Team:IIT Madras/Experiments
From 2009.igem.org
Ramakrishna (Talk | contribs) (→Comparing the differences in the growth rates of cells with and without plasmids in variuos media) |
Ramakrishna (Talk | contribs) (→Comparing the differences in the growth rates of cells with and without plasmids in various media) |
||
| Line 40: | Line 40: | ||
[[Image:growth curves.jpg|700px]] | [[Image:growth curves.jpg|700px]] | ||
| - | This experiment helps in comparing the growth rates of various strains in different antibiotic media. First we inoculate a colony from the plate containing the required strain into its corresponding media (the media with the required antibiotic). That is, DH5a will be inoculated into LB without any antibiotic, RFP (in pSB1C3) will be inoculated into LB containing Chloramphenicol (Chl), CFP (in pSB1A2) will be inoculated into LB containing Ampicillin (Amp) and RFP-CFP ccotransformed cells into LB containing both the antibiotics. These are then grown for about 4 hours or till they reach an OD600 value between 0.1 to 0.5. | + | |
| + | This experiment helps in comparing the growth rates of various strains in different antibiotic media. | ||
| + | |||
| + | First we inoculate a colony from the plate containing the required strain into its corresponding media (the media with the required antibiotic). That is, DH5a will be inoculated into LB without any antibiotic, RFP (in pSB1C3) will be inoculated into LB containing Chloramphenicol (Chl), CFP (in pSB1A2) will be inoculated into LB containing Ampicillin (Amp) and RFP-CFP ccotransformed cells into LB containing both the antibiotics. These are then grown for about 4 hours or till they reach an OD600 value between 0.1 to 0.5. | ||
Then the culture of each strain is taken and inoculted into each of the 5ml broths which contains no antibiotic, Amp, Chl and Amp-Chl so that the OD value is 0.01. So we have an array of 16 tubes with various possible combinations: | Then the culture of each strain is taken and inoculted into each of the 5ml broths which contains no antibiotic, Amp, Chl and Amp-Chl so that the OD value is 0.01. So we have an array of 16 tubes with various possible combinations: | ||
* DH5a in no antibiotic, DH5a in Amp, DH5a in Chl and Dh5a in Amp-Chl | * DH5a in no antibiotic, DH5a in Amp, DH5a in Chl and Dh5a in Amp-Chl | ||
| Line 55: | Line 58: | ||
# This inoculum is allowed to grow for about 4 hours or till the OD600 of each sample crosses 0.1. | # This inoculum is allowed to grow for about 4 hours or till the OD600 of each sample crosses 0.1. | ||
# Then we use OD 1 x Vol 1 = OD 2 x Vol 2 to measure how much to inoculte from this 4 hour culture to each of the fresh 5ml LB medium with different antibiotic combinations so that the starting OD is 0.01. In this case, OD 1 is the OD600 of the 4 hour culture, Vol 1 is the volume of this 4 hour culture that needs to be inoculted in to the fresh 5ml culture, OD 2 is 0.01 (starting OD for all the 5 ml cultures) and the Vol 2 is (5ml + vol 1). | # Then we use OD 1 x Vol 1 = OD 2 x Vol 2 to measure how much to inoculte from this 4 hour culture to each of the fresh 5ml LB medium with different antibiotic combinations so that the starting OD is 0.01. In this case, OD 1 is the OD600 of the 4 hour culture, Vol 1 is the volume of this 4 hour culture that needs to be inoculted in to the fresh 5ml culture, OD 2 is 0.01 (starting OD for all the 5 ml cultures) and the Vol 2 is (5ml + vol 1). | ||
| - | # | + | # From this freshly inoculated sample, 150ul of the culture is used to measure the OD every hour starting from the point if inoculation. The 150ul of the sample is diluted 5 times to 750ul and then the OD600 is measured. |
