Team:Aberdeen Scotland/WetLab/lacilatch
From 2009.igem.org
| (8 intermediate revisions not shown) | |||
| Line 6: | Line 6: | ||
The LacI latch module was designed to maintain the system in the ON state once it has been triggered, by repressing LacI production, and to induce timed cell lysis by repressing TetR production. These two functions enable production of our target proteins even after IPTG is no longer present, thus producing Elastin for a certain time period after a leak has been closed, allowing for extra strengthening of the weak spot. | The LacI latch module was designed to maintain the system in the ON state once it has been triggered, by repressing LacI production, and to induce timed cell lysis by repressing TetR production. These two functions enable production of our target proteins even after IPTG is no longer present, thus producing Elastin for a certain time period after a leak has been closed, allowing for extra strengthening of the weak spot. | ||
| - | [[Image: | + | [[Image:6DanAberdeenigem2009fixed.png|center|500px]] |
The LacI latch module consists of a promoter with a CI operator, a Tet Repressor gene and the LacI gene. The TetOperon and the Lysis Cassete which was developed by the 2008 Berkley iGEM team is then added to the module. | The LacI latch module consists of a promoter with a CI operator, a Tet Repressor gene and the LacI gene. The TetOperon and the Lysis Cassete which was developed by the 2008 Berkley iGEM team is then added to the module. | ||
| - | The autolysis mechanism is governed by the activity of bacteriophage encoded holin which induces the formation of hole in the bacterial cytoplasmic inner membrane. The holin pore complex facilitates the insertion of the endolysin, a soluble muralytic enzyme for disrupting the structure of the peptidoglycan in the outer membrane [4]. This tuned activity of holin and endolysin will deliver our glue component at the specific site following the induction of IPTG and quorum sensing. At the cellular level, the function of holin is inhibited by another protein called antiholin. The holin and antiholin are coded from the same coding region of the mRNA and hence generating a difference in only two amino acids [2]. A simple basis for this inhibitory function of antiholin could be the ionic interaction between the negatively charged inner surface of the cytoplasmic membrane and the positively charged lysine in the antiholin structure [1]. The literature [3] suggests that the presence of a thousand holin molecule is required for achieving a lysis timing of 40-45 minutes. This was tested and modelled by the UC Berkeley’08 iGEM team. In our system design, the antihloin is expressed under the control of a weak constitutive promoter so that holin production is able to take over antiholin produced at the time of lysis | + | The autolysis mechanism is governed by the activity of bacteriophage encoded holin which induces the formation of hole in the bacterial cytoplasmic inner membrane. The holin pore complex facilitates the insertion of the endolysin, a soluble muralytic enzyme for disrupting the structure of the peptidoglycan in the outer membrane [4]. This tuned activity of holin and endolysin will deliver our glue component at the specific site following the induction of IPTG and quorum sensing. At the cellular level, the function of holin is inhibited by another protein called antiholin. The holin and antiholin are coded from the same coding region of the mRNA and hence generating a difference in only two amino acids [2]. A simple basis for this inhibitory function of antiholin could be the ionic interaction between the negatively charged inner surface of the cytoplasmic membrane and the positively charged lysine in the antiholin structure [1]. The literature [3] suggests that the presence of a thousand holin molecule is required for achieving a lysis timing of 40-45 minutes. This was tested and modelled by the UC Berkeley’08 iGEM team. In our system design, the antihloin is expressed under the control of a weak constitutive promoter so that holin production is able to take over antiholin produced at the time of lysis. |
| - | An efficient autolysis mechanism | + | |
| + | An efficient autolysis and LacI latch mechanism are indispensable for ensuring the desirable time delay (longer than 40 minutes) required for enough glue (tropoelastain and lysyl oxidase ) production and their subsequent mixing . This will be achieved by the slow decay rate in the level of TetR production following the induction by inducer IPTG and the formation of antiholin which is an inhibitor of holin complex formation . | ||
