|
|
| (77 intermediate revisions not shown) |
| Line 1: |
Line 1: |
| - | {| cellpadding="0" cellspacing="0" border="0" width="900" align="center" | + | {{Tsinghua/Header+}} |
| - | !align="center"|[[Team:Tsinghua|Home]]
| + | {{Tsinghua/Header2}} |
| - | !align="center"|[[Team:Tsinghua/Team|The Team]]
| + | {{Tsinghua/ProjectHeader}} |
| - | !align="center"|[[Team:Tsinghua/Project|The Project]]
| + | |
| - | !align="center"|[[Team:Tsinghua/Protocol|Protocols]]
| + | |
| - | !align="center"|[[Team:Tsinghua/Parts|Parts]]
| + | |
| - | !align="center"|[[Team:Tsinghua/Modeling|Modeling]]
| + | |
| - | !align="center"|[[Team:Tsinghua/Notebook|Notebook]]
| + | |
| - | !align="center"|[[Team:Tsinghua/Brainstorming|Brainstorming]]
| + | |
| - | |}
| + | |
| | | | |
| - | {|align="justify"
| |
| - | |You can write a background of your team here. Give us a background of your team, the members, etc. Or tell us more about something of your choosing.
| |
| - | |[[Image:Example_logo.png|200px|right]]
| |
| - | |-
| |
| - | |
| |
| - | ''Tell us more about your project. Give us background. Use this is the abstract of your project. Be descriptive but concise (1-2 paragraphs)
| |
| | | | |
| - | This page will be further decorated later with necessary figures incorporated.
| + | [[Image:project.png|center|frameless|Overall Project|500px]] |
| - | ''
| + | |
| - | |[[Image:Team.png|right]]
| + | |
| - | |-
| + | |
| - | |}
| + | |
| | | | |
| - | <!--- The Mission, Experiments --->
| |
| | | | |
| - | {| style="color:#1b2c8a;background-color:#0c6;" cellpadding="3" cellspacing="1" border="1" bordercolor="#fff" width="70%" align="center"
| + | =Overall Project= |
| - | !align="center"|[[Team:Tsinghua|Home]]
| + | Our aim is to construct a targeted gene therapy vector with high cellular specificity, considerable capacity and the potential for mass production and universal modification. Analogizing the characteristics of bacteriophage lambda and adenovirus, we genomically engineered the fiber protein of adenovirus with the pC of bacteriophage lambda, with the knob region modified by cell-specific peptides generated by phage display (called targeted bioBrick). After inducing the vector genome (generated by bottom-up or top-down approach) into BL21 DE3 E.coli strain, we applied a co-transformed therapeutic DNA (namely a cosmid with a capacity of 40-50 kb) for mass production of our targeted gene therapy vectors containing the desired genes to be delivered. With the targeted bioBrick mediating the attachment and RGD domain mediating the internalization of the targeted vector, we are able to accomplish the targeted gene therapy. |
| - | !align="center"|[[Team:Tsinghua/Team|The Team]]
| + | |
| - | !align="center"|[[Team:Tsinghua/Brainstorming|Brainstorming]]
| + | |
| - | !align="center"|[[Team:Tsinghua/Project|The Project]]
| + | |
| - | !align="center"|[[Team:Tsinghua/Parts|Parts]]
| + | |
| - | !align="center"|[[Team:Tsinghua/Modeling|Modeling]]
| + | |
