Team:Washington/Project/Secretion
From 2009.igem.org
| Line 8: | Line 8: | ||
</style></html> | </style></html> | ||
<div> | <div> | ||
| - | <div style="float:left;">'''[https://2009.igem.org/Team:Washington/Project | + | <div style="float:left;">'''[https://2009.igem.org/Team:Washington/Project Project Description]'''</div> |
<div style="float:right;">'''[https://2009.igem.org/Team:Washington/Project/Display Display >]'''</div> | <div style="float:right;">'''[https://2009.igem.org/Team:Washington/Project/Display Display >]'''</div> | ||
</div> | </div> | ||
Revision as of 05:14, 19 October 2009
Background
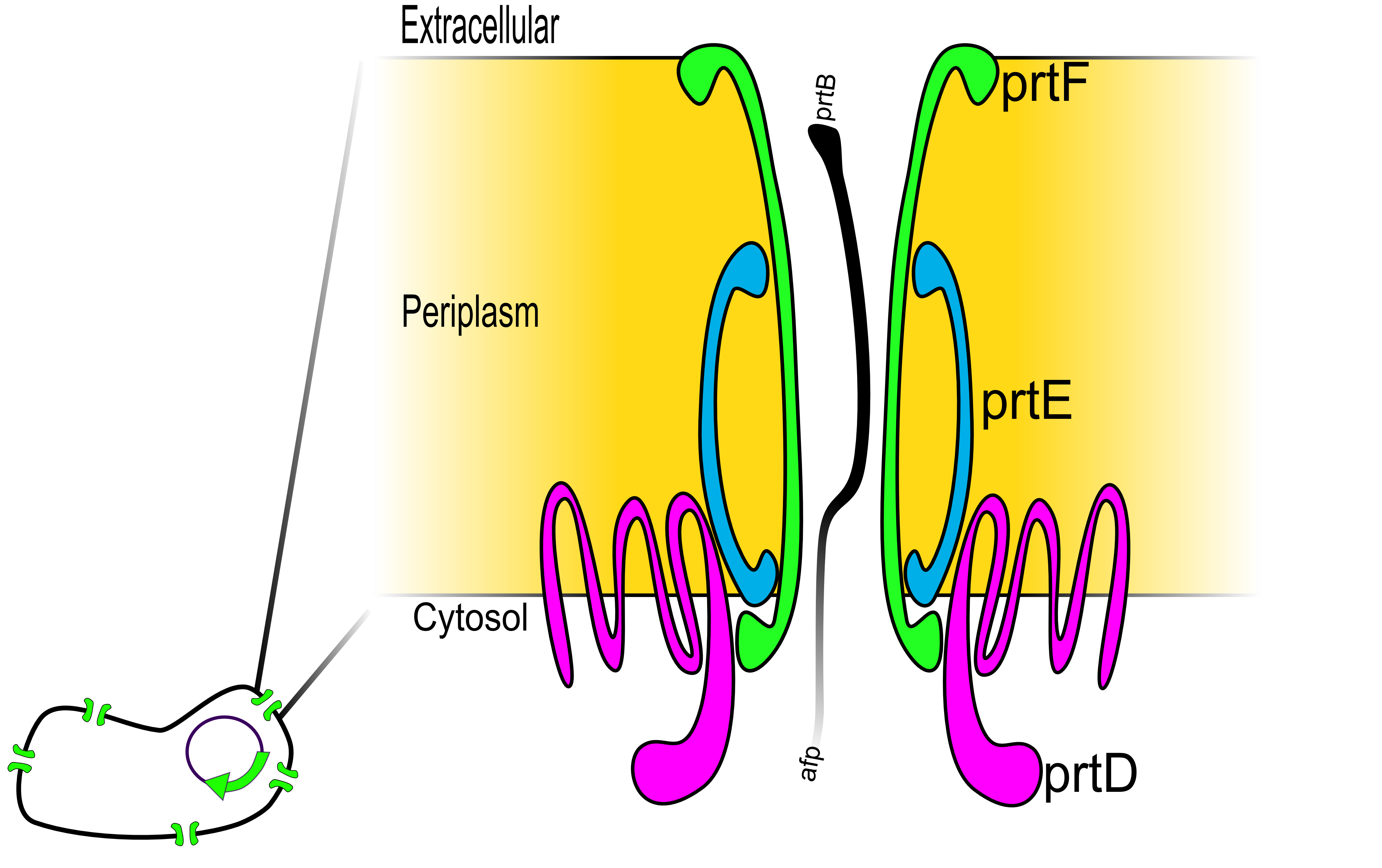
The ability to secrete our target protein completely out of the cell and into the media is an essential feature of our project. While the laboratory E. coli strain K-12 contains genes for a type II system (the genes have been silenced 1), it lacks a type I secretion system capable of exporting proteins to the extracellular space. To implement this ability into E. coli, we chose to install the type I secretion system of Erwinia chrysanthemi 2. We choose a Type I secretion system over more traditional secretion systems since it has been shown to secrete a protein of interest all the way from the cytosol to the medium with any extra machinery 2,3,4. More traditional system, such as the Tat or Sec pathways, only secrete the protein of interest to the periplasim. 5
