Team:UNIPV-Pavia/Project/Motivation
From 2009.igem.org
(l) |
(l) |
||
| Line 88: | Line 88: | ||
Molecular formula C<sub>2</sub>H<sub>6</sub>O | Molecular formula C<sub>2</sub>H<sub>6</sub>O | ||
| + | |||
{|align="center"|[[Image:Ethanol.JPG|thumb|600px|]]|} | {|align="center"|[[Image:Ethanol.JPG|thumb|600px|]]|} | ||
Revision as of 10:53, 21 October 2009

|
|
|
Motivation |
|||
|
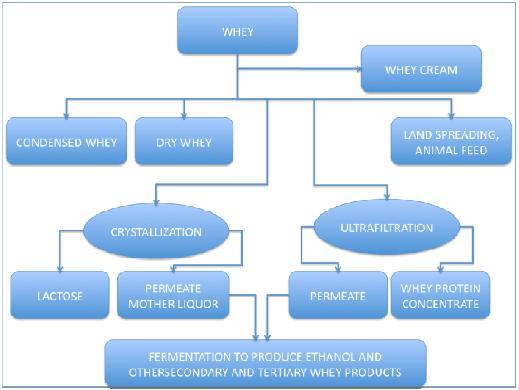
WHY WHEY?Whey is a component of milk, which separates after curdling when rennet or an edible acidic substance are added. It represents one of the main by-products of the cheese and casein industry. It is a mixture of different substances, some of which potentially valuable if extracted and individually processed. As reported in the following table, the main solid components of whey are soluble proteins, lactose, mineral salts and vitamins. In this section a summary of "Ethanol? Whey not!" project results is reported. For more information you can visit Parts Characterization section, in which you can find a documentation for each every single BioBrick involved in this project. This liquid waste is commonly regarded as the "environmental problem" of the cheese factories, which generally produce wheys of different content depending on the specific productive process and kind of cheese. In Italy, as in other countries with an important casearian tradition, the amount of whey produced every year is huge (8-10 million m^3/year). Differently from other countries in the central/nord Europe, in Italy the 85% of the milk produced is transformed in cheese by an high number of small/medium creameries, spread on the whole territory. Until some year ago, whey was given to pigs or introduced directly into the river systems, thus contributing to the organic pollution of environment and giving rise to water asphyxia. Its high nutrient load, in fact, causes the proliferation of microorganisms which deplete oxygen levels in the surface water systems. For this reason, the Italian law classifies whey as "special waste". Two indicators are commonly used to measure the nutrient load: B.O.D.5 and C.O.D..
B.O.D. 5 (Biochemical Oxigen Domand 5) It is an index to measure the oxygen need within 5 days (in mg/l), for the oxidation processes of the organic substances present in waste performed by microorganisms. C.O.D. (Chemical Oxygen Demand) It is a measure of the amount of oxygen used to oxidize organic and inorganic compounds contained in a water sample. It provides a global measurement of all chemicals in the water that can be oxidized and so it is an indirect index of pollutants in a water sample.
Nowadays, it is well known that the management of the whey disposal is expensive and that whey is considered as a troublesome reject substance.
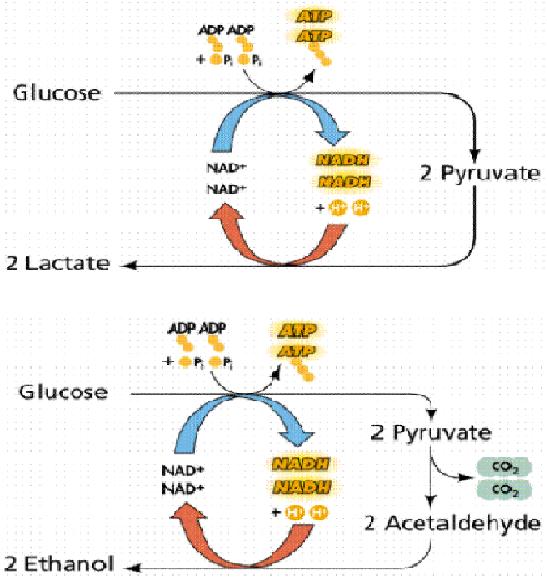
FermentationFermentation is an anaerobic biochemical process, in which an organic compound is reduced to ethanol or lactic acid (typically) as final products in order to produce NAD+, a coenzyme involved in the standard energy-producing pathway, that is glicolysis. Fermentation is wide-spread in living beings: it is the only energy production mechanism coupled to glicolysis in prokaryotes and is also present in eukaryotes were it is an alternative to the more efficient oxidative phosphorilation. The starting compound in the fermentation pathway is pyruvate, which is usually produced from sugar substrates, such as glucose and lactose through the glicolysis pathway. In lactic fermentation, pyruvate is directly transformed in lactate by the enzyme Lactate Dehydrogenase. In alcoholic fermentation, pyruvate is first transformed in acetaldehyde by pyruvate decarboxylase, with the production of a carbon dioxide molecule and then in ethanol by the enzyme alcohol dehydrogenase. From a molecule of glucose, 2 molecules of ATP, the "molecular unit" of energy inside the cells, are produced. Fermentation is exploited industrially in a wide range of preparations: bread and wine-making, beer brewing, cheese and yogurt production. Lactose transformation into ethanol is a process that involves both glycolisis and alcoholic fermentation. Since glicolysis starting point is a molecule of glucose, lactose molecules have to be firstly broken into the two monosaccharide units, glucose and β-galactose. The latter sugar is then converted into glucose by the action of three different enzymes, known as the Leloir pathway. The two molecules of glucose are then oxidized in the glicolysis pathway, in which we have the production of a total of 4 ATP molecules and 4 molecules of pyruvate per lactose molecule. The pyruvate is then reduced into ethanol and carbon dioxide with a 1:1 stochiometry. EthanolMolecular formula C2H6O | ||||
 "
"