==How it works:== | ==How it works:== | ||
| Line 16: | Line 17: | ||
| - | [[Image: | + | [[Image:7DanAberdeenigem2009fixed.png|center|500px]] |
The normal state of our system is illustrated in the picture above. LacI is expressed and binds to the Lac Operator thus repressing expression of our glue and the CI repressor. At the same time TetR is expressed and and blocks expression of Holin, and Endolysin. Note that Antiholin is not affected by this and is in fact expressed since it is under control of a separate constitutive promoter. | The normal state of our system is illustrated in the picture above. LacI is expressed and binds to the Lac Operator thus repressing expression of our glue and the CI repressor. At the same time TetR is expressed and and blocks expression of Holin, and Endolysin. Note that Antiholin is not affected by this and is in fact expressed since it is under control of a separate constitutive promoter. | ||
| - | [[Image: | + | [[Image:8DanAberdeenigem2009fixed.png|center|500px]] |
Once IPTG is induced our target Gene starts being expressed in addition to this the CI repressor gene can be expressed. | Once IPTG is induced our target Gene starts being expressed in addition to this the CI repressor gene can be expressed. | ||
| - | [[Image: | + | [[Image:9DanAberdeenigem2009fixed.png|center|500px]] |
| - | Once the CI repressor is synthesised it binds to the CI | + | Once the CI repressor is synthesised it binds to the CI operator, this halts expression of LacI and TetR. LacI and TetR levels then start to fall inside of the cell since no more molecules are being made. At this point TetR is still repressing Holin and Endolysin, which creates a time delay before Holin and Endolysin are expressed and the cell lysis. |
| - | [[Image: | + | [[Image:9bDanAberdeenigem2009fixed.png|center|500px]] |
| - | Once TetR and LacI have completely degraded, the system is latched on, and it will carry on expressing our product until it lysis | + | Once TetR and LacI have completely degraded, the system is latched on, and it will carry on expressing our product until it lysis. |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | == References == | |
| - | 3. Christos G. Savva, Jill S. Dewey, John Deaton, Rebecca L. White, Douglas K. Struck, Andreas Holzenburg and Ry Young. The holin of bacteriophage lambda forms rings with large diameter. Molecular Microbiology 69(4), 784–793. 2008. | + | [1]. Blasi, U., C.-Y. Chang, M. T. Zagotta, K. Nam, and R. Young. 1990. The lethal lambda S gene encodes its own inhibitor. EMBO J. 9:981–989. |
| - | 4. Young, R., I. N. Wang, and W. D. Roof. 2000. Phages will out: strategies of host cell lysis. Trends Microbiol. 8:120–128. | + | |
| - | 5. https://2008.igem.org/Team:UC_Berkeley/LysisDevice | + | [2]. Chang, C.-Y., K. Nam, U. Bla¨si, and R. Young. 1993. Synthesis of two bacteriophage lambda S proteins in an in vivo system. Gene 133:9–16. |
| + | |||
| + | [3]. Christos G. Savva, Jill S. Dewey, John Deaton, Rebecca L. White, Douglas K. Struck, Andreas Holzenburg and Ry Young. The holin of bacteriophage lambda forms rings with large diameter. Molecular Microbiology 69(4), 784–793. 2008. | ||
| + | |||
| + | [4]. Young, R., I. N. Wang, and W. D. Roof. 2000. Phages will out: strategies of host cell lysis. Trends Microbiol. 8:120–128. | ||
| + | |||
| + | [5]. https://2008.igem.org/Team:UC_Berkeley/LysisDevice | ||
| Line 47: | Line 51: | ||
<tr> | <tr> | ||
<td> | <td> | ||
| - | <a href=" | + | <a href="https://2009.igem.org/Team:Aberdeen_Scotland/WetLab/andgate/results"><img src="https://static.igem.org/mediawiki/2009/e/ed/Aberdeen_Left_arrow.png"> Back to AND Gate Results</a> |
</td> | </td> | ||
<td align="right"> | <td align="right"> | ||
| - | <a href=" | + | <a href="https://2009.igem.org/Team:Aberdeen_Scotland/WetLab/lacilatch/cloning">Continue to LacI-Latch Cloning Strategy <img src="https://static.igem.org/mediawiki/2009/4/4c/Aberdeen_Right_arrow.png"></a> |
</td> | </td> | ||
</tr> | </tr> | ||
Latest revision as of 21:41, 23 September 2009
University of Aberdeen - Pico Plumber
Introduction:
The LacI latch module was designed to maintain the system in the ON state once it has been triggered, by repressing LacI production, and to induce timed cell lysis by repressing TetR production. These two functions enable production of our target proteins even after IPTG is no longer present, thus producing Elastin for a certain time period after a leak has been closed, allowing for extra strengthening of the weak spot.
