Team:UCSF/Project
From 2009.igem.org
Jasonpark1 (Talk | contribs) m (image size and caption edits) |
Jasonpark1 (Talk | contribs) m |
||
| Line 173: | Line 173: | ||
[[Image:cellbeads_scheme.jpg|400px|thumb|center|'''Modular and generalizable scheme for attaching beads to cells''']] | [[Image:cellbeads_scheme.jpg|400px|thumb|center|'''Modular and generalizable scheme for attaching beads to cells''']] | ||
| - | Some important early considerations were therefore: | + | Some important early considerations for us were therefore: 1) Best to use a scheme that does not require much (or any) genetic manipulation, so that we can use our scheme quickly and easily with any cell or cell type, 2) Use well-established chemistries and reagents, 3) Be able to swap and interchange modular parts. |
| - | + | ||
| - | + | ||
| - | + | ||
For our pilot experiments this summer, we used the simplest possible parts that we could think of. For cellular attachment, we used the jack bean lectin protein [http://en.wikipedia.org/wiki/Concanavalin_A Concanavalin A] (ConA), which binds mainly to internal and nonreducing terminal alpha-mannosyl groups. We left the linker out in some cases, though in others we used [http://en.wikipedia.org/wiki/Polyethylene_glycol polyethylene glycol (PEG)] linkers. For bead attachment, we used either streptavidin-biotin chemistry, passive adsorption of protein on the surface of polystyrene beads, or [http://en.wikipedia.org/wiki/Carbodiimide carbodiimide chemistry] with the crosslinker EDC. | For our pilot experiments this summer, we used the simplest possible parts that we could think of. For cellular attachment, we used the jack bean lectin protein [http://en.wikipedia.org/wiki/Concanavalin_A Concanavalin A] (ConA), which binds mainly to internal and nonreducing terminal alpha-mannosyl groups. We left the linker out in some cases, though in others we used [http://en.wikipedia.org/wiki/Polyethylene_glycol polyethylene glycol (PEG)] linkers. For bead attachment, we used either streptavidin-biotin chemistry, passive adsorption of protein on the surface of polystyrene beads, or [http://en.wikipedia.org/wiki/Carbodiimide carbodiimide chemistry] with the crosslinker EDC. | ||
Revision as of 10:25, 21 October 2009
Contents |
Part 1: NAVIGATION: Rewiring the cell to move toward new chemical signals
Motivation: Why is this useful?
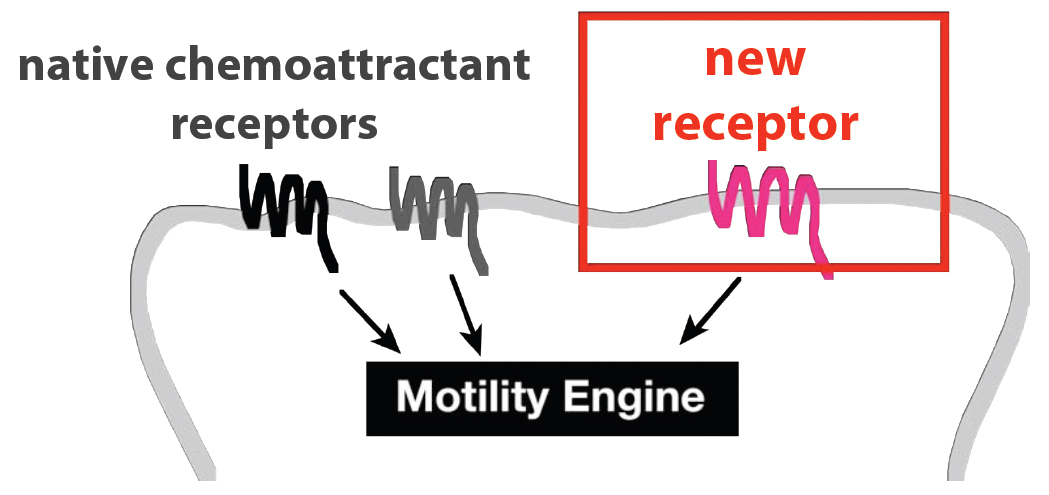
We envision a cellular robot that could travel to practically any site in the human body. This would provide a flexible platform that could be used for a variety of therapeutic tasks. The first step toward achieving this goal is to broaden the range of possible chemotactic targets for our cells. Ideally, we could connect virtually any input to chemotaxis in a generalized way.
