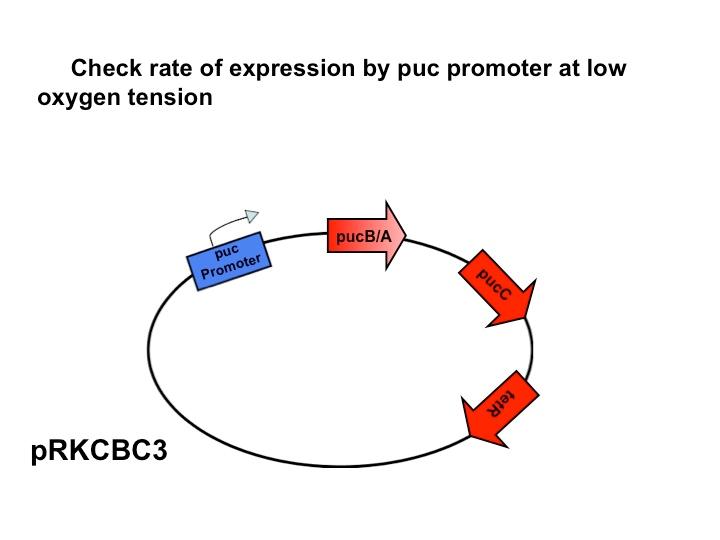
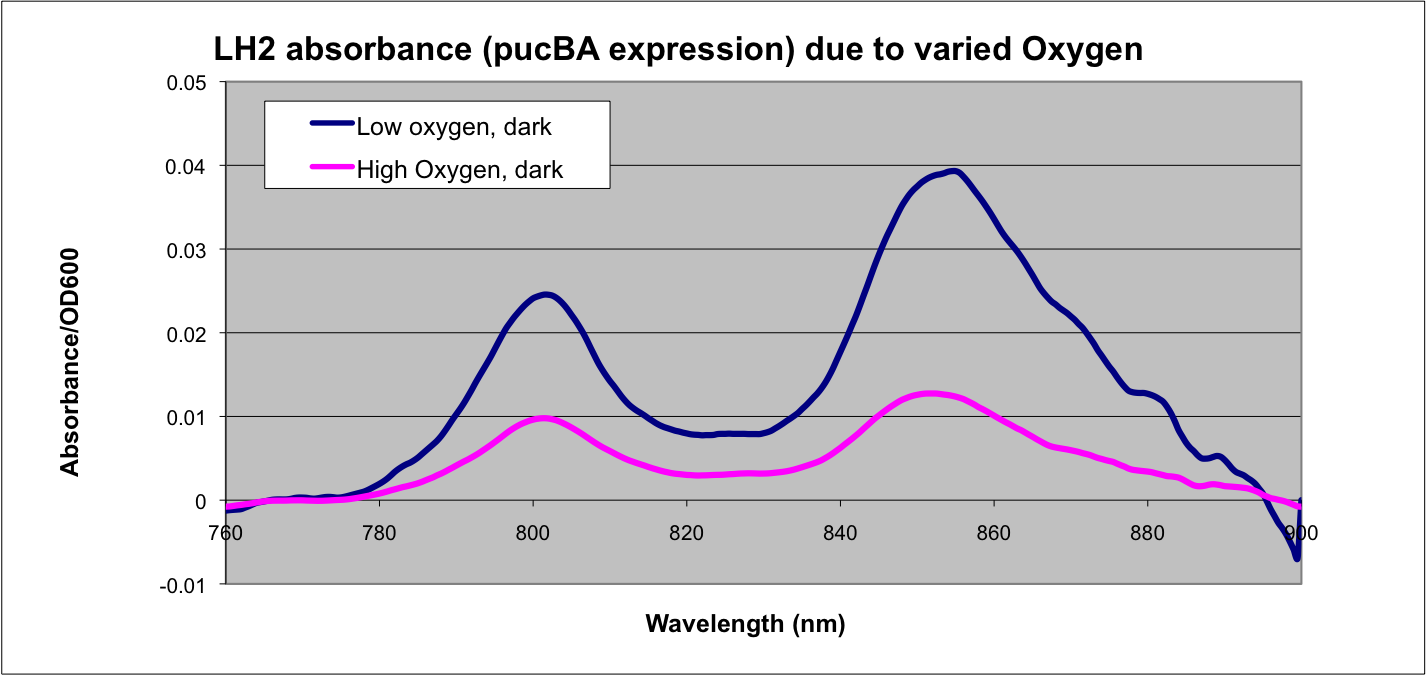
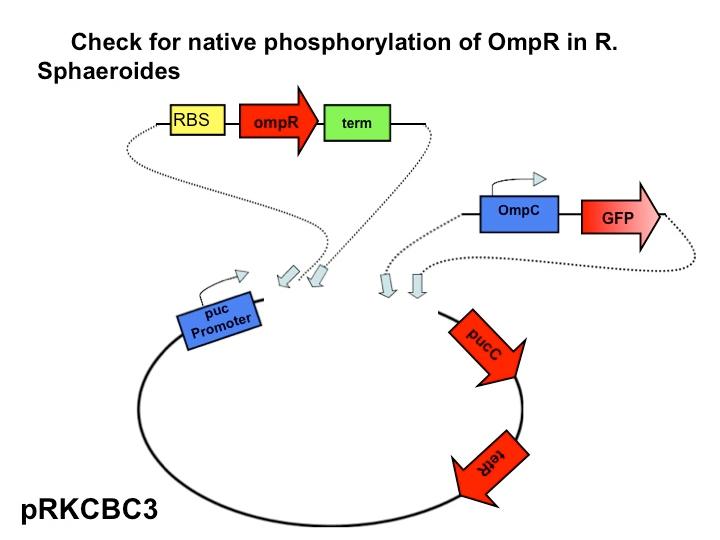
Team:Wash U/Biological Parts
From 2009.igem.org
(→Simulating a Bioreactor) |
|||
| Line 364: | Line 364: | ||
<font size="2"> | <font size="2"> | ||
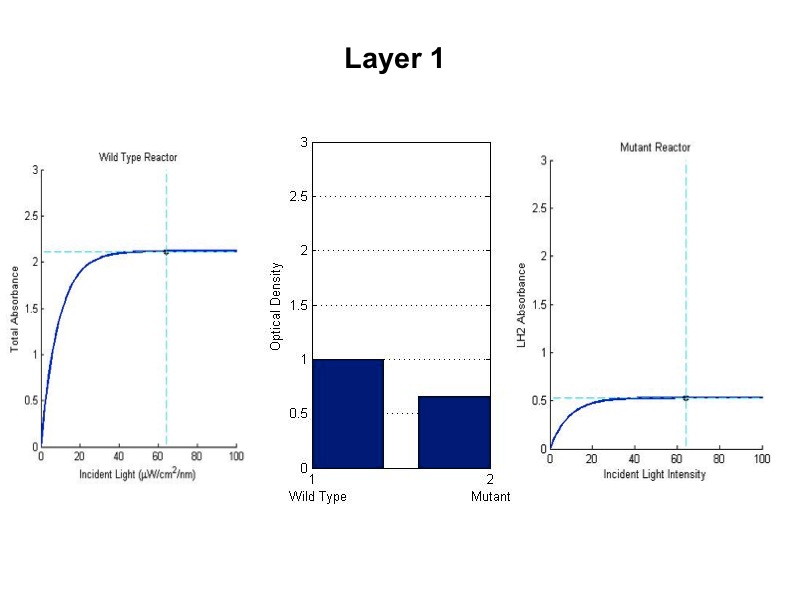
| - | '''Problem:''' In a typical reactor, cells at the surface absorb more than enough light to saturate their photosynthetic apparatus, transmitting less energy to deeper layers. For wild type cells, the “saturation curve” is approximately the same for all cells, regardless of their incident light intensity.<br> | + | '''Problem:''' In a typical reactor, cells at the surface absorb more than enough light to saturate their |
| - | '''Simulating our | + | |
| - | For our mutant cells, | + | photosynthetic apparatus, transmitting less energy to deeper layers. Cells operating past the saturation point |
| + | |||
| + | waste incident photons by non-photochemical quenching and possibly undergo photodamage. For wild type cells, the | ||
| + | |||
| + | “saturation curve” is assumed to be approximately the same for all cells in all layers, regardless of their | ||
| + | |||
| + | incident light intensity. This means that layers of cells on the exterior of the reactor nearest a light source | ||
| + | |||