The LacI latch module consists of a promoter with a CI operator, a Tet Repressor gene and the LacI gene. The TetOperon and the Lysis Cassete which was developed by the 2008 Berkley iGEM team is then added to the module.
The autolysis mechanism is governed by the activity of bacteriophage encoded holin which induces the formation of hole in the bacterial cytoplasmic inner membrane. The holin pore complex facilitates the insertion of the endolysin, a soluble muralytic enzyme for disrupting the structure of the peptidoglycan in the outer membrane [4]. This tuned activity of holin and endolysin will deliver our glue component at the specific site following the induction of IPTG and quorum sensing. At the cellular level, the function of holin is inhibited by another protein called antiholin. The holin and antiholin are coded from the same coding region of the mRNA and hence generating a difference in only two amino acids [2]. A simple basis for this inhibitory function of antiholin could be the ionic interaction between the negatively charged inner surface of the cytoplasmic membrane and the positively charged lysine in the antiholin structure [1]. The literature [3] suggests that the presence of a thousand holin molecule is required for achieving a lysis timing of 40-45 minutes. This was tested and modelled by the UC Berkeley’08 iGEM team. In our system design, the antihloin is expressed under the control of a weak constitutive promoter so that holin production is able to take over antiholin produced at the time of lysis.
An efficient autolysis and LacI latch mechanism are indispensable for ensuring the desirable time delay (longer than 40 minutes) required for enough glue (tropoelastain and lysyl oxidase ) production and their subsequent mixing . This will be achieved by the slow decay rate in the level of TetR production following the induction by inducer IPTG and the formation of antiholin which is an inhibitor of holin complex formation .
How it works:
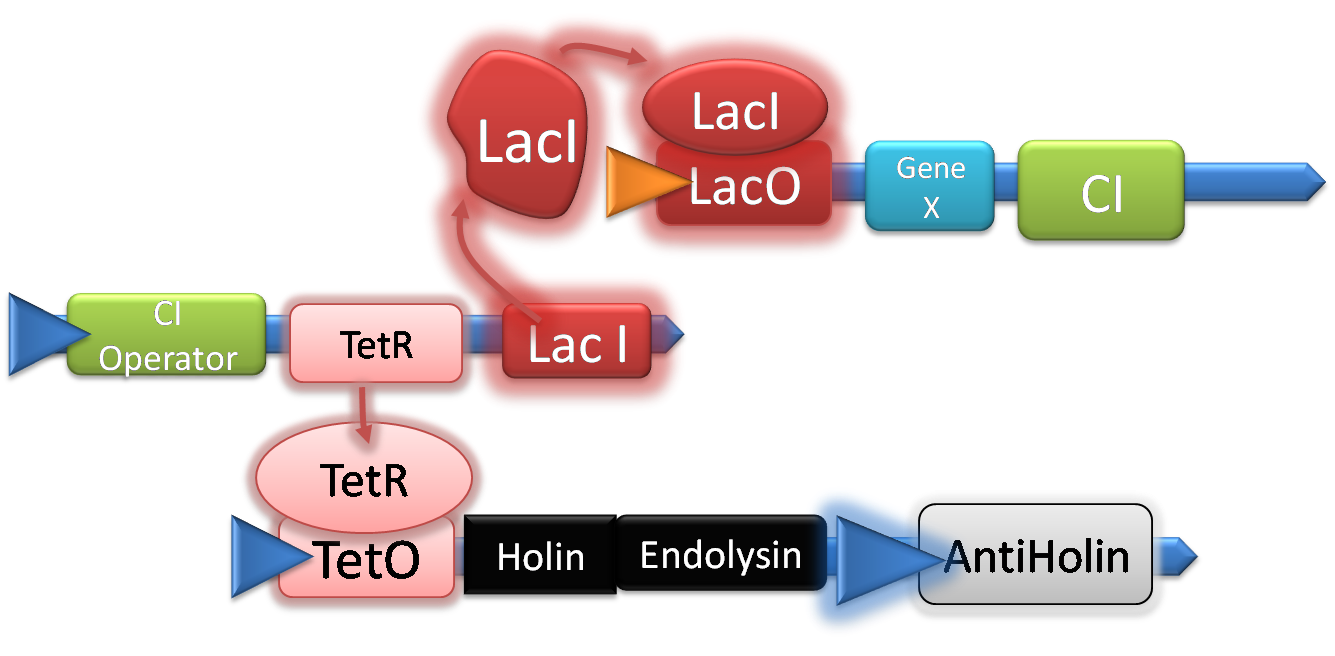
Please note that for the purpose of simplicity Quorum sensing is ignored in this section. The system would not turn on with just the IPTG stimuli.
The normal state of our system is illustrated in the picture above. LacI is expressed and binds to the Lac Operator thus repressing expression of our glue and the CI repressor. At the same time TetR is expressed and and blocks expression of Holin, and Endolysin. Note that Antiholin is not affected by this and is in fact expressed since it is under control of a separate constitutive promoter.
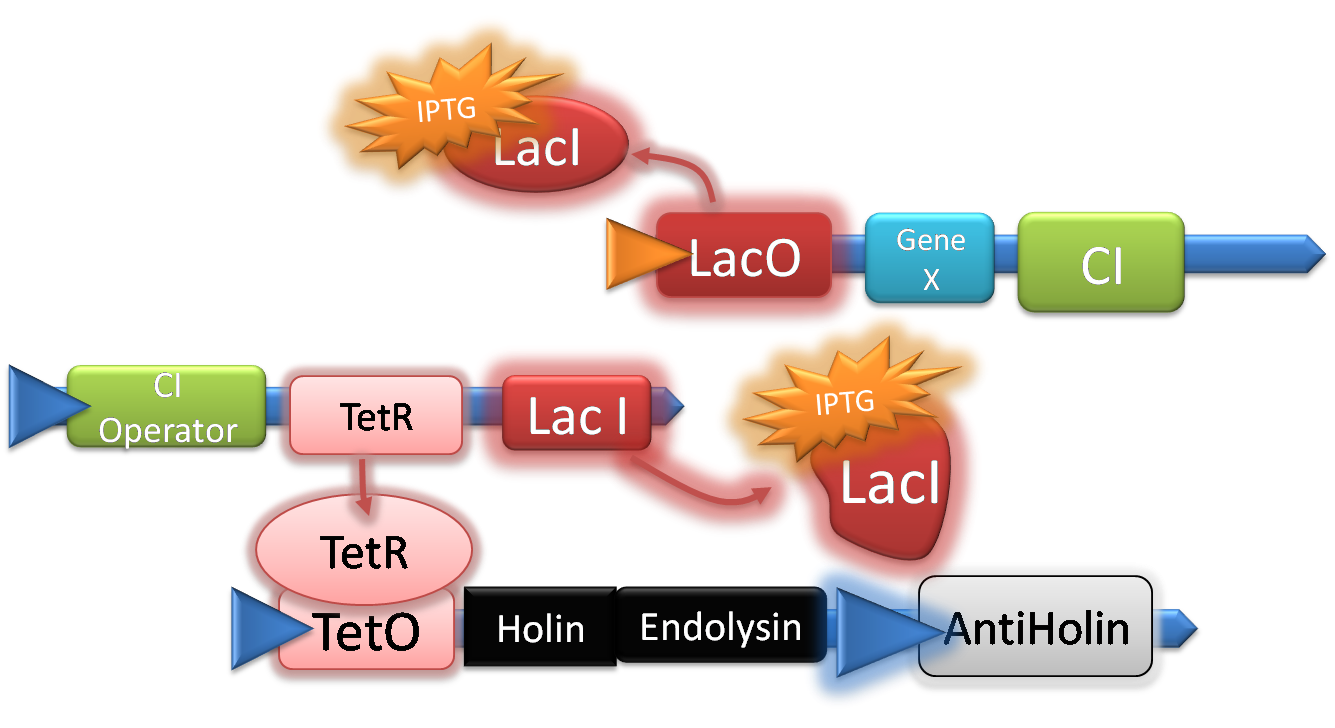
Once IPTG is induced our target Gene starts being expressed in addition to this the CI repressor gene can be expressed.