Approach:
Neutrophils sense most of their chemotactic signals through [http://en.wikipedia.org/wiki/G_protein_coupled_receptor G protein-coupled receptors] (GPCRs). The spectrum of chemical signals to which these cells respond is therefore determined, at least in part, by the set of GPCRs they express. Can this spectrum be broadened arbitrarily by the introduction of new GPCRs? We tested this idea by transiently expressing 23 exogenous GPCRs in HL-60 (neutrophil-like) cells.
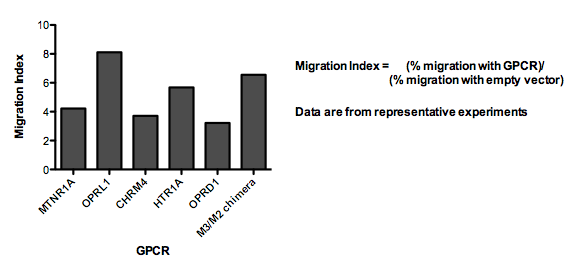
These cells were then tested for their ability to migrate toward ligands for the new GPCRs in multiwell [http://en.wikipedia.org/wiki/Chemotaxis_assay#Two-chamber_techniques Boyden chamber] assays. We measured the fold change in % of cells migrating toward the new ligand (with vs without added GPCR) at the peak response. We refer to this ratio as the "Migration Index." For receptors that appeared to activate a migration response (Migration Index > 3), we also conducted time-lapse microscopy to determine whether the cell movement was directed toward the gradient of ligand.
Results:
6 of the GPCRs we transiently expressed in our cells resulted in a Migration Index > 3.
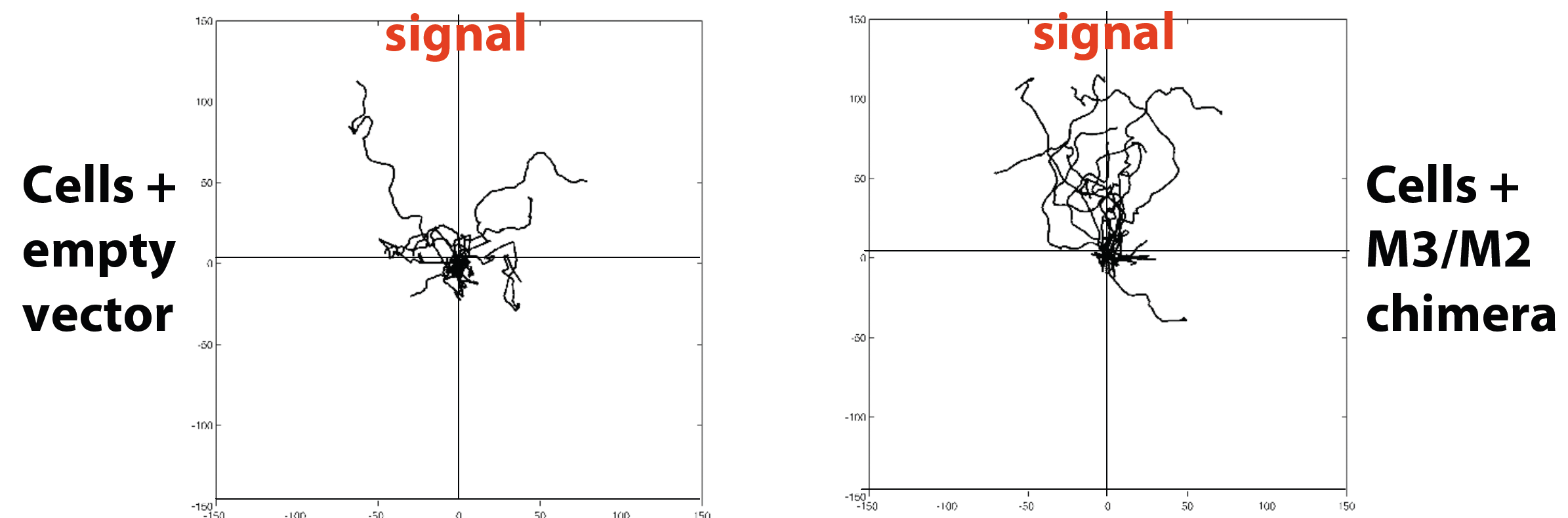
Here, we show an example of one of these receptors (M3/M2 chimera) mediating directional migration up a stable, linear gradient of ligand. Transfected cells are fluorescent, and the concentration of ligand increases in the direction corresponding to the top of the image:
To directly compare these cells to those transfected with empty vector, we plotted center-zeroed tracks of individuals cells in each treatment. Qualitatively, cells expressing the chimera tend to move more directly toward the source of ligand.