The secretion system is composed of three genes prtD, prtE, and prtF2. The PrtD, PrtE, and PrtF proteins are thought to form a selective pore that connects the cytoplasm directly to the external medium. As the diagram shows, PrtD and PrtE interact with the inner membrane, while PrtF interacts with the outer membrane. This system recognizes the C-terminal tail of the target (the prtB secretion tag) and selectively shuttles it from the cytosol, all the way through the periplasim, and out to the extracellular space. The exact mechanism of how this occurs is not currently understood.
Experiments
For the Secretion System our goal was to:
- Construct the Secretion Plasmid
- Characterize the Secretion System
Constructing the Secretion System
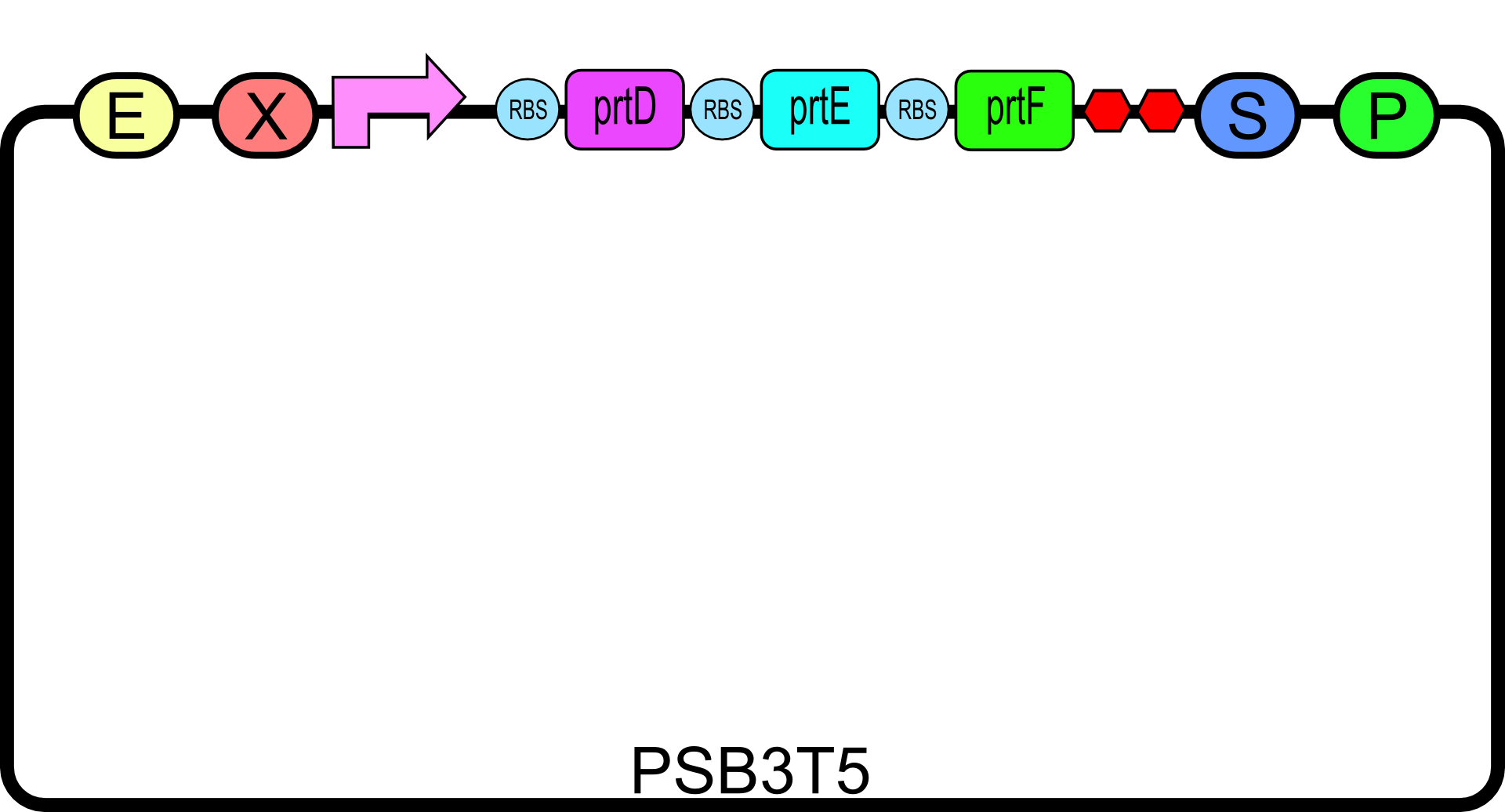
To create the secretion system, we first synthesized a coding sequence that would produce the 3 gene constructs described above2. To do this, we synthesized biobrick compatible versions of each gene and their native ribosome binding sites from oligos as described in our gene synthesis protocol.
After synthesis of each individual gene w/RBS biobrick, they were then pieced together using biobrick standard assembly to form a single construct. Three different versions of the secretion construct were then created with the placement of one of three different promoters in front of the gene construct, a high ([http://partsregistry.org/wiki/index.php?title=Part:BBa_J23100 BBa_J23100]), a medium ([http://partsregistry.org/wiki/index.php?title=Part:BBa_J23114 BBa_J23114]), or a low ([http://partsregistry.org/wiki/index.php?title=Part:BBa_J23113 BBa_J23113]) strength promoter . All three designs were placed in pSB3T5. For our experiments, only the high strength promoter was used.