| - | !align="center"|[[Team:Tsinghua/Notebook|Notebook]]
| + | |
| - | |}
| + | |
| - | (''Or you can choose different headings. But you must have a team page, a project page, and a notebook page.'') | + | |
| | | | |
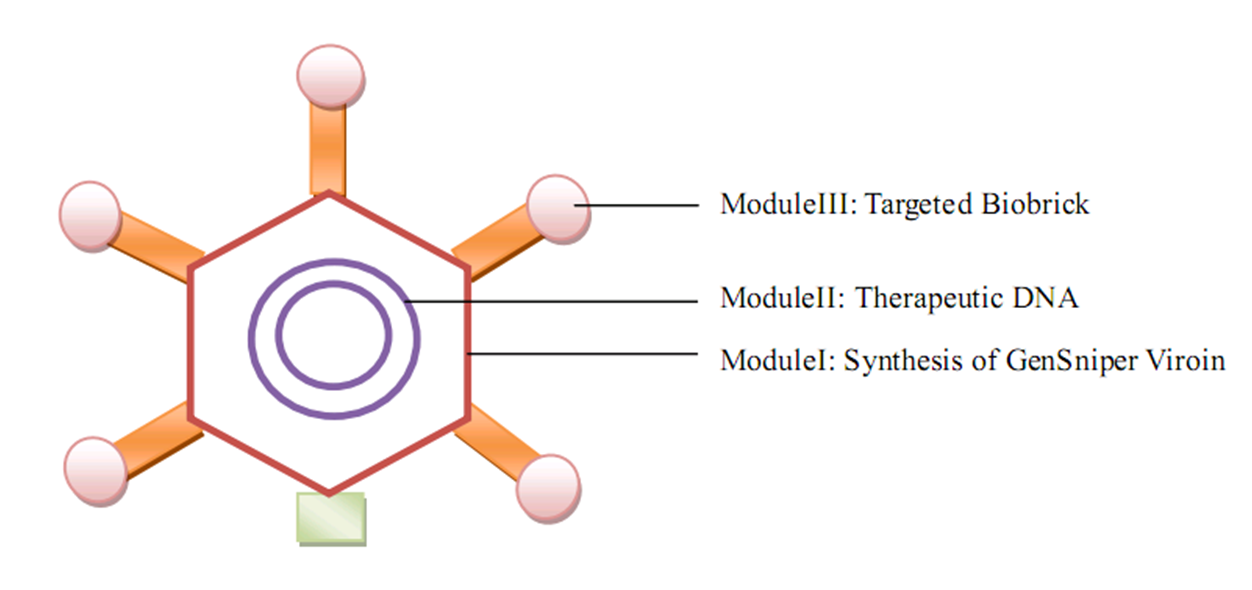
| | + | ==ModuleI: Synthesis of GenSniper Viroin== |
| | + | Using two synthetic biological strageties (bottom-up and top-down), we will construct a genome of our desired gene therapy vector, which consists of the structural proteins for bacteriophage lambda head assembly and lysis genes (and targeted bioBrick later). Our aim in this module is to generate the viroin of our gene therapy vector (namely the head structure of bacteriophage lambda). |
| | | | |
| | + | ==ModuleII: Therapeutic DNA== |
| | + | In this module, we plan to construct a cosmid (a molecular cloning with a cos site) encoding the conceptual therapeutic genes, because the structural proteins of bacteriophage lambda will only package the circular DNA with cos site and 40-50 kb in size. Here we will also identify the function of this module through the expression of RFP. |
| | | | |
| - | == '''Overall project''' == | + | ==ModuleIII: Targeted BioBrick== |
| | + | This module is the modification of bacteriophage lambda protein C by fiber shaft at its N termini. When constructing this targeted bioBrick, we will apply PCR to add the targeted peptide (in wetlab we just use TAT peptide) and RGD sequence into the fusion protein, forming a peptide-fiber shaft-RGD-C reading frame. |
| | | | |
| - | Your abstract
| + | {{Tsinghua/Pic}} |
| - | | + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | == '''Background''' ==
| + | |
| - | | + | |
| - | === Gene Therapy ===
| + | |
| - | Gene therapy is the insertion of genes into an individual's cells and tissues to treat a disease, such as a hereditary disease in which a deleterious mutant allele is replaced with a functional one[1]. Although the technology is still in its infancy, it is one of the most promising and active research fields in medicine[1,2]. Antisense therapy is not strictly a form of gene therapy, but is a genetically-mediated therapy and is often considered together with other methods[1].
| + | |
| - | | + | |
| - | === Vectors in Gene Therapy ===
| + | |
| - | Despite substantial progress, a number of key technical issues need to be resolved before gene therapy can be safely and effectively applied in the clinic, and an ideal gene delivery vector is one of the bottlenecks in gene therapy application[3]. Generally, vectors applied in gene therapy can be classified into viral or non-viral.
| + | |
| - | | + | |
| - | ==== Viral Methods ====
| + | |
| - | All viruses bind to their hosts and introduce their genetic material into the host cell as part of their replication cycle. This genetic material contains basic 'instructions' of how to produce more copies of these viruses, hijacking the body's normal production machinery to serve the needs of the virus[1].