| + | receive an overabundance of photons and in turn block the interior layers from receiving enough light. In an | ||
| + | |||
| + | optimal reactor, all layers would operate near their respective saturation points to maximize the photosynthetic | ||
| + | |||
| + | channeling of incident light energy. | ||
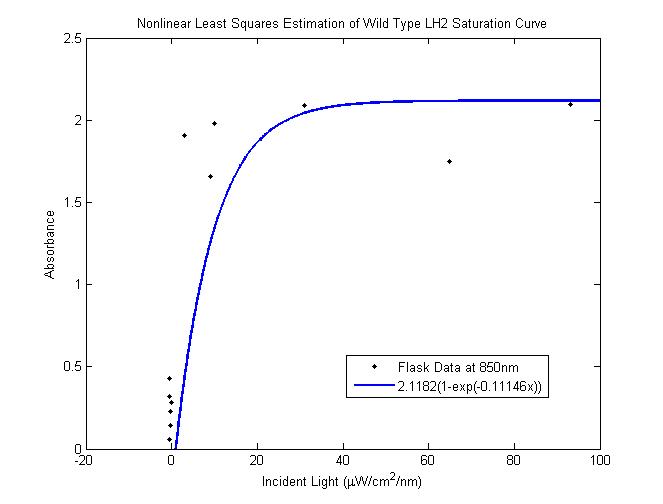
| + | The total saturation curve for wild type R. Sphaeroides was fit with a nonlinear least squares regression of the | ||
| + | |||
| + | form in Equation A. Data points were generated from calculating absorbance as the negative logarithm of the ratio | ||
| + | |||
| + | of the absolute irradiance detected on the next layer to incident absolute irradiance on a layer of cells. A | ||
| + | |||
| + | logistic form was chosen to account for the diminishing returns to absorption of further photons past a threshold | ||
| + | |||
| + | operating capacity of the photosynthetic apparatus. (1) | ||
| + | <br> | ||
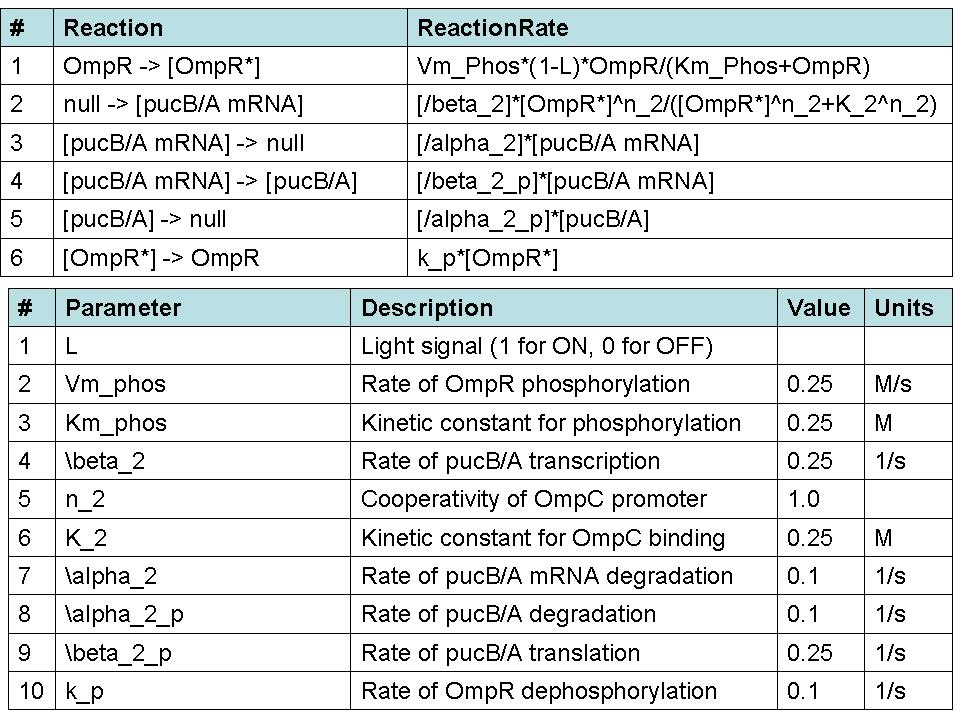
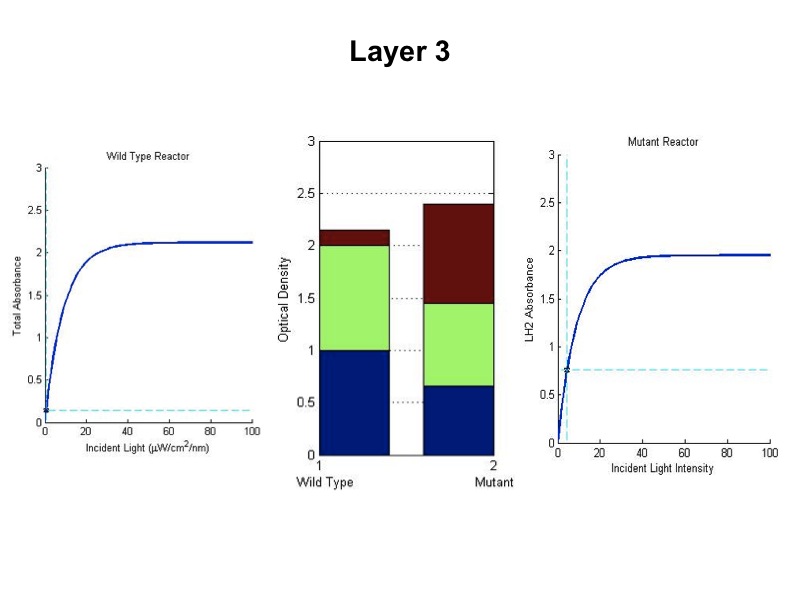
| + | '''Simulating our Mutant's advantage in a Bioreactor''' | ||
| + | For our mutant cells, the LH2 saturation curve for each layer scales as a function of light intensity. This | ||
| + | |||
| + | predicted behavior in the mutant is due to negative regulation of LH2 complex production as incident light | ||
| + | |||