Characterization of Secretion System (Secreting the Target Protein)
To test the functionality of our secretion system, we decided to see how well the system could export the Target protein into the media. To do this, we cloned GFP (BBa_E0040) into our Target vector, and transformed the vector into cells (strain BL21 lacq) containing the Secretion plasmid. The cells containing our TargetGFP vector and Secretion plasmid were then grown in a large culture (50 ml) and expression of the Target protein was induced via IPTG. After a period of growth, the culture was then spun down to separate the cells from the media. The amount of fluorescence found in the supernatant was then used to quantify the amount of Target protein being secreted (a detailed protocol of the experiment can be found here).
Results
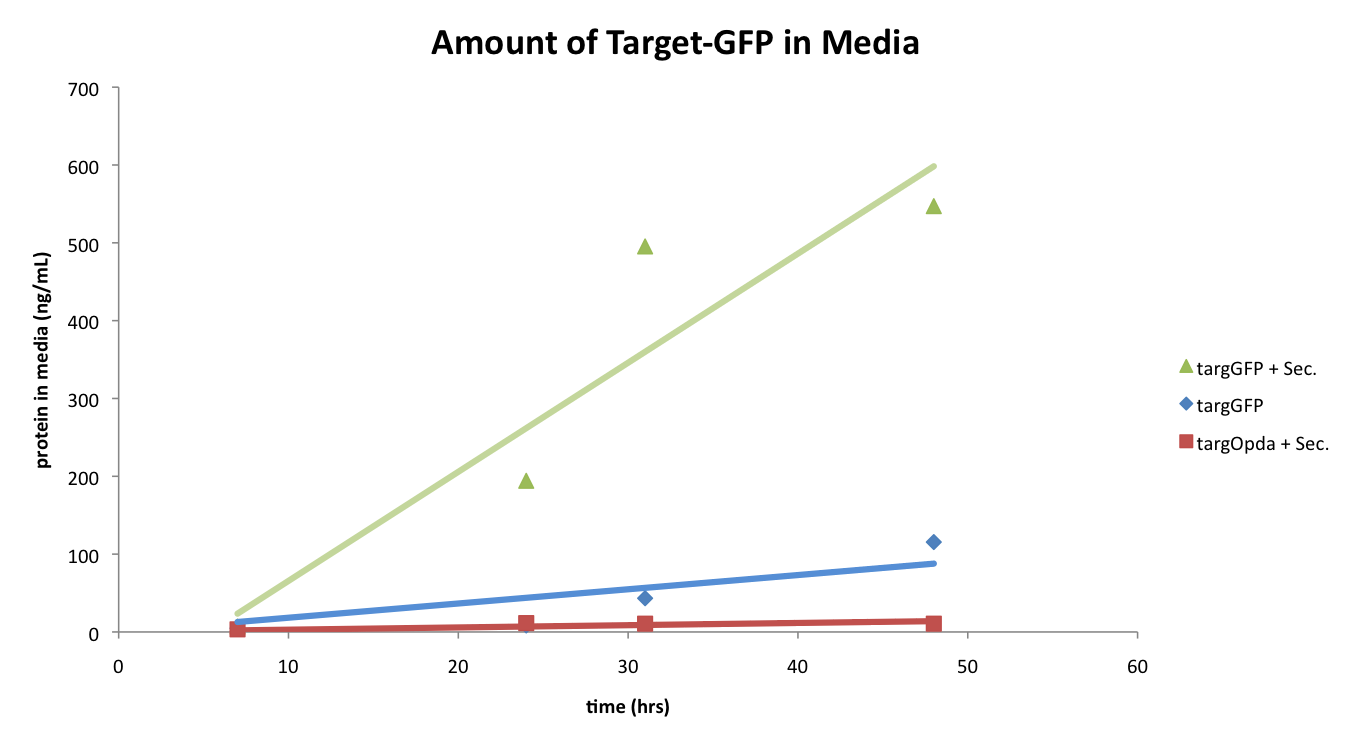
In our first round of experiments with the cells containing both the TargetGFP construct and Secretion plasmid, two controls were also tested: cells containing only the TargetGFP construct and cells containing the TargetOpdA construct and the Secretion plasmid.
The plot below shows the results of our first secretion test.
As shown above, the cells containing both the TargetGFP and Secretion plasmid released a much higher amount of the target protein into the media when compared to the two controls. This result was quite exciting as it appeared that our secretion system was working great.
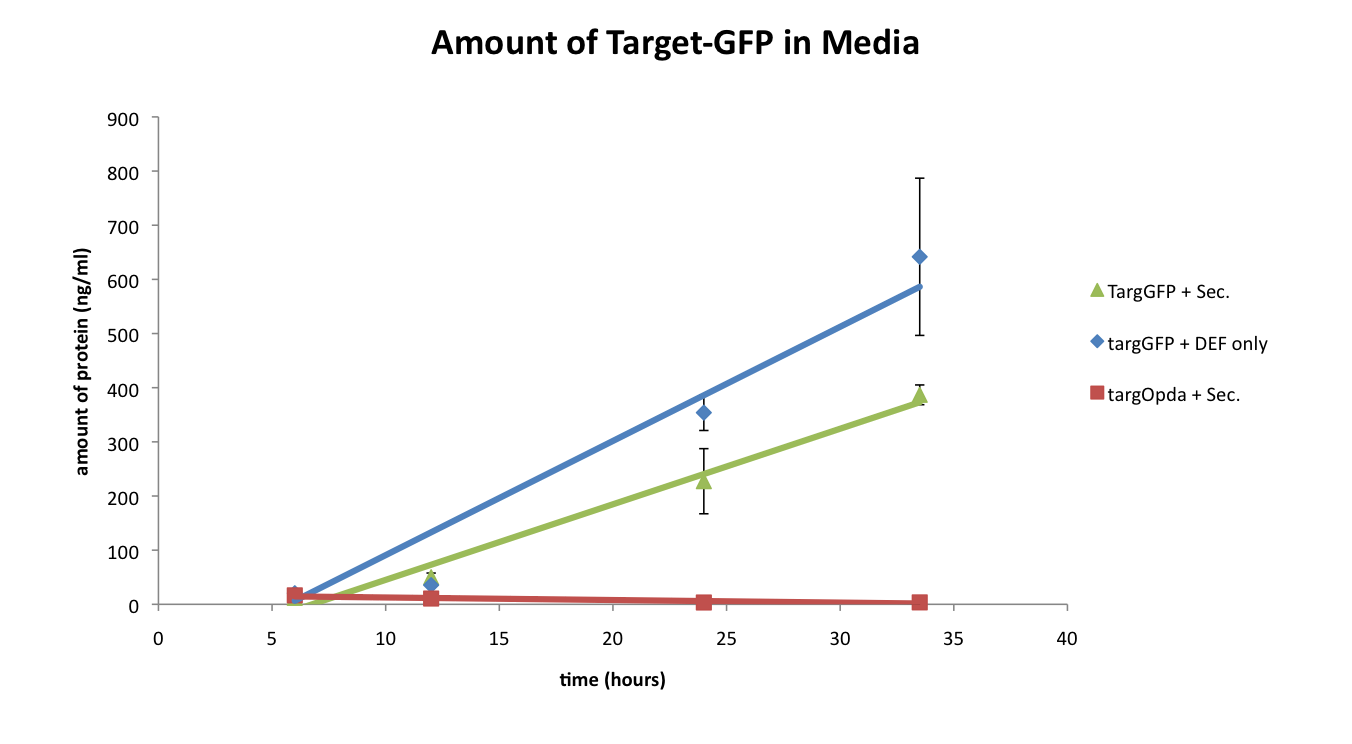