| + | |
| - | | + | |
| - | Adenovirus.
| + | |
| - | | + | |
| - | Adeno-associated virus.
| + | |
| - | | + | |
| - | Retroviruses.
| + | |
| - | | + | |
| - | ==== Non-Viral Methods ====
| + | |
| - | Non-viral methods present certain advantages over viral methods, with simple large scale production and low host immunogenicity being just two. Previously, low levels of transfection and expression of the gene held non-viral methods at a disadvantage; however, recent advances in vector technology have yielded molecules and techniques with transfection efficiencies similar to those of viruses[1].
| + | |
| - | | + | |
| - | Naked DNA.
| + | |
| - | | + | |
| - | Oligonucleotides.
| + | |
| - | | + | |
| - | Lipoplexes and polyplexes.
| + | |
| - | | + | |
| - | === Frontiers in Gene Therapy Research ===
| + | |
| - | | + | |
| - | ==== Induced Pluripotent Stem Cell (iPS) and Gene Delivery System====
| + | |
| - | | + | |
| - | ==== Therapeutic microRNA Delivery====
| + | |
| - | Therapeutic strategies based on modulation of microRNA (miRNA) activity hold great promise due to the ability of these small RNAs to potently influence cellular behavior[4].
| + | |
| - | | + | |
| - | ==== Cancer Gene Therapy====
| + | |
| - | As a primary threat of human health, cancer causes about 13% of all human deaths[5]. According to the American Cancer Society, 7.6 million people died from cancer in the world during 2007[6]. Current treatments often have far reaching negative side effects[7]. The systemic toxicity of chemotherapy regimens still often result in acute and delayed nausea, mouth ulcerations and mild cognitive impairments[8].
| + | |
| - | | + | |
| - | ==== References====
| + | |
| - | [1] http://en.wikipedia.org/wiki/Gene_therapy
| + | |
| - | | + | |
| - | [2] SM Selkirk. Gene therapy in clinical medicine. ''Postgraduate Medical Journal''.2004;80:560-570; doi:10.1136/pgmj.2003.017764
| + | |
| - | | + | |
| - | [3] RC Mulligan. The basic science of gene therapy. ''Science'', 1993, Vol 260, Issue 5110, 926-932.
| + | |
| - | | + | |
| - | [4] Janaiah Kota, Raghu R. Chivukula, Kathryn A. O’Donnell, Erik A. Wentzel, Chrystal L. Montgomery, Hun-Way Hwang, Tsung-Cheng Chang, Perumal Vivekanandan, Michael Torbenson, K. Reed Clark, Jerry R. Mendell,and Joshua T. Mendell. Therapeutic microRNA Delivery Suppresses Tumorigenesis in a Murine Liver Cancer Model. ''Cell''. 2009, 137. 1005–1017.
| + | |
| - | | + | |
| - | [5] WHO (February 2006). "Cancer". World Health Organization. http://www.who.int/mediacentre/factsheets/fs297/en/. Retrieved on 2007-06-25.
| + | |
| - | | + | |
| - | [6] American Cancer Society (December 2007). "Report sees 7.6 million global 2007 cancer deaths". Reuters. http://www.reuters.com/article/healthNews/idUSN1633064920071217. Retrieved on 2008-08-07.
| + | |
| - | | + | |
| - | [7] Peter Sinnaeve, Olivier Varenne, Désiré Collen, and Stefan Janssens. Gene therapy in the cardiovascular system: an update. ''Cardiovasc Res'', Dec 1999; 44: 498 - 506.
| + | |
| - | | + | |
| - | [8] Chemotherapy and you: A guide to self-help during cancer treatment. National Institutes of Health Web site. Available at: http://www.cancer.gov/PDF/b21d0a74-b477-41ec-bdc0-a60bbe527786/chemoandyou.pdf. Accessed July 31, 2006.
| + | |
| - | | + | |
| - | == Project Design==
| + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | | + | |
| - | === Introduction ===
| + | |
| - | Although several successive gene therapeutic approaches have been reported[1,2], an ideal gene delivery system with targeted specificity, high efficiency and safety is still not available[1,3,4]. Generally speaking, these factors to a large extent depend on the gene therapy vectors used[5]. In other words, the gene delivery system is still a bottleneck as well as a universal problem in the practical fields of gene therapy.