| + | intensity increases. The scalar of the magnitude of the saturation curve was altered according to a predicted | ||
| + | |||
| + | exponential curve of LH2 production in repsone to changes in incident light. It was assumed that the system could | ||
| + | |||
| + | vary expression levels such that at high light intensities, the saturation curve is scaled to 25% of that of the | ||
| + | |||
| + | wild type. At low light intensities, LH2 production was assumed to have the potential to be up-regulated to 150% | ||
| + | |||
| + | of wild type expression levels. | ||
| + | The advantage this mutant would confer stems from the adaptive nature of the saturation curve heights. Cells | ||
| + | |||
| + | receiving the most light on the outside of the bioreactor saturate at low absorption levels. This allows more | ||
| + | |||
| + | light to transmit to further layers, which have elevated saturation curves due to lower incident light. | ||
| + | <br><br> | ||
| + | Assumptions: | ||
| + | - Light intensity at next layer is given by transmittance from previous layer (assumes no backscattering).<br> | ||
| + | - Total energy funneled to photosynthetic pathways is estimated as the sum of light absorbed by each layer. This | ||
| + | |||
| + | generalizes to the optical density measurement of cell culture density. | ||
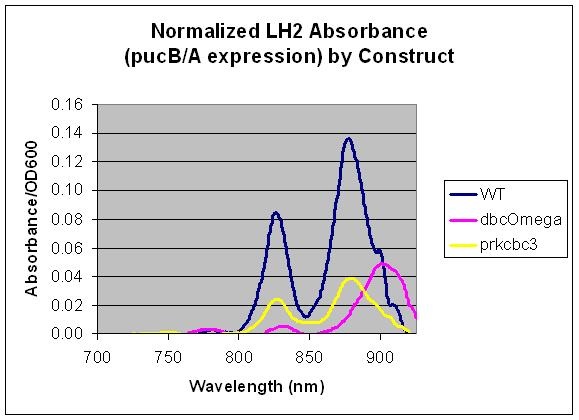
