Team:HKU-HKBU/Speed Control Results
From 2009.igem.org
| Line 10: | Line 10: | ||
===Colony PCR=== | ===Colony PCR=== | ||
| + | |||
[[Image:HKU-HKBU_delta_cheZ.png|center|300px]] | [[Image:HKU-HKBU_delta_cheZ.png|center|300px]] | ||
| - | By using recombineering technology, cheZ could be precisely replaced by seletive resistance marker with homologous arms(about 50bp). To test the result, colonies were selected on the agar plate with chloramphenicol resistance followed by colony PCR test. In PCR, forward primers were designed in the upstream of cheZ locus in the chromosome and reverse primer was designed inside the chlromphenicol gene. The result of colony PCR test could examine whether the recombination procedure was successful or not. The expected DNA size of this test was about 400bp. The DNA agrose gel picture indicated the cheZ was knocked out both in ''YBE01'' and ''YBS01'' strains. | + | |
| + | |||
| + | By using recombineering technology, ''cheZ'' could be precisely replaced by seletive resistance marker with homologous arms(about 50bp). To test the result, colonies were selected on the agar plate with chloramphenicol resistance followed by colony PCR test. In PCR, forward primers were designed in the upstream of ''cheZ'' locus in the chromosome and reverse primer was designed inside the chlromphenicol gene. The result of colony PCR test could examine whether the recombination procedure was successful or not. The expected DNA size of this test was about 400bp. The DNA agrose gel picture indicated the cheZ was knocked out both in ''YBE01'' and ''YBS01'' strains. | ||
===Swimming Test=== | ===Swimming Test=== | ||
Based on the theory of speed control, the ''cheZ'' knockout strain could not swim at all. Therefore 3 percent half solid agar were used for testing the swimming ability. After obseveing swimming assay constantly for 8 hours, the patterns of ''YBE01'' and ''YBS01'' strains were formed as follows and the ''cheZ'' knockout strains showed that the ineffectiveness of the swimming ability of the two bacteria strains, which was an indication of the successful knockout of ''cheZ'' gene in ''YBE01'' and ''YBS01''. | Based on the theory of speed control, the ''cheZ'' knockout strain could not swim at all. Therefore 3 percent half solid agar were used for testing the swimming ability. After obseveing swimming assay constantly for 8 hours, the patterns of ''YBE01'' and ''YBS01'' strains were formed as follows and the ''cheZ'' knockout strains showed that the ineffectiveness of the swimming ability of the two bacteria strains, which was an indication of the successful knockout of ''cheZ'' gene in ''YBE01'' and ''YBS01''. | ||
| + | |||
| + | |||
[[Image:HKU-HKBU_speed_control_experiments_Fig2.png |center|500px]] | [[Image:HKU-HKBU_speed_control_experiments_Fig2.png |center|500px]] | ||
| + | |||
| + | |||
==Regulation of ''cheZ'' expression== | ==Regulation of ''cheZ'' expression== | ||
| Line 28: | Line 35: | ||
The samples were collected from differnt cultivation time and various IPTG concentrations. Obvious concentration gradient was observed with the induce time of 24 hours with different IPTG concentrations, which indicated that the inducer IPTG could control the expression level of ''cheZ''. | The samples were collected from differnt cultivation time and various IPTG concentrations. Obvious concentration gradient was observed with the induce time of 24 hours with different IPTG concentrations, which indicated that the inducer IPTG could control the expression level of ''cheZ''. | ||
| + | |||
| + | |||
[[Image:HKU-HKBU speed control western2.png |center|500px]] | [[Image:HKU-HKBU speed control western2.png |center|500px]] | ||
| + | |||
| + | |||
===Speed control === | ===Speed control === | ||
Revision as of 15:02, 20 October 2009
Contents |
Results
cheZ knockout detection in YBE01 and YBS01
cheZ knockout is a crutial step for speed control. We believed that by controlling the expression level of cheZ, we could manipulate the ratio of flagella rotating direction (clockwise & counterclockwise). Therefore, higher concentration of expressed CheZ could lead to faster swimming speed. This part played a key role as an "adapter" of bactomotor.
Colony PCR
By using recombineering technology, cheZ could be precisely replaced by seletive resistance marker with homologous arms(about 50bp). To test the result, colonies were selected on the agar plate with chloramphenicol resistance followed by colony PCR test. In PCR, forward primers were designed in the upstream of cheZ locus in the chromosome and reverse primer was designed inside the chlromphenicol gene. The result of colony PCR test could examine whether the recombination procedure was successful or not. The expected DNA size of this test was about 400bp. The DNA agrose gel picture indicated the cheZ was knocked out both in YBE01 and YBS01 strains.
Swimming Test
Based on the theory of speed control, the cheZ knockout strain could not swim at all. Therefore 3 percent half solid agar were used for testing the swimming ability. After obseveing swimming assay constantly for 8 hours, the patterns of YBE01 and YBS01 strains were formed as follows and the cheZ knockout strains showed that the ineffectiveness of the swimming ability of the two bacteria strains, which was an indication of the successful knockout of cheZ gene in YBE01 and YBS01.
Regulation of cheZ expression
For controlling the expression level of cheZ, a series of plasmids were constructed, such as plsmid plac/ara-his-cheZ-cm, which could be induced by IPTG or arabinose. This plasmid was transformed to YBS02 and YBE02. Different concentrations 0.001, 0.002, 0.004, 0.008, 0.012mmol/L of IPTG were added into the cultivating solution LB to induce cheZ expression.
Western Blotting
The samples were collected from differnt cultivation time and various IPTG concentrations. Obvious concentration gradient was observed with the induce time of 24 hours with different IPTG concentrations, which indicated that the inducer IPTG could control the expression level of cheZ.
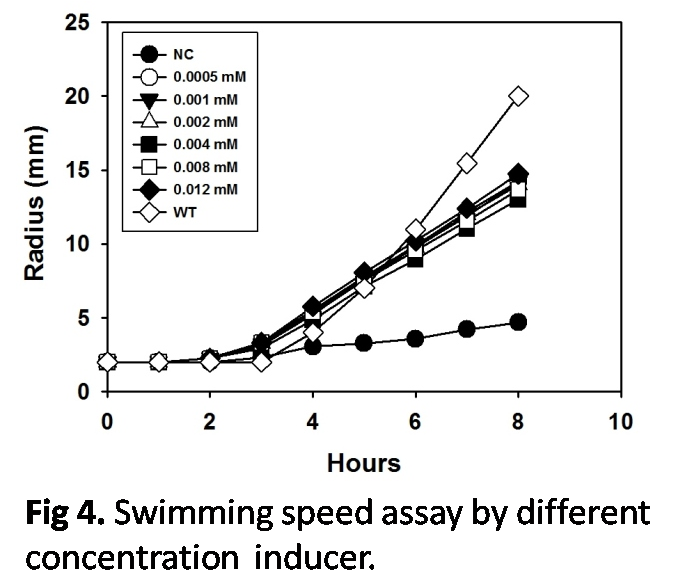
Speed control
The speed was detected by the swimming assay. The swimming radius was measured each hour continously for 8 hours. The results indicated that without IPTG inducer, the bacteria only tumbled; and when IPTG was added into cultivation LB broth, the bacteria were started and swam. However, similar speed bacteria with differnt IPTG concentration indicated that the alternation of IPTG concentrations was not an effective speed control method. Although it was hard to manipulate the speed via this method, the turn-on and turn-off of swimming could be achieved.
 "
"