| + | |
| - | | + | |
| - | | + | |
| - | Our project is aimed at applying the ideas of synthetic biology at the genomic level and building a targeted gene therapy vector that can be applied with respect to the certain need of specificity. Also, we intend to propose a procedure for selecting certain synthetic gene vectors with specificity of one’s interest based our design gene therapy vectors. Mathematic modeling focus on both the synthesis of the targeted gene therapy vectors as well as the evaluation of the specificity selection procedure.
| + | |
| - | | + | |
| - | | + | |
| - | Generally, we have two sub-projects. One is the synthetic biology approach in simulation with the established viral vectors. However, the synthetic gene therapy vector will not be as cytotoxic as the commonly applied viral vectors. The other is the synthetic biology approach on the basis of naked plasmid vectors, while the efficiency and specificity of the gene therapy vector can be ensured by synthetic biology modification. The two sub-projects are interconnected in that the flexible synthetic domains of the key proteins are truly interchangeable.
| + | |
| - | | + | |
| - | ===Project1===
| + | |
| - | ==== Basic Idea ====
| + | |
| - | Project1 is aimed at synthesizing a gene therapy vector which is structurally and functionally similar to a commonly used viral vector termed adenovirus vector[6]. However, in order to achieve the synthetic biology standard for human practice in the realm of gene therapy, this gene therapy vector should be industrially easy to manipulate in its production and genetically easy to modify in its specificity. Also, based on the social implication of synthetic biology[7,8], the synthetic gene therapy vector must be safe for possible clinical use.
| + | |
| - | | + | |
| - | | + | |
| - | We compared and contrasted the structure of viron between adenovirus and bacteriophage lambda, and found the following facts: 1) the shapes of the viron of both adenovirus and bacteriophage lambda is a regular icosohedron[9], while the adenovirus protein (Fiber) that determines its specificity is positioned on the vertices of the icosohedral viron, which can specifically bind with a receptor called CAR[10]; 2) CAR is widely distributed on the plasma membrane of various types of cells[11-13], which partially contributes to its poor specificity to the target cells as well as its potential cytotoxicity[9,10]; 3) the proliferation of bacteriophage lambda is solely on the basis of its host E.coli, while the production of adenovirus gene therapy vector normally depends on eukaryotic cell lines which are more cost-inefficient and time-consuming; 4) the vertices of adenovirus viron are composed of pentamer of protein III attached to trimer of protein fiber, while the vertices of bacteriophage lambda viron are composed of protein C encoded by lambda phage genome.
| + | |
| - | | + | |
| - | | + | |
| - | Thus, if we can manage to synthesize a bacteriophage-lambda-based gene therapy vector in simulation to the adenovirus vector but modified at the vertices position of its viron, then the production of the synthetic gene therapy vector can be simplified and much easier to manipulate. In addition, considering the low immunogenicity of lambda phage proteins[6,14], the safety of the gene therapy can be improved compared with conventional adenovirus vector.
| + | |
| - | | + | |
| - | | + | |
| - | Our Project1 is equivalent to apply synthetic biology concepts and standards at the genomic level, constructing a genome which is neither adenovirus nor bacteriophage lambda genome. This synthetic genome, however, is capable of producing standardized and targeted gene therapy vectors for human clinical practice. Also, we use the abstraction principle in our design of this genome in order to make the synthetic gene therapy vector easy to be further modified, improved and industrialized. Project1 implicates the evolvement of synthetic biology to a higher level of living organism, the genome, which meets the trends of synthetic biology innovation.
| + | |
| - | | + | |
| - | ==== Synthesis of the Gene Therapy Production System====
| + | |
| - | Based on the standard of synthetic biology, we apply two approaches to synthesize the genome of our gene therapy vector
| + | |
| - | [[Image:Team.png|right]]
| + | |
| - | | + | |
| - | ===== Bottom-Up Approach =====
| + | |
| - | As for the bottom-up approach, we amplify the target genes from the target genome(mostly structural genes from lambda phage genome, and also L5 (fiber) gene from adenovirus genome) and recombine them into one or two molecular cloning vectors according to the biobrick standard. In the synthetic genome of the gene therapy vector, gene C (which encodes the protein at the vertices of the lambda viroin) and L5 should be fused according to certain standardiztion consideration (termed targeted biobrick).