| + | - The constant wild type saturation curve inherently includes both LH2 and LH1 contributions to absorbance. The | ||
| + | |||
| + | mutant's variable saturation curve only accounts for LH2 absorbance, since this is the only complex under the | ||
| + | |||
| + | light-sensing regulation. To account for the component of absorbance provided by LH1, the proportion of total | ||
| + | |||
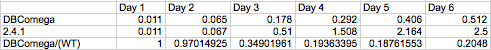
| + | optical density due strictly to LH1 was investigated by comparing the growth of wild type and LH2-knockout | ||
| + | |||
| + | (dbcOmega) cultures. It is evident that by day three the proportion of Optical Density accounted for by LH1 | ||
| + | |||
| + | absorption converges to a value near 0.2. In other words, at the phase the layers of cells have grown in the model, | ||
| + | |||
| + | 20% of total optical density can be attributed to LH1. To account for this, the absorption in the mutant was | ||
| + | |||
| + | divided by the factor (1-0.2) = 0.8. Then, the total optical density of the mutant cultures reflects total | ||
| + | |||
| + | absorption by both LH1 and LH2. | ||
| + | |||
| + | '''Model Assumptions''' <br> | ||
| + | - Light intensity at next layer is given by transmittance from previous layer (assumes no backscattering).<br> | ||
| + | - Total energy funneled to photosynthetic pathways is estimated as the sum of light absorbed by each layer. This | ||