However, after verifying these results, we realized that one of the controls was not entirely fair since the culture containing only the TargetGFP vector had just one plasmid and thus was only grown with one antibiotic (ampicillin). To make a more comparable control, the cells containing only the TargetGFP construct were also transformed with a promoter-less Secretion plasmid, and the experiment was repeated.
The plot below shows the results of the second secretion test.
As shown above, the addition of the promoter-less secretion plasmid into the TargetGFP control also caused the culture to release an elevated amount of target protein into the media. From this result, it appeared that the Secretion system was not responsible for the elevated amount of TargetGFP in the media, but that it was actually an artifact caused by the extra plasmid that the Secretion system was on (pSB3T5). We hypothesize that this artifact could be the result of a couple different reasons: the extra plasmid (and thus extra antibiotic resistance needed) causes an elevated amount of cell stress that causes premature cell lysis, or the cell membrane was rendered more permeable since the extra plasmid encoded tetracycline resistance via a membrane pump protein.
A couple different variations of the above experiments were then tried to show secretion:
- We tried varying the amount of IPTG used for induction (1-500 uM) to make sure we weren't overloading the cells
- We changed the cell strain from BL21(lacq) to DH5a, the cell strain used in reference 2
- We used PSB3T5 with out an insert, to eliminate the potential leaky expression from a promoter less gene
- We varied the temperature of expression to see if the proteins would be more stable