| + | |
| - | | + | |
| - | ===== Top-Down Approach=====
| + | |
| - | As for the top-down approach, we transplant the whole gene expression element under the upsteam regulation of promoter R' into a molecular cloning vector. Fortunately, the lambda promoter R' can be strongly enhanced by protein Q in the late state of wildtype phage infection, which makes a feasible scheme to regulate the structural proteins on the synthetic genome.
| + | |
| - | | + | |
| - | ====Synthesis of the Targeted Biobrick====
| + | |
| - | The synthesized genome will be capable of producing gene therapy "viroins" with protein C specially modified. This targeted biobrick enables the synthetic gene therapy vector to be targeted specifically for certain types of cells. We generally decouple this biobrick into four "modules" (will be expressed to different domains in the C-Fiber fusion protein).
| + | |
| - | | + | |
| - | ====Synthesis of the Therapeutic DNA====
| + | |
| - | | + | |
| - | ===Project2===
| + | |
| - | ==== Basic Idea ====
| + | |
| - | | + | |
| - | === References(Project1) ===
| + | |
| - | [1] David A.Williams, and Christopher Baum. Gene Therapy—New Challenges Ahead. Science. 2003, 302, 400-401.
| + | |
| - | | + | |
| - | [2] Marina Cavazzana-Calvo et al.. Immunodeficiency (SCID)-X1 Disease Gene Therapy of Human Severe Combined. Science. 2000, 288, 669-672.
| + | |
| - | | + | |
| - | [3] Esmail D. Zanjani, and W. French Anderson. Prospects for in utero human gene therapy. Science. 1999, 285, 2084-2088.
| + | |
| - | | + | |
| - | [4] Leland H. Hartwell, Leroy Hood, Michael L. Goldberg, Ann E. Reynolds, Lee M. Silver, Ryth C. Veres. Genetics: From Genes to Genome. McGrawHall, 3rd edition, 2008.
| + | |
| - | | + | |
| - | [5] http://en.wikipedia.org/wiki/Gene_therapy
| + | |
| - | | + | |
| - | [6] Jerry Guo, and Hao Xin. Splicing out the West?. Science. 2007, 314, 1232-1235.
| + | |
| - | | + | |
| - | [7] Chopra Paras, and Akhil Kamma. Engineering life through Synthetic Biology. In Silico Biology 6. http://www.bioinfo.de/isb/2006/06/0038. Retrieved on 2008-06-09.
| + | |
| - | | + | |
| - | [8] http://www.syntheticbiology.org
| + | |
| - | | + | |
| - | [9] Michael T. M., John M. M., and Jack P. Brock Biology of Microorganisms. Prentice Hall, 12th edition, 2008.
| + | |
| - | | + | |
| - | [10] Glen RN, and Phoebe LS. Role of αv integrins in adenovirus cell entry and gene delivery. Microbiology and Molecular Biology reviews. 1999, 63, 725-734.
| + | |
| - | | + | |
| - | [11] Yuanming Zhang, and Jeffrey M. Bergelson. Adenovirus Receptors. J. Virol. 2005, 79, 12125–12131.
| + | |
| - | | + | |
| - | [12] Miyazawa N, Crystal RG, and Leopold PL. Adenovirus serotype 7 retention in a late endosomal compartment prior to cytosol escape is modulated by fiber protein. J. Virol. 2001, 75, 1387–1400.
| + | |
| - | | + | |
| - | [13] Shayakhmetov DM, Eberly AM, Li ZY, and Lieber A. Deletion of penton RGD motifs affects the efficiency of both the internalization and the endosome escape of viral particles containing adenovirus serotype 5 or 35 fiber knobs. J. Virol. 2005, 79, 1053–1061.
| + | |
| - | | + | |
| - | == The Experiments ==
| + | |
| - | | + | |
| - | ==== Project 1 ====
| + | |
| - | | + | |
| - | ==== Project 2 ====
| + | |
| - | | + | |
| - | ==== Project 1 and Project 2 ====
| + | |
| - | | + | |
| - | == Results ==
| + | |
| - | | + | |
| - | ==== Project 1 ====
| + | |
| - | | + | |
| - | ==== Project 2 ====
| + | |
 "
"