| + | |||
| + | generalizes to the optical density measurement of cell culture density. <br> | ||
| + | - The constant wild type saturation curve inherently includes both LH2 and LH1 contributions to absorbance. The | ||
| + | |||
| + | mutant's variable saturation curve only accounts for LH2 absorbance, since this is the only complex under the | ||
| + | |||
| + | light-sensing regulation. To account for the component of absorbance provided by LH1, the proportion of total | ||
| + | |||
| + | optical density due strictly to LH1 was investigated by comparing the growth of wild type and LH2-knockout | ||
| + | |||
| + | (dbcOmega) cultures. It is evident that by day three the proportion of Optical Density accounted for by LH1 | ||
| + | |||
| + | absorption converges to a value near 0.2. In other words, at the phase the layers of cells have grown in the model, | ||
| + | |||
| + | 20% of total optical density can be attributed to LH1. To account for this, the absorption in the mutant was | ||
| + | |||
| + | divided by the factor (1-0.2) = 0.8. Then, the total optical density of the mutant cultures reflects total | ||
| + | |||
| + | absorption by both LH1 and LH2. <br> | ||
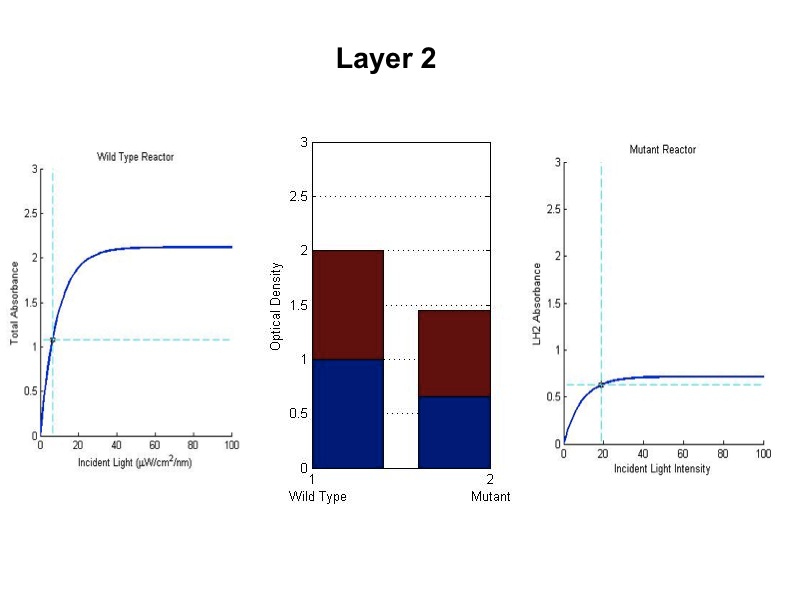
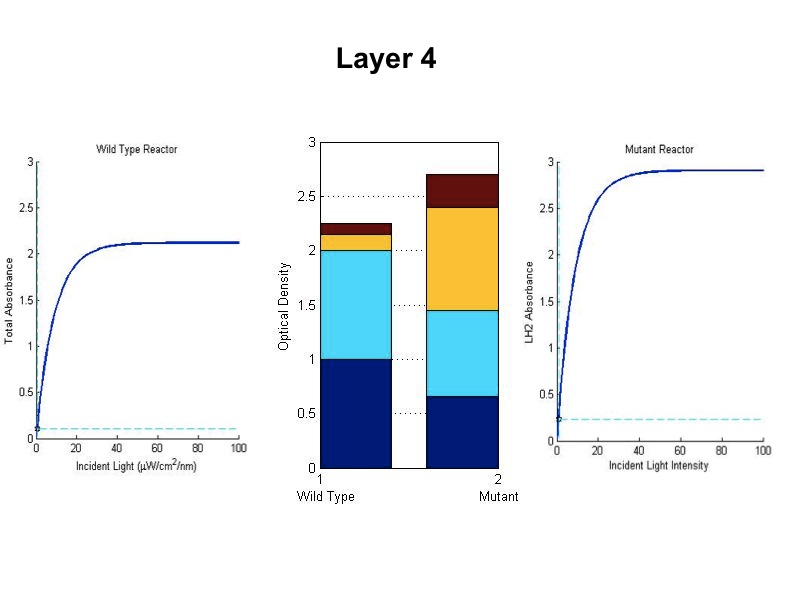
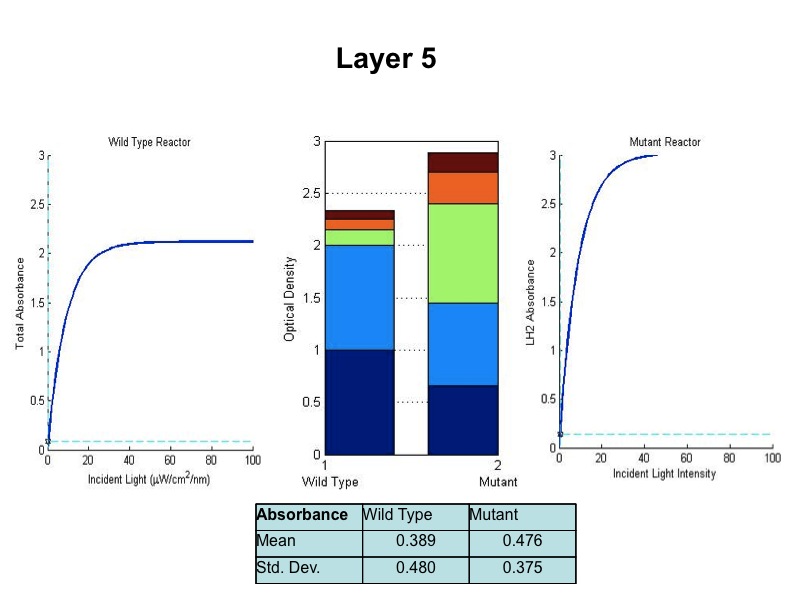
