Team:HKU-HKBU/Polar Expression Results
From 2009.igem.org
(→Fluorescence Microscopy) |
(→Fluorescence Microscopy) |
||
| Line 23: | Line 23: | ||
===Fluorescence Microscopy=== | ===Fluorescence Microscopy=== | ||
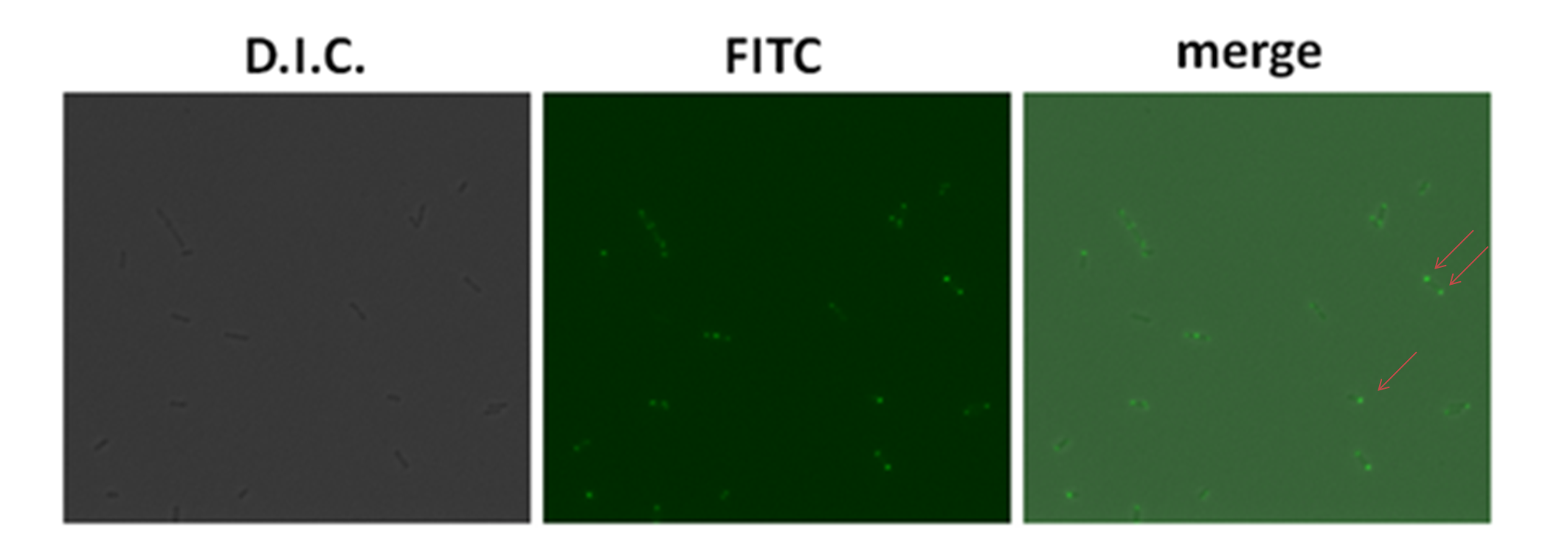
| - | After literature review, we found the ''E.coli BL21'' was also LPS complete strains. Therefore, we used the plasmid ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K283001 '''BBa_K283001'''] ) containing T7 promoter which controlled the expression of '''GFP-Strp-AIDA'''. ''BL21'' strain contained T7 polymerase, therefore when the IPTG was added into the culture medium, '''GFP-Strp-AIDA''' would be strongly induced. In the figure below, it showed the expression of '''GFP'''. This expression was observed under the fluorescent microscope using oil lens with the manification of 600 times. In the merge picture of dark field and FITC field, the fluorescent proteins were showed at one end of the bacteria. | + | After literature review, we found the ''E.coli BL21'' was also LPS complete strains. Therefore, we used the plasmid ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K283001 '''BBa_K283001''']) containing T7 promoter which controlled the expression of '''GFP-Strp-AIDA'''. ''BL21'' strain also contained T7 polymerase, therefore when the IPTG was added into the culture medium, '''GFP-Strp-AIDA''' would be strongly induced. In the figure below, it showed the expression of '''GFP'''. This expression was observed under the fluorescent microscope using oil lens with the manification of 600 times. In the merge picture of dark field and FITC field, the fluorescent proteins were showed at one end of the bacteria. |
Revision as of 13:41, 21 October 2009
Contents |
Strain Selecion
Swimming Ability Assay
The purpose of this part is to find one or several strains of bacteria which could swim. Therefore, we tested bacteria of Escherichia coli and Salmonella typhimurium. The swimming plates of BL21, NCM3722 and MG3 yielded negative results. We found a Salmonella typhimurium strain - YBS01 is a bacteria with high swimming abilities (~4.5 mm/h), and a strain of Escherichia coli - YBE01 showed a stronger swim ability, with a speed of approximate 5.5 mm increase in radius every hour at the end of eight-hour-experiment.
