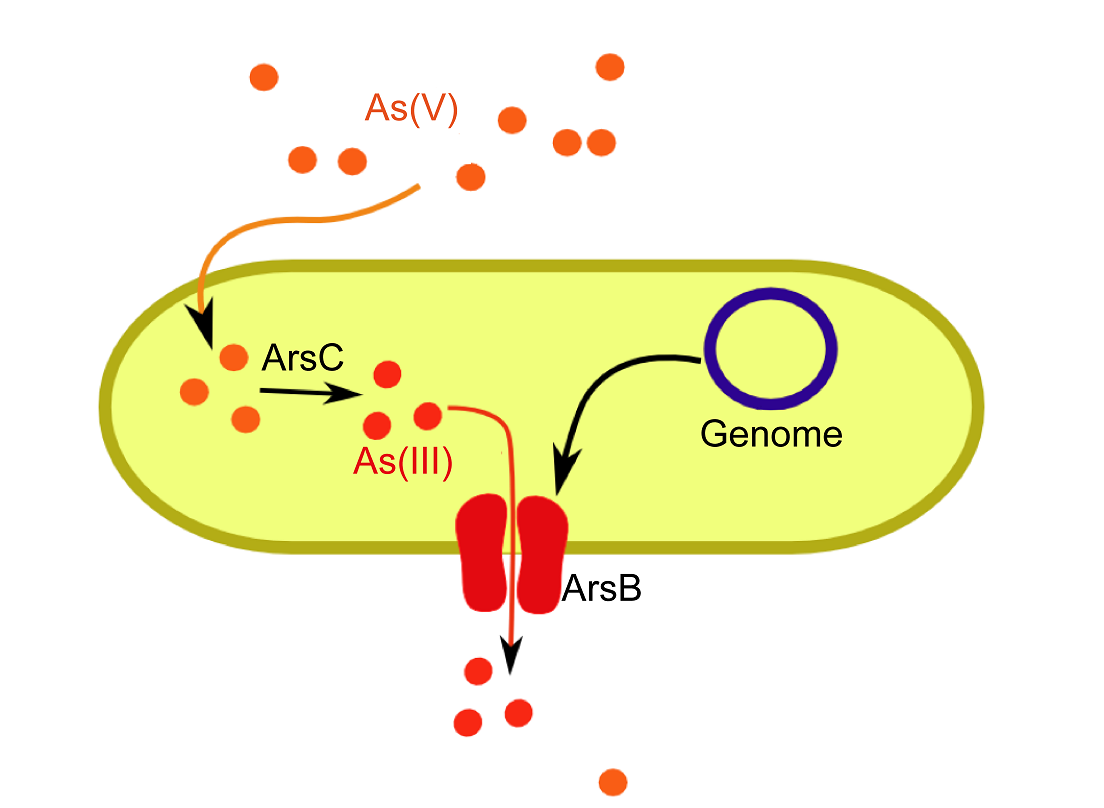
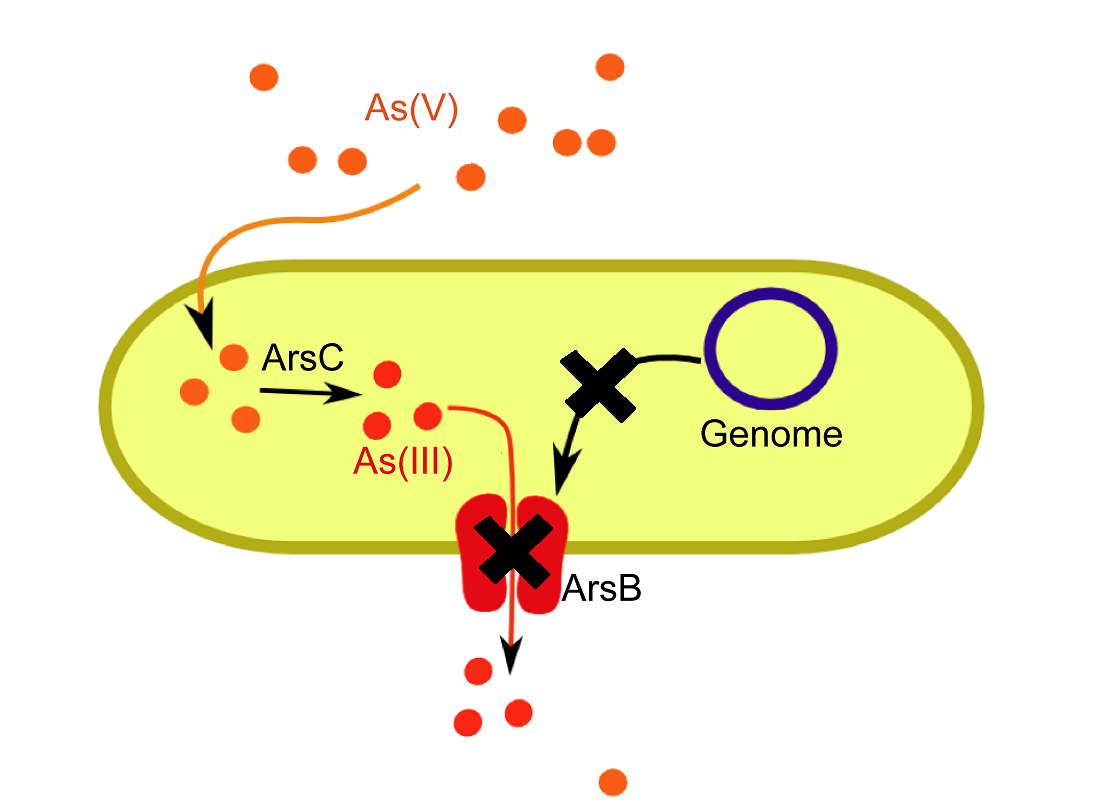
ArsB Knock-out
Escherichia coli uses the [http://ecoliwiki.net/colipedia/index.php/arsB arsB] ion transporter to specifically transport arsenite (As(III)) ions out of the cytoplasm. We obtained a [http://ecoli.naist.jp/GB6/info.jsp?id=JW3469 single gene knock-out for arsB] from the [http://ecoli.naist.jp/gb6/Resources/deletion/deletion.html Keio Collection]. This KO strain (JW3469) performed was verified in control growth curves in the presence of arsenic. (See Results).