| + | - The model was revised upon gathering optical density data from the five layers of the bioreactor setup. (See | ||
| + | |||
| + | Results Figure 2a.) In the wild type, the optical density of the first flask of cells was much lower than | ||
| + | |||
| + | predicted, a phenomenon that was attributed to photodamage of the cells due to exposure to a large quantity of | ||
| + | |||
| + | light past the saturation point of the LH2 complexes. In the optical density data for the flasks, both the | ||
| + | |||
| + | dbcOmega knockout and the wild type logistically grew to an absorbance value of 1. This gave reason to put a hard | ||
| + | |||
| + | limit of 1 on the first flask's potential optical density. Any light left over from this cutoff was transmitted to | ||
| + | |||
| + | the next layer, as evidenced by in the increased growth of the second wild type flask in the Optical Density data. | ||
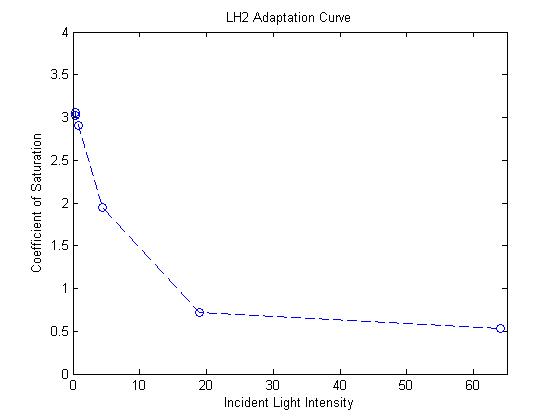
| + | - The response curve for the coefficient of saturation for the mutant due to changes in light intensity was modeled | ||
| + | |||
| + | as an inverse exponential form. In other words, the system reacts to increasing light intensity by exponentially | ||
| + | |||
| + | tapering the coefficient of saturation. | ||
<div style="text-align: left;"> | <div style="text-align: left;"> | ||
Revision as of 03:13, 22 October 2009

 "
"