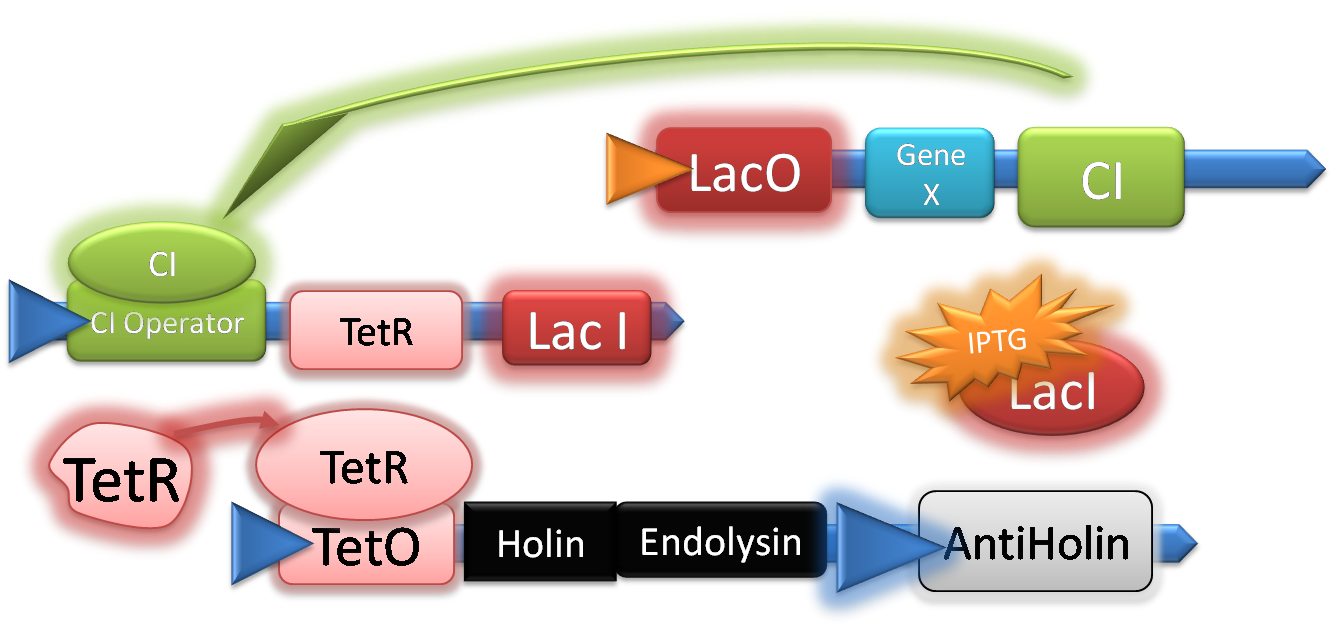
Once the CI repressor is synthesised it binds to the CI operator, this halts expression of LacI and TetR. LacI and TetR levels then start to fall inside of the cell since no more molecules are being made. At this point TetR is still repressing Holin and Endolysin, which creates a time delay before Holin and Endolysin are expressed and the cell lysis.
