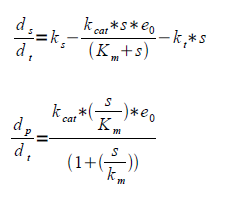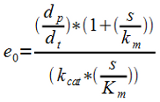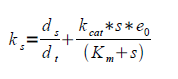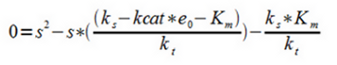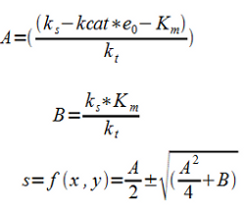Team:Uppsala-Sweden/Modelling
From 2009.igem.org
| Line 14: | Line 14: | ||
[[Image:ek_0.png]] | [[Image:ek_0.png]] | ||
| - | Where e is the amount of free enzyme and es is the amount of enzyme bound to substrate. | + | Where ''e'' is the amount of free enzyme and ''es'' is the amount of enzyme bound to substrate. |
[[Image:ek_1_2.png]] | [[Image:ek_1_2.png]] | ||
| - | Where ks is the rate of substrate formation (flux of the glycolysis), kcat is the maximum number of enzymatic reactions catalyzed per second, kt is the rate constant loss of substrate to other pathways, in our case primarily to the Krebbs cycle, Km is the substrate concentration at half the maximum reaction rate (Vmax) and s is the substrate concentration. | + | Where ''ks'' is the rate of substrate formation (flux of the glycolysis), ''kcat'' is the maximum number of enzymatic reactions catalyzed per second, ''kt'' is the rate constant loss of substrate to other pathways, in our case primarily to the Krebbs cycle, ''Km'' is the substrate concentration at half the maximum reaction rate (''Vmax'') and ''s'' is the substrate concentration. |
| - | Having the Km and Kcat values gives e0 from the level of product formation. | + | Having the ''Km'' and ''Kcat'' values gives ''e0'' from the level of product formation. |
[[Image:ek_3.png]] | [[Image:ek_3.png]] | ||
| - | 3. Having e0 and letting kt go towards zero gives ks. | + | 3. Having ''e0'' and letting ''kt'' go towards zero gives ''ks''. ''kt'' can be assumed to approach zero provided that the Krebbs cycle is the main native consumer of pyruvate and that the inhibition of the PDC is successful. |
[[Image:ek_4.png]] | [[Image:ek_4.png]] | ||
| - | 4. Assuming Ks to be constant within the interval and assuming steady-state | + | 4. Assuming ''Ks'' to be constant within the interval and assuming steady-state |
[[Image:ek_5.png]] | [[Image:ek_5.png]] | ||
Revision as of 13:33, 21 October 2009

Modelling of Advanced Ethanol Production
For our modell we have some basic assumptions that are
1. Co-evolution of the of the ethanol producing pathway, making km and kcat being highly similar for pdc as well as ADH. This assumption makes it possible for us to model the system as a single step reaction from pyruvate to ethanol. Greatly increasing the ease of simulation.
2. Assuming quasi steady state (Michaelis Menten Kinetics), that is constant total enzyme concentration. This is reasonable during log-phase before ethanol starts to inhibit growth.
Where e is the amount of free enzyme and es is the amount of enzyme bound to substrate.
Where ks is the rate of substrate formation (flux of the glycolysis), kcat is the maximum number of enzymatic reactions catalyzed per second, kt is the rate constant loss of substrate to other pathways, in our case primarily to the Krebbs cycle, Km is the substrate concentration at half the maximum reaction rate (Vmax) and s is the substrate concentration.
Having the Km and Kcat values gives e0 from the level of product formation.
3. Having e0 and letting kt go towards zero gives ks. kt can be assumed to approach zero provided that the Krebbs cycle is the main native consumer of pyruvate and that the inhibition of the PDC is successful.
4. Assuming Ks to be constant within the interval and assuming steady-state
S is a function of f(x,y) where x is proportional to e0 and y is proportional to kt
Measure for different x and y and fit the function
 "
"