August/13 August 2009
From 2009.igem.org
(Difference between revisions)
(New page: 1.<B>before Min prep</b><br> Checked the cell cultures transformed and plated out yesterday (8/10) for colony formation and made an approximate count of the number of colonies. (plate n...) |
|||
| (8 intermediate revisions not shown) | |||
| Line 3: | Line 3: | ||
(plate number)-(location on plate) (no. of colonies) | (plate number)-(location on plate) (no. of colonies) | ||
| - | + | [http://partsregistry.org/wiki/index.php?title=Part:BBa_B0034 1-2M] 100~ (Amp) | |
| - | + | [http://partsregistry.org/wiki/index.php?title=Part:BBa_F1610 2-24G] 1? | |
| - | + | [http://partsregistry.org/wiki/index.php?title=Part:BBa_R0071 1-12C] 10 | |
| - | + | [http://partsregistry.org/wiki/index.php?title=Part:BBa_R0040 1-6I] 10< | |
| - | + | [http://partsregistry.org/wiki/index.php?title=Part:BBa_C0171 2-8I] 10< | |
| - | 2-6E | + | [http://partsregistry.org/wiki/index.php?title=Part:BBa_F2620 2-6E] 10< |
| - | 1-16G | + | [http://partsregistry.org/wiki/index.php?title=Part:BBa_R0078 1-16G] 10 |
| - | 1-12A | + | [http://partsregistry.org/wiki/index.php?title=Part:BBa_R0079 1-12A] 10< |
| - | 2-8O | + | [http://partsregistry.org/wiki/index.php?title=Part:BBa_C0161 2-8O] 10< |
| - | 2-11N 10 (Kan) | + | [http://partsregistry.org/wiki/index.php?title=Part:BBa_I14015 2-11N] 10 (Kan) |
| - | + | [http://partsregistry.org/wiki/index.php?title=Part:BBa_I14017 1-18L] 50 | |
inoculate to iquid medium(5mL + Amp 5uL or Kan 25uL) | inoculate to iquid medium(5mL + Amp 5uL or Kan 25uL) | ||
| Line 20: | Line 20: | ||
after ligation | after ligation | ||
EpsE + terminator 3 → liquid medium | EpsE + terminator 3 → liquid medium | ||
| - | RBS + LasR(2-8M) 10 → rapid check | + | RBS + LasR([http://partsregistry.org/wiki/index.php?title=Part:BBa_C0179 2-8M]) 10 → rapid check |
| Line 29: | Line 29: | ||
digestion with restriction enzyme | digestion with restriction enzyme | ||
| - | + | '''K204003''' | |
| - | + | <table border="1" Frame="box"> | |
| - | + | <tr><th colspan="2">Vector</th><th colspan="2">Insert</th></tr> | |
| - | + | <tr><td>RBS([http://partsregistry.org/wiki/index.php?title=Part:BBa_B0034 1-2M])</td><td>10</td><td>cinR([http://partsregistry.org/wiki/index.php?title=Part:BBa_C0077 1-14F])</td><td>6</td></tr> | |
| - | + | <tr><td>SpeI</td><td>1</td><td>XbaI</td><td>1</td><tr> | |
| - | + | <tr><td>PstI</td><td>1</td><td>PstI</td><td>1</td><tr> | |
| - | + | <tr><td> No.2 Buffer</td><td>2</td><td>No.2</td><td>2</td></tr> | |
| - | + | <tr><td>dH<sub>2</sub>O</td><td>6</td><td>dH<sub>2</sub>O</td><td>10</td></tr> | |
| - | + | <tr><td>total</td><td>20uL</td><td>total</td><td>20uL</td></tr> | |
| - | + | </table> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| + | '''K204004''' | ||
| + | <table border="1" Frame="box"> | ||
| + | <tr><th colspan="2">Vector</th><th colspan="2">Insert</th></tr> | ||
| + | <tr><td>RBS([http://partsregistry.org/wiki/index.php?title=Part:BBa_B0034 1-2M])</td><td>10</td><td>LasR([http://partsregistry.org/wiki/index.php?title=Part:BBa_C0179 2-8M])</td><td>10</td></tr> | ||
| + | <tr><td>EcoRI</td><td>1</td><td>EcoRI</td><td>1</td><tr> | ||
| + | <tr><td>XbaI</td><td>1</td><td>SpeI</td><td>1</td><tr> | ||
| + | <tr><td> No.2</td><td>2</td><td>No.2</td><td>2</td></tr> | ||
| + | <tr><td>dH<sub>2</sub>O</td><td>4</td><td>dH<sub>2</sub>O</td><td>6</td></tr> | ||
| + | <tr><td>total</td><td>20uL</td><td>total</td><td>20uL</td></tr> | ||
| + | </table> | ||
| + | |||
| - | + | '''K204005''' | |
| - | + | ||
| - | + | <table border="1" Frame="box"> | |
| - | + | <tr><th colspan="2">Vector</th><th colspan="2">Insert</th></tr> | |