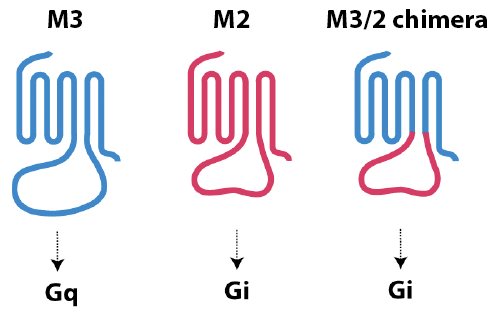
Common characteristics of chemotaxis receptors: All 6 receptors we identified couple to the [http://en.wikipedia.org/wiki/Gi_alpha_subunit Gi] signaling pathway. The behavior of the M3/M2 chimera, however, suggests that it may be possible to convert receptors with different coupling specificities into chemotaxis receptors. To generate this chimera, the third intracellular loop (i3) from the M3 muscarinic acetylcholine receptor ([http://en.wikipedia.org/wiki/Gq_alpha_subunit Gq] coupled) was exchanged with that of M2 muscarinic receptor (Gi coupled). It has previously been shown that this chimera now couples to Gi.
Why the i3 loop allows the M3/M2 chimeric receptor to signal to the cell's chemotaxis machinery remains a question for further study. However, the possibility exists that more Gq-coupled (and possibly [http://en.wikipedia.org/wiki/Gs_alpha_subunit Gs]-coupled) receptors could be converted in this way, thus dramatically increasing the number of potential chemotaxis targets.
Summary and outlook:
We have shown that we can program our cells to migrate to new chemical signals by expressing exogenous GPCRs. One of these GPCRs, a chimeric protein, suggests that there may be a way to convert even more GPCRs into chemotaxis receptors. In the future, we are interested in understanding more about why certain receptors mediate chemotaxis while others do not. It would also be interesting to go back to the receptors that did not work, and confirm that they are functional and signaling to other known pathways.
Part 2: NAVIGATION: Tuning receptor sensitivity
Motivation: Why is this useful?
In our experiments, we could control exactly how much ligand was presented to our cells. In "real-life," however, we would want our cellular robots to be able to respond to a variety of signal strengths: from very low to very high. To accomplish this, we would want to be able to control the sensitivity of our receptors, or how the receptor's output changes when the measured quantity of ligand changes.
Approach:
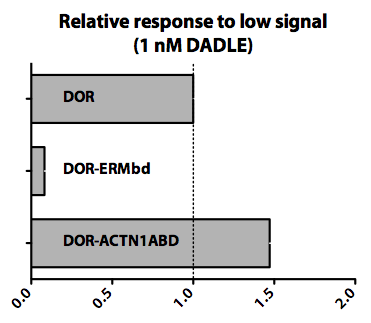
We felt that one key determinant of sensitivity would be the number of receptors present at the plasma membrane of the cell. Therefore, we measured the migration response of a receptor (delta Opioid receptor) whose recycling behavior could be engineered by fusing different recycling interaction modules to the C terminus of the GPCR. We tested a number of such receptor-module fusions for migration response and compared them to receptor alone. The primary assay here was again the Boyden chamber (transwell) assay.