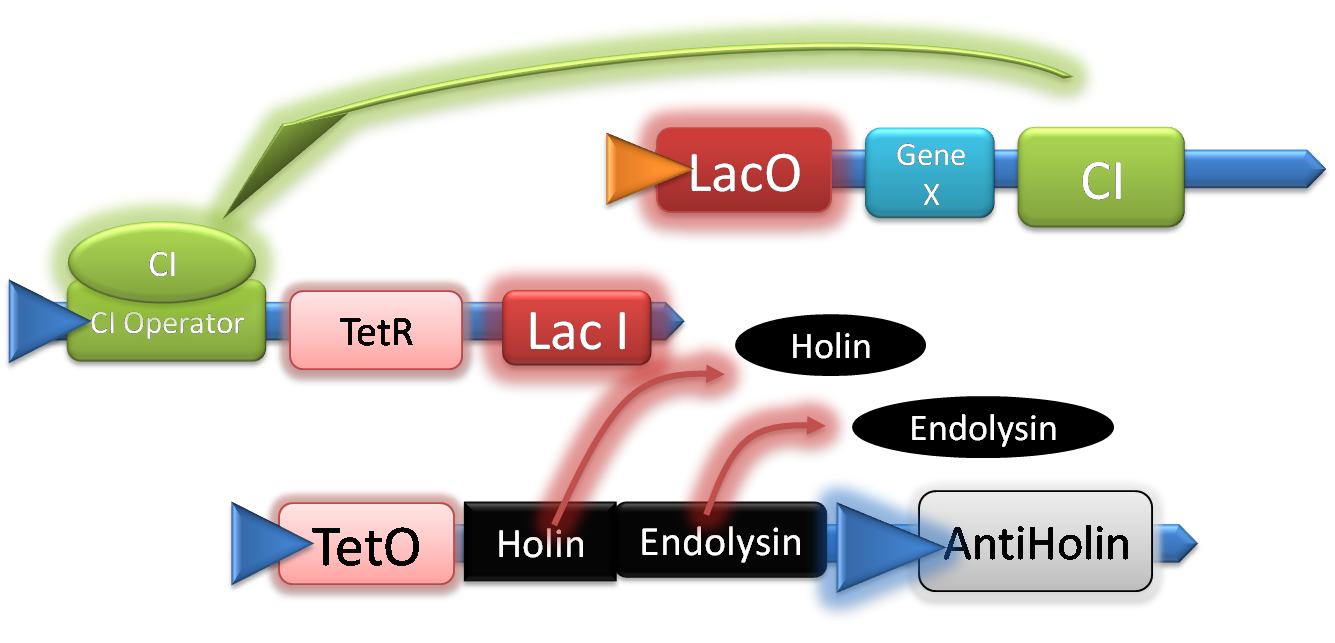
Once TetR and LacI have completely degraded, the system is latched on, and it will carry on expressing our product until it lysis.
References
[1]. Blasi, U., C.-Y. Chang, M. T. Zagotta, K. Nam, and R. Young. 1990. The lethal lambda S gene encodes its own inhibitor. EMBO J. 9:981–989.
[2]. Chang, C.-Y., K. Nam, U. Bla¨si, and R. Young. 1993. Synthesis of two bacteriophage lambda S proteins in an in vivo system. Gene 133:9–16.
[3]. Christos G. Savva, Jill S. Dewey, John Deaton, Rebecca L. White, Douglas K. Struck, Andreas Holzenburg and Ry Young. The holin of bacteriophage lambda forms rings with large diameter. Molecular Microbiology 69(4), 784–793. 2008.
[4]. Young, R., I. N. Wang, and W. D. Roof. 2000. Phages will out: strategies of host cell lysis. Trends Microbiol. 8:120–128.
[5]. https://2008.igem.org/Team:UC_Berkeley/LysisDevice
 Back to AND Gate Results Back to AND Gate Results
|
Continue to LacI-Latch Cloning Strategy 
|
 "
"