Sequestration Mechanism
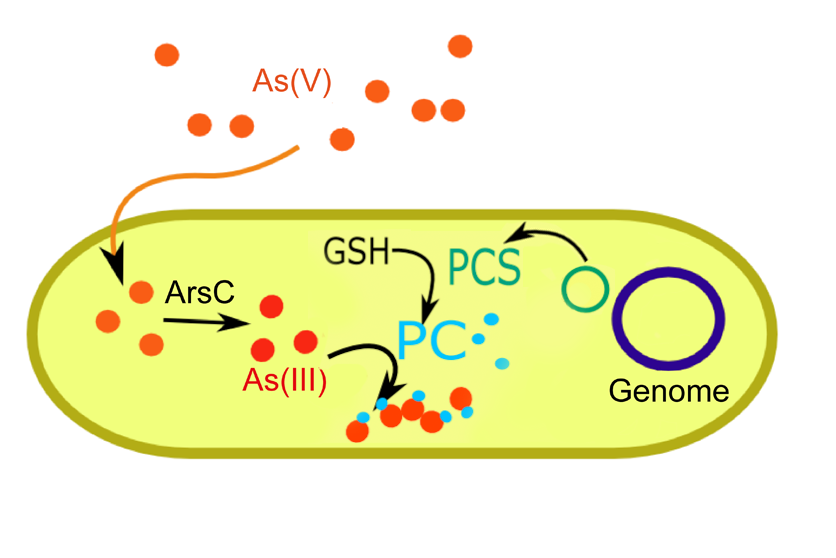
Most microbial detoxification/resistance strategies involve efflux of heavy metals. In plants such as Arabidopsis such functions have not been observed. They may be explained by plants' inability to simply pump toxins into a surrounding solute. Instead they have evolved the ability to synthesize metallothioneins. Arabidopsis specifically synthesizes phytochelatins, a type of metallothionein. These cysteine rich peptides provide alternative binding sites for arsenic and other heavy metals.
Phytochelatin synthase
Part: [http://partsregistry.org/Part:BBa_K231001 BBa_K231001] This enzyme catalyzes the conversion of glutathione (GSH) to phytochelatin (PC). Its precursor GSH exists endogenously in E. coli making integration of PCS function relatively simple. The gene was codon and synthesized in an IPTG inducable pJexpress vector from DNA2.0. This construct was then expressed in JW3469. Induced samples displayed significantly faster growth in the presence of arsenic compared to uninduced samples. (See Results).
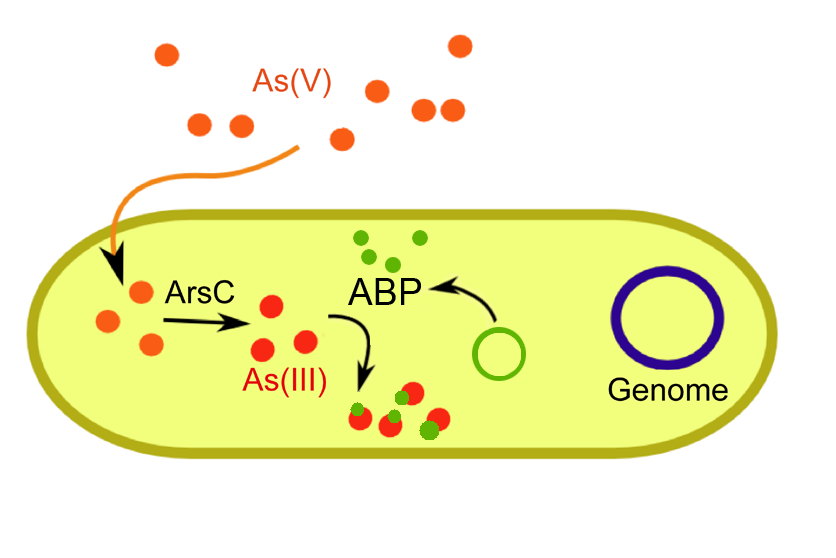
Arsenic binding peptide
Part: [http://partsregistry.org/Part:BBa_K231000 BBa_K231000] This synthetic peptide has the same properties as the natural PC which give it arsenic binding function. However we eliminated the need for the metabolically stressful synthesis and operation of an additional protein. The end product is relatively simple therefore it can be directly synthesized from amino acid precursors. ABP takes advantage of rational peptide engineering to optimize its binding properties.
ABP was synthesized in an inducible expression vector much like PCS and expressed in KO cells. (See Results).
Safety
- Would any of your project ideas raise safety issues in terms of: (researcher safety, public safety, or environmental safety?)
- The University's Environmental Health and Safety department was involved in our project from the beginning. Team members absolved training to handle arsenic and proper precautions were taken in the lab for storage.
- Is there a local biosafety group, committee, or review board at your institution?
- See above.
- What does your local biosafety group think about your project?
- See above.
- Do any of the new BioBrick parts that you made this year raise any safety issues?
- Hopefully they will make the world safer rather than raise safety issues! (No)
 "
"