| - | + | <tr><td>[http://partsregistry.org/wiki/index.php?title=Part:BBa_I14017 1-18L]</td><td>8</td><td>[http://partsregistry.org/wiki/index.php?title=Part:BBa_I1466 1-23J]</td><td>8</td></tr> | |
| - | + | <tr><td>SpeI</td><td>1</td><td>XbaI</td><td>1</td><tr> | |
| - | + | <tr><td>PstI</td><td>1</td><td>PstI</td><td>1</td><tr> | |
| - | + | <tr><td> No.2 </td><td>2</td><td>No.2</td><td>2</td></tr> | |
| + | <tr><td>dH<sub>2</sub>O</td><td>8</td><td>dH<sub>2</sub>O</td><td>8</td></tr> | ||
| + | <tr><td>total</td><td>20uL</td><td>total</td><td>20uL</td></tr> | ||
| + | </table> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | + | '''K204006''' | |
| + | |||
| + | <table border="1" Frame="box"> | ||
| + | <tr><th colspan="2">Vector</th><th colspan="2">Insert</th></tr> | ||
| + | <tr><td>RBS([http://partsregistry.org/wiki/index.php?title=Part:BBa_B0034 1-2M])</td><td>10</td><td>[http://partsregistry.org/wiki/index.php?title=Part:BBa_C0076 1-14D]</td><td>10</td></tr> | ||
| + | <tr><td>SpeI</td><td>1</td><td>XbaI</td><td>1</td><tr> | ||
| + | <tr><td>PstI</td><td>1</td><td>PstI</td><td>1</td><tr> | ||
| + | <tr><td> No.2 </td><td>2</td><td>No.2</td><td>2</td></tr> | ||
| + | <tr><td>dH<sub>2</sub>O</td><td>6</td><td>dH<sub>2</sub>O</td><td>8</td></tr> | ||
| + | <tr><td>total</td><td>20uL</td><td>total</td><td>20uL</td></tr> | ||
| + | </table> | ||
| + | |||
| - | + | ↓<br> | |
| - | + | 37°C, 2hr<br> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
↓<br> | ↓<br> | ||
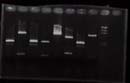
gel electrophoresis<br> | gel electrophoresis<br> | ||
| + | [[Image:Osaka090813.jpg]]<br> | ||
| + | No. 3 , 4 , 5 , 6 , ladder | ||
gel cut<br> | gel cut<br> | ||
↓<br> | ↓<br> | ||
| Line 118: | Line 97: | ||
total 50uL | total 50uL | ||
↓ | ↓ | ||
| - | + | 16°C overnight | |
| Line 127: | Line 106: | ||
3.1. EpsE + terminater<br> | 3.1. EpsE + terminater<br> | ||
Result of Nanodrop concentration check | Result of Nanodrop concentration check | ||
| - | |||
(sample No.) (concentration) | (sample No.) (concentration) | ||
2-A 21.5 ng/uL | 2-A 21.5 ng/uL | ||
| Line 136: | Line 114: | ||
digestion with restriction enzyme | digestion with restriction enzyme | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | + | <table border="1" frame="box"> | |
| - | + | <tr><td>2-A/2-B/2-C</td><td>8</td></tr> | |
| - | + | <tr><td>XbaI</td><td>1</td></tr> | |
| - | + | <tr><td>PstI</td><td>1</td></tr> | |
| - | + | <tr><td>No.2</td><td>2</td></tr> | |
| + | <tr><td>dH<sub>2</sub>O</td><td>8</td></tr> | ||
| + | <tr><td>total</td><td>20uL</td></tr> | ||
| + | </table> | ||
| - | |||
| - | |||
| - | 3.2. | + | <table border="1" frame="box"> |
| + | <tr><td>[http://partsregistry.org/wiki/index.php?title=Part:BBa_B0015 1-23L](vector)</td><td>8</td></tr> | ||
| + | <tr><td>XbaI</td><td>1</td></tr> | ||
| + | <tr><td>No.2</td><td>2</td></tr> | ||
| + | <tr><td>dH<sub>2</sub>O</td><td>9</td></tr> | ||
| + | <tr><td>total</td><td>20uL</td></tr> | ||
| + | </table> | ||
| + | |||
| + | ↓<br> | ||
| + | 37°C over night<br> | ||
| + | <br> | ||
| + | <br> | ||
| + | 3.2. rapid check <br> | ||
| + | [[Image:Osaka090813-2.jpg]] | ||
| + | (RBS + LasR([http://partsregistry.org/wiki/index.php?title=Part:BBa_C0179 2-8M])) | ||
No.3 and No.8 → to iquid medium(5mL + Amp 5uL) | No.3 and No.8 → to iquid medium(5mL + Amp 5uL) | ||
| + | |||
| + | |||
| + | [https://2009.igem.org/Team:Osaka/NOTES back to NOTES] | ||
Latest revision as of 07:52, 12 October 2009
1.before Min prep
Checked the cell cultures transformed and plated out yesterday (8/10) for colony formation and made an approximate count of the number of colonies.