LPS Completeness Search
LPS takes vital part in AIDA polar expression system. Due to many mutations may exist in the strains of Escherichia coli or Salmonella typhimurium, some of these mutations may cause the defciency in there LPS layers. There mutants could survive in the culture. But for AIDA expression, when the host's LPS is incomplete, the AIDA will express all over the bacteria surface; while the host's LPS is complete, the AIDA will express only on one side of the bacteria. After literature review, E. coli-YBE01 [1] and S.typhimurium-YBS01 [2] are identified to possess complete LPS layer. Their ability to express desirable proteins on the head is examined in later experiment.
Polar Expression
AIDA Polar Expression System
Fluorescence Microscopy
After literature review, we found the E.coli BL21 was also LPS complete strains. Therefore, we used the plasmid ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K283001 BBa_K283001]) containing T7 promoter which controlled the expression of GFP-Strp-AIDA. BL21 strain also contained T7 polymerase, therefore when the IPTG was added into the culture medium, GFP-Strp-AIDA would be strongly induced. In the figure below, it showed the expression of GFP. This expression was observed under the fluorescent microscope using oil lens with the manification of 600 times. In the merge picture of dark field and FITC field, the fluorescent proteins were showed at one end of the bacteria.
For this project, we required a strain that expresses specific proteins in only one side, and has swimming ability at the same time. However, the BL21 strain couldn't swim. Therefore we need to apply one side expression system into our new candidate YBE01. While YBE01 didn't contain T7 polymerase, being not able to activate T7 promoter in this plasmid. Therefore, a new plasmid with strongly inducible or constitutive expression system should be constructed. Attempts were made to use pcI857 as thermo-inducible promoter which was turn-on at 37 celsius degree and turn-off at 30 celsius degree. We also tried plac without lacI as a constitutive promoter. However, only weak and unstable expression of GFP could be observed. One of the possible explanations could be that these two systems were not strong or stable enough to ensure the consistent expression of our required proteins.
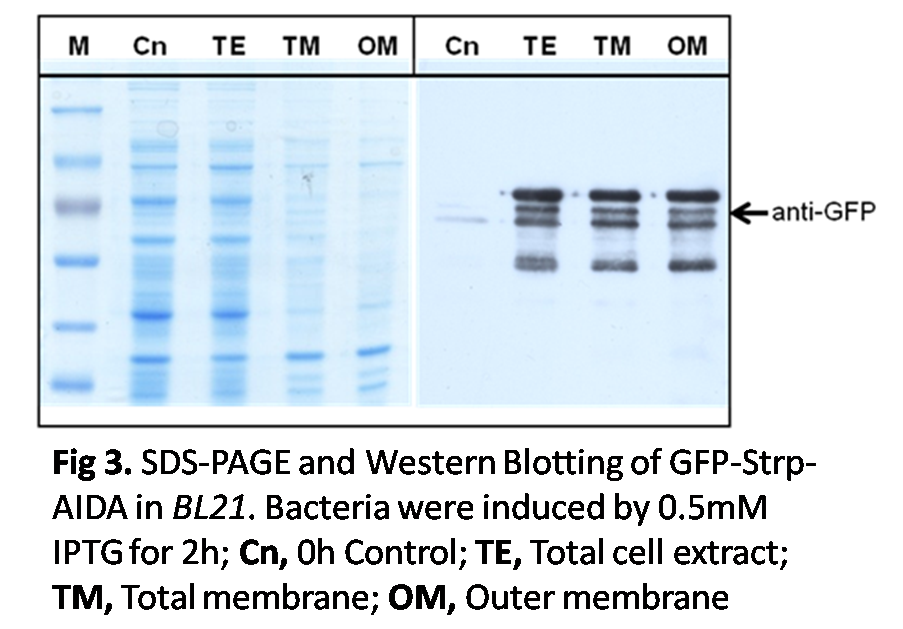
SDS PAGE and Western Blotting
The surface expression of streptavidin was essential to specifically bind to biotin-coated motor. To prove that the GFP-streptavidin was expressed on the outer membrane of the bacteria through AIDA system, different contents of the bacteria were separated by ultracentrifuge. After separation, the outer membrane, inner membrane, total membrane and cytoplasm were collected as different samples. SDS PAGE and Western Blotting were conducted for verification. Each sample was quantified by BCA assay to make sure the equal loading of proteins. The western blotting results showed that most GFP proteins were localized on the outer membrane of the bacteria, which corresponded with the expected result that the specific expressions of proteins were on the outer membrane.