===Modeling=== | ===Modeling=== | ||
Revision as of 06:43, 21 October 2009
Contents |
Experiments
Comparing the differences in the growth rates of cells with and without plasmids in various media
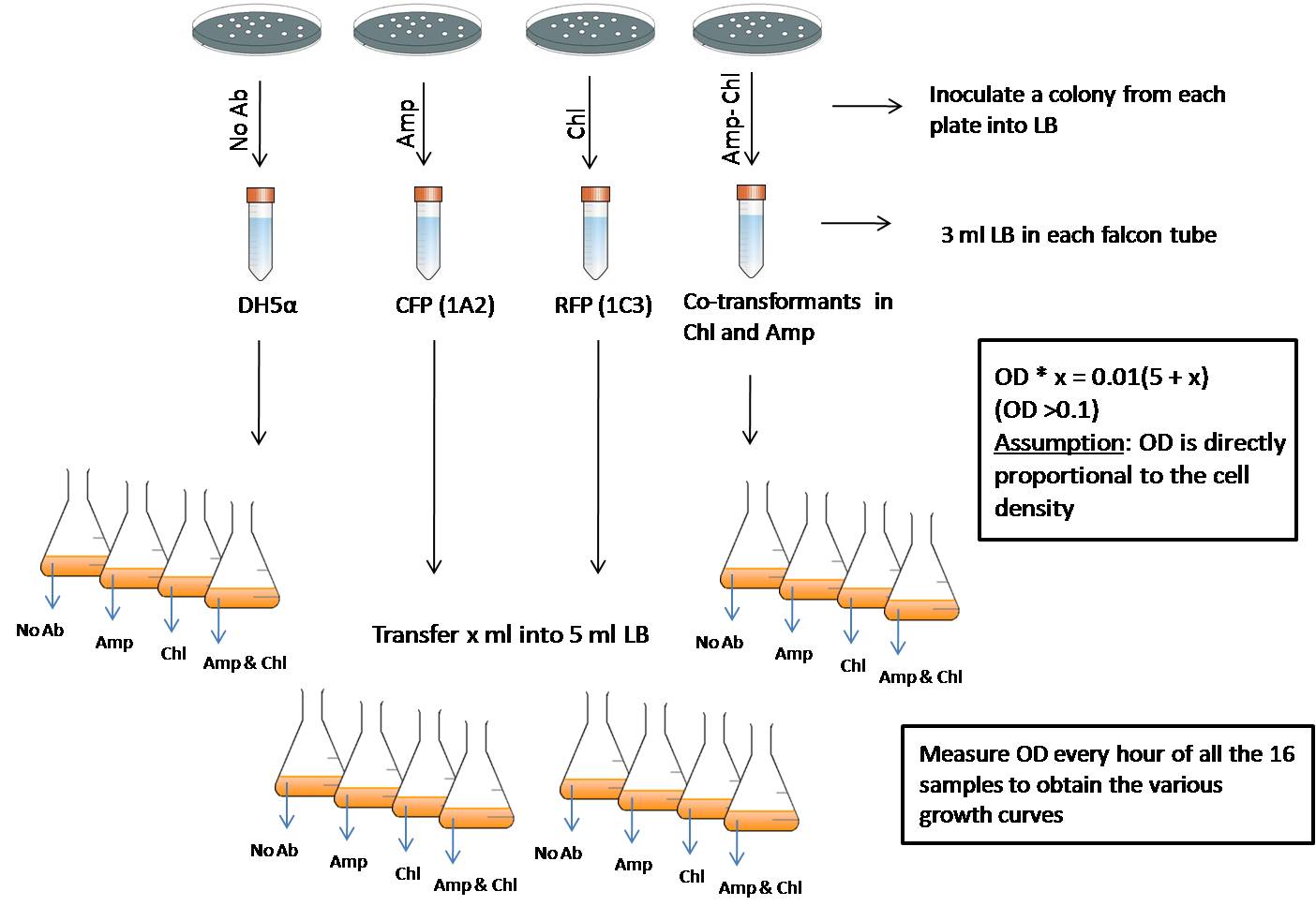
This experiment helps in comparing the growth rates of various strains in different antibiotic media.
First we inoculate a colony from the plate containing the required strain into its corresponding media (the media with the required antibiotic). That is, DH5a will be inoculated into LB without any antibiotic, RFP (in pSB1C3) will be inoculated into LB containing Chloramphenicol (Chl), CFP (in pSB1A2) will be inoculated into LB containing Ampicillin (Amp) and RFP-CFP ccotransformed cells into LB containing both the antibiotics. These are then grown for about 4 hours or till they reach an OD600 value between 0.1 to 0.5. Then the culture of each strain is taken and inoculted into each of the 5ml broths which contains no antibiotic, Amp, Chl and Amp-Chl so that the OD value is 0.01. So we have an array of 16 tubes with various possible combinations:
- DH5a in no antibiotic, DH5a in Amp, DH5a in Chl and Dh5a in Amp-Chl
- RFP (pSB1C3) in no antibiotic, RFP (pSB1C3)in Amp, RFP (pSB1C3)in Chl and RFP (pSB1C3)in Amp-Chl
- CFP (pSB1A2) in no antibiotic, CFP (pSB1A2)in Amp, CFP (pSB1A2)in Chl and CFP (pSB1A2)in Amp-Chl
- RFP (1C3)-CFP (1A2) in no antibiotic, RFP (1C3)-CFP (1A2) in Amp, RFP (1C3)-CFP (1A2) in Chl and RFP (1C3)-CFP (1A2)in Amp-Chl
Thus the starting point for all the samples is the same - an OD600 of 0.01. Every hour starting from the point of inoculation, the OD of all the 16 samples is measured. This will give a fair idea of the growth rates of variuos strains in different antibiotic media
Experimental protocol:
- A colony of DH5a is inoculated into 3ml LB without any antibiotic in a 50ml tarson tube. Similarly, RFP (1C3) colony is inoculated into 3ml LB with Chl, CFP (1A2) colony into 3ml LB with Amp and RFP-CFP colony into 3ml LB with Amp-Chl.
- This inoculum is allowed to grow for about 4 hours or till the OD600 of each sample crosses 0.1.
- Then we use OD 1 x Vol 1 = OD 2 x Vol 2 to measure how much to inoculte from this 4 hour culture to each of the fresh 5ml LB medium with different antibiotic combinations so that the starting OD is 0.01. In this case, OD 1 is the OD600 of the 4 hour culture, Vol 1 is the volume of this 4 hour culture that needs to be inoculted in to the fresh 5ml culture, OD 2 is 0.01 (starting OD for all the 5 ml cultures) and the Vol 2 is (5ml + vol 1).
- From this freshly inoculated sample, 150ul of the culture is used to measure the OD every hour starting from the point if inoculation. The 150ul of the sample is diluted 5 times to 750ul and then the OD600 is measured.
Modeling
Fluorescent Imaging
 "
"