(plate number)-(location on plate) (no. of colonies)
[http://partsregistry.org/wiki/index.php?title=Part:BBa_B0034 1-2M] 100~ (Amp)
[http://partsregistry.org/wiki/index.php?title=Part:BBa_F1610 2-24G] 1?
[http://partsregistry.org/wiki/index.php?title=Part:BBa_R0071 1-12C] 10
[http://partsregistry.org/wiki/index.php?title=Part:BBa_R0040 1-6I] 10<
[http://partsregistry.org/wiki/index.php?title=Part:BBa_C0171 2-8I] 10<
[http://partsregistry.org/wiki/index.php?title=Part:BBa_F2620 2-6E] 10<
[http://partsregistry.org/wiki/index.php?title=Part:BBa_R0078 1-16G] 10
[http://partsregistry.org/wiki/index.php?title=Part:BBa_R0079 1-12A] 10<
[http://partsregistry.org/wiki/index.php?title=Part:BBa_C0161 2-8O] 10<
[http://partsregistry.org/wiki/index.php?title=Part:BBa_I14015 2-11N] 10 (Kan)
[http://partsregistry.org/wiki/index.php?title=Part:BBa_I14017 1-18L] 50
inoculate to iquid medium(5mL + Amp 5uL or Kan 25uL)
after ligation
EpsE + terminator 3 → liquid medium
RBS + LasR([http://partsregistry.org/wiki/index.php?title=Part:BBa_C0179 2-8M]) 10 → rapid check
2.digestion and ligation
digestion with restriction enzyme
K204003
| Vector | Insert | ||
|---|---|---|---|
| RBS([http://partsregistry.org/wiki/index.php?title=Part:BBa_B0034 1-2M]) | 10 | cinR([http://partsregistry.org/wiki/index.php?title=Part:BBa_C0077 1-14F]) | 6 |
| SpeI | 1 | XbaI | 1 |
| PstI | 1 | PstI | 1 |
| No.2 Buffer | 2 | No.2 | 2 |
| dH2O | 6 | dH2O | 10 |
| total | 20uL | total | 20uL |
K204004
| Vector | Insert | ||
|---|---|---|---|
| RBS([http://partsregistry.org/wiki/index.php?title=Part:BBa_B0034 1-2M]) | 10 | LasR([http://partsregistry.org/wiki/index.php?title=Part:BBa_C0179 2-8M]) | 10 |
| EcoRI | 1 | EcoRI | 1 |
| XbaI | 1 | SpeI | 1 |
| No.2 | 2 | No.2 | 2 |
| dH2O | 4 | dH2O | 6 |
| total | 20uL | total | 20uL |
K204005
| Vector | Insert | ||
|---|---|---|---|
| [http://partsregistry.org/wiki/index.php?title=Part:BBa_I14017 1-18L] | 8 | [http://partsregistry.org/wiki/index.php?title=Part:BBa_I1466 1-23J] | 8 |
| SpeI | 1 | XbaI | 1 |
| PstI | 1 | PstI | 1 |
| No.2 | 2 | No.2 | 2 |
| dH2O | 8 | dH2O | 8 |
| total | 20uL | total | 20uL |
K204006
| Vector | Insert | ||
|---|---|---|---|
| RBS([http://partsregistry.org/wiki/index.php?title=Part:BBa_B0034 1-2M]) | 10 | [http://partsregistry.org/wiki/index.php?title=Part:BBa_C0076 1-14D] | 10 |
| SpeI | 1 | XbaI | 1 |
| PstI | 1 | PstI | 1 |
| No.2 | 2 | No.2 | 2 |
| dH2O | 6 | dH2O | 8 |
| total | 20uL | total | 20uL |
↓
37°C, 2hr
No. 3 , 4 , 5 , 6 , ladder
gel cut
↓
purification by [QIAquick Nucleotide Removal Kit]
↓
ligation
ligation DNA 44 10* buffer 5 ligation 1 total 50uL ↓ 16°C overnight
3.ligation check
3.1. EpsE + terminater
Result of Nanodrop concentration check
(sample No.) (concentration)
2-A 21.5 ng/uL
2-B 15.8 ng/uL
2-C 16.7 ng/uL
↓
digestion with restriction enzyme
| 2-A/2-B/2-C | 8 |
| XbaI | 1 |
| PstI | 1 |
| No.2 | 2 |
| dH2O | 8 |
| total | 20uL |
| [http://partsregistry.org/wiki/index.php?title=Part:BBa_B0015 1-23L](vector) | 8 |
| XbaI | 1 |
| No.2 | 2 |
| dH2O | 9 |
| total | 20uL |
↓
37°C over night
3.2. rapid check
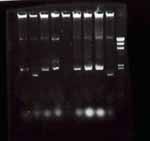
(RBS + LasR([http://partsregistry.org/wiki/index.php?title=Part:BBa_C0179 2-8M])) No.3 and No.8 → to iquid medium(5mL + Amp 5uL)
 "
"