Results:
We found that virtually any protein domain/module known to alter the recycling of delta Opioid receptor (DOR) affected cellular migration to a low concentration of ligand (1 nM DADLE). Below, we show two examples of such domains. The actin binding domain from alpha-Actinin-1 (ACTN1ABD) appears to potentiate cellular migration when compared to the wild-type receptor. On the other hand, cellular response is inhibited at this concentration when DOR is bound to a domain of EBP50 (ERMbd) that binds to ERM (ezrin/radixin/moesin) family of proteins.
Summary and outlook:
We have shown that we can tune the sensitivity of a receptor both up and down by fusing it to different recycling modules. Next, we would like to measure recycling directly, and determine whether changes in recycling are necessary for this difference in chemotactic response. All experiments in HL-60 cells would benefit the generation of stable cell lines, which would allow us to more precisely quantify these new behaviors.
Part 3: SPEED: Engineering accelerators and brakes
A cellular cruise control by modulating cell polarity with feedback loops
Motivation: Why is this useful?
Just like we have control over speed in a car – we can brake or accelerate – it would be useful to engineer such behavior into our cellular nanorobots. Just think about it: We could speed cells up so that they reach their targets faster and stop them once they have arrived or do not behave properly.
Background:
For these experiments we chose Dictyostelium discoideum cells to test our prototypical brakes and accelerators quickly. We expect that our brakes and accelerators can be used in a plug and play fashion because Dicty’s way of movement is very similar to a neutrophil’s:
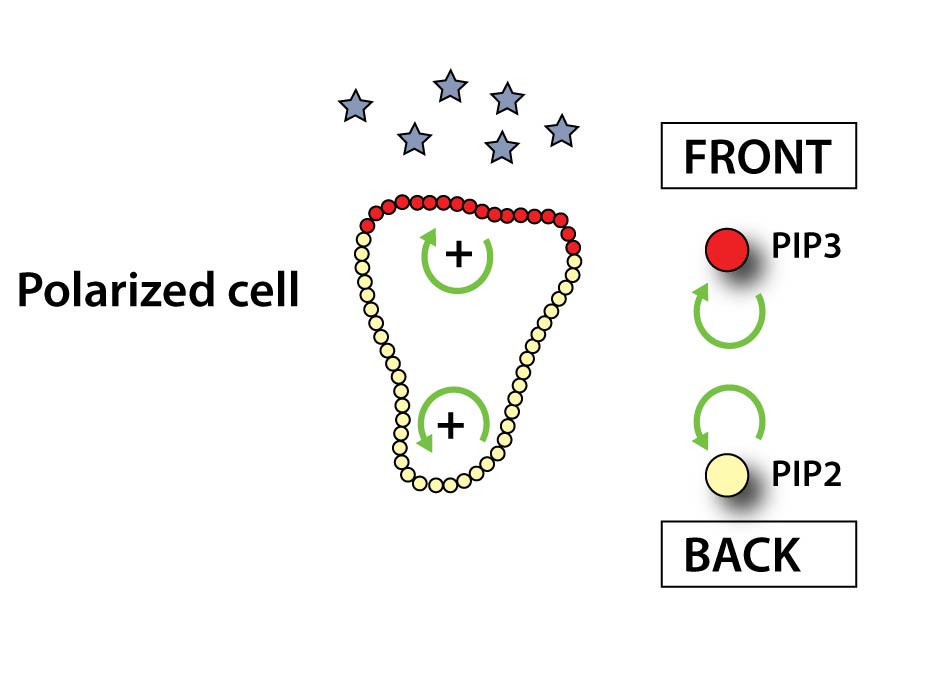
