Team:Illinois/3'-Acting sRNAs
From 2009.igem.org
(→mRNA Degradation Team) |
(→mRNA Degradation Team) |
||
| Line 47: | Line 47: | ||
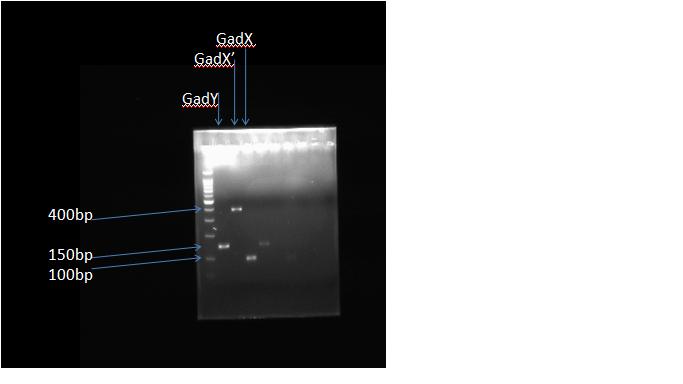
GadX 3' UTR (SwaI, XbaI restriction sites)-102bp, 393bp : | GadX 3' UTR (SwaI, XbaI restriction sites)-102bp, 393bp : | ||
| + | == '''July 9''' == | ||
| - | |||
We cloned the GadY gene as well as the 3' UTR of gadX. We used two different primers for gadX because we weren't sure which design would work the best. Primer 2 goes all the way to the start codon of gadW. | We cloned the GadY gene as well as the 3' UTR of gadX. We used two different primers for gadX because we weren't sure which design would work the best. Primer 2 goes all the way to the start codon of gadW. | ||
| Line 58: | Line 58: | ||
We then purified the PCR product that was not used in the gel. We also prepared an overnight of dh5ap. transformed with pXG-1. | We then purified the PCR product that was not used in the gel. We also prepared an overnight of dh5ap. transformed with pXG-1. | ||
| - | '''July 10 | + | == '''July 10''' == |
Today we performed a midiprep to extract the plasmid DNA from dH5ap. | Today we performed a midiprep to extract the plasmid DNA from dH5ap. | ||
Revision as of 19:04, 10 July 2009
Contents |
mRNA Degradation Team
Members:
Francis Lee
Lucy McCommas
Van Ceu
Goals:
Our goal is to work with sRNAs that bind to the 3' UTR. We will study this regulation system using the Vogel protocol. We want to have two sRNAs controlling one gene, which could be accomplished through using a combination of 5' and 3' acting sRNAs.
Background:
Colony Boil-Streak plate E. Coli K12 on LB agar, incubate overnight at 37C. Pick healthy colony with sterile glass pasteur pipette and innoculate 100 uL dH20. Boil for 10 min, centrifugate for 5 min at 14K rpm. Extract 1 uL of supernatant as template DNA for PCR.
GadY Gene Sequence: (K-12 substr. MG1655: 3662867-3663047)
ACTGAGAGCA CAAAGTTTCC CGTGCCAACA GGGAGTGTTA TAACGGTTTA TTAGTCTGGA GACGGCAGAC TATCCTCTTC CCGGTCCCCT ATGCCGGGTT TTTTTTATGT CTGAGTAAAA CTCTATAATC TTATTCCTTC
GadX 3' UTR Sequence: (K-12 substr. MG1655: 3662641-3662717)
AGTTTTACTC AGACATAAAA AAAACCCGGC ATAGGGGACC GGGAAGAGGA TAGTCTGCCG TCTCCAGACT AATAAA
GadX 3' UTR Sequence (2): (K-12 substr. MG1655: 3662641-3663008)
AGTTTTACTC AGACATAAAA AAAACCCGGC ATAGGGGACC GGGAAGAGGA TAGTCTGCCG TCTCCAGACT AATAAACCGT TATAACACTC CCTGTTGGCA CGGGAAACTT TGTGCTCTCA GTAAGTTAAA TATAACTTTT ACTGGAAATA AGATCAGCCA TTTTTTTATA AACATAAGCT ATACGCTGTG CGAAAATATA TTCGTGCTGC ATTTACTTAT TATCAATTAA CTGTTATGCA AAACTACTTT GTGGATAAAT TTTGGTCCTA CCAAATCTGG CAGTTTTTGC GCTAAGAAAC AGTCTGGCAT CATTTCATTA GTATACTGAA ATTGAAATAA TCGCAGTATG AAATATAAGG GATAATC
Primers Used:
- Forward/sense gadY (sRNA gene) primer: ACTGAGAGCACAAAGTTTCC
- Reverse/antisense gadY (sRNA gene) primer: (XbaI)-GTTTTTTCTAGAGAAGGAATAAGATTATAGAG
- Forward/sense gadX (sRNA target sequence) primer: (SwaI)-GTTTTTATTTAAATAGTTTTACTCAGACATAAAAAAAACC
- Reverse/antisense gadX (sRNA target sequence) primer: (XbaI)-GTTTTTTCTAGATTTATTAGTCTGGAGACGGCAG
- Reverse/antisense gadX (sRNA target sequence) primer 2: (XbaI)-GTTTTTTCTAGAGATTATCCCTTATATTTCATAC
PCR Products:
GadY GENE (XbaI restriction site)-152bp
GadX 3' UTR (SwaI, XbaI restriction sites)-102bp, 393bp :
July 9
We cloned the GadY gene as well as the 3' UTR of gadX. We used two different primers for gadX because we weren't sure which design would work the best. Primer 2 goes all the way to the start codon of gadW.
We did this with a successful PCR.
We then purified the PCR product that was not used in the gel. We also prepared an overnight of dh5ap. transformed with pXG-1.
July 10
Today we performed a midiprep to extract the plasmid DNA from dH5ap.
References
[http://jb.asm.org/cgi/content/abstract/186/20/6698 GadY, a Small-RNA Regulator of Acid Response Genes in Escherichia coli] Jason A. Opdyke, Ju-Gyeong Kang, and Gisela Storz
 "
"