Unfortunately we obtained the same results for all of these additional experiments, in which there was no increase of protein concentration in the media when the secretion system was present (data not shown).
Conclusion
Based on these results, we have yet to show that the Secretion system is functioning. However, this system has been shown to work properly in the literature2, and there are many parameters that we have yet to reproduce and optimize. Proteins that have been secreted by this Type I system and other similar ones include GFP2,3, lipase3, Trichoderma harzianum endochitinase2, trout growth hormone2, ompC2, and lacZ2. This causes us to believe that our part, which has been sequenced verified, should work given a little more tweaking, to see our ideas see Future Directions
Continue to Display
Citations
- Olivera. Expression of the endogenous type II secretion pathway in Escherichia coli leads to chitinase secretion.
- Palacios et al. [http://www.ncbi.nlm.nih.gov/pubmed/11157948 Subset of Hybrid Eukaryotic Proteins Is Exported by the Type I Secretion System of Erwinia chrysanthemi, Secrevtion of GFP in E. Coli]
- Chung et al. [http://www.ncbi.nlm.nih.gov/pubmed/19178697 Export of recombinant proteins in Escherichia coli using ABC transporter, Secretion of GFP in E. Coli]
- Holland et al. [http://www.ncbi.nlm.nih.gov/pubmed/16092522 Type 1 protein secretion in bacteria, the ABC-transporter dependent pathway]
- [http://www.ncbi.nlm.nih.gov/pubmed/15763404 Mergulhaoa and Summersb and Monteiroa. Recombinant protein secretion in Escherichia coli]
 "
"