Observing the moving strains in the microscope, we found that all the GFP were expressed at the forehead end of the rod shaped bacteria. Therefore, this system was a perfect choice for binding with biotin-coated motor. It can express streptavidin at pole(s), which allows cells to adhere in the same orientation to a microrotatory motor through biotin-streptavidin interaction.
Lpp-OmpA System
Fluorescent Microscope
For AIDA system, the requirements for strains were critical, such as LPS completeness, strong activators, etc. Another polar expression system was constructed as our back-up plan. Lpp consists of 9 amino acids of signal peptide for targeting the inner membrane of the bacteria. OmpA (91-261) is a 5-cross trans-membrane protein which could bring cargos to the outer membrane of the bacteria. We constructed a plasmid by fusing GFP-strp with Lpp-OmpA system.
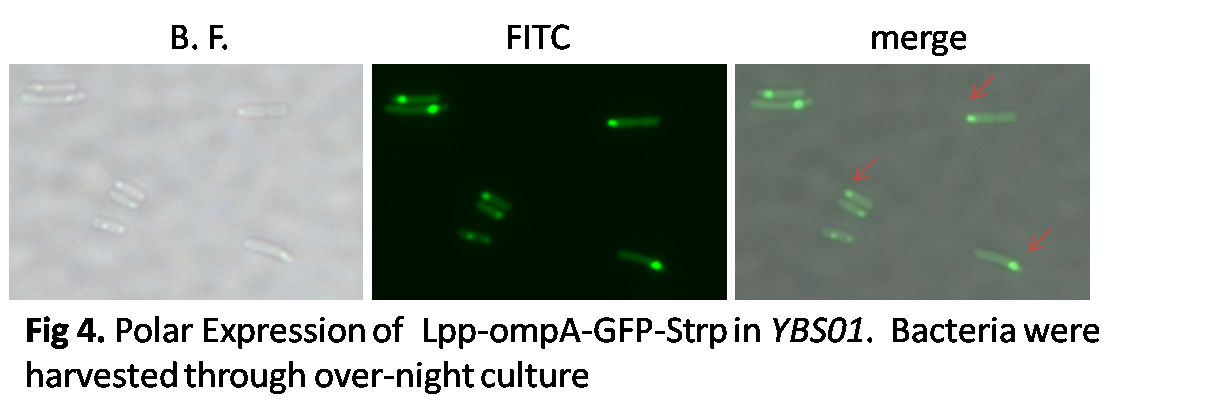
The surface expression of Lpp-ompA-GFP-Strp was consistent, however the polar expression of this plasmid couldn't be achieved. Therefore, this plasmid was transformed into various strains of E.coli and Salmonella. We found that in the stage phase of YBS01, polar expression of the plamid was observed.
The left panel showed the polar expression of the plasmid in the stationary phase of YBS01 as a negative control. The right panel showed the merge picture of the bright field and FITC field under the fluorescent microscope with a magnification of 600 times. The result indicated that polar expression of proteins was achieved by this system.
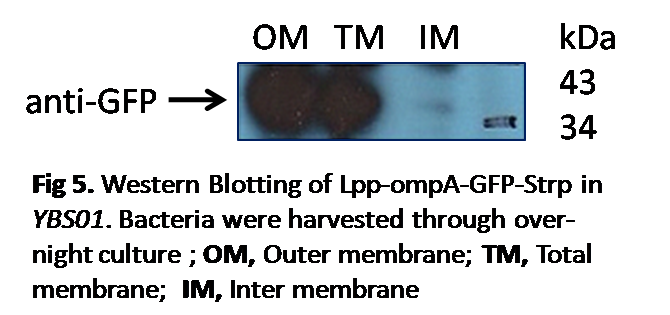
Western Blotting
To verify our experimental result in a more convincing way, Western Blotting was conducted, which could reflect the surface expression of proteins. By studying the relative concentrations of expressed protein GFP in different samples, an obvious darker band was observed in the outer membrane samples. This results verified our hypothesis that the GFP proteins are particularly expressed on the outer membrane.
Reference
- Sumita Jain, Peter van Ulsen, Inga Benz, M. Alexander Schmidt, Rachel Fernandez, Jan Tommassen, and Marcia B. Goldberg, Polar Localization of the Autotransporter Family of Large Bacterial Virulence Proteins, Journal of Bacteriology, July 2006, p. 4841-4850, Vol. 188, No. 13
- Maurien M. A. Olsthoorn, Bent O. Petersen, Siegfried Schlecht, Johan Haverkamp, Klaus Bock, Jane E. Thomas-Oates and Otto Holst, Identification of a Novel Core Type in Salmonella Lipopolysaccharide, The Journal of Biological Chemistry, Vol. 273, No. 7, Issue of February 13, pp. 3817-3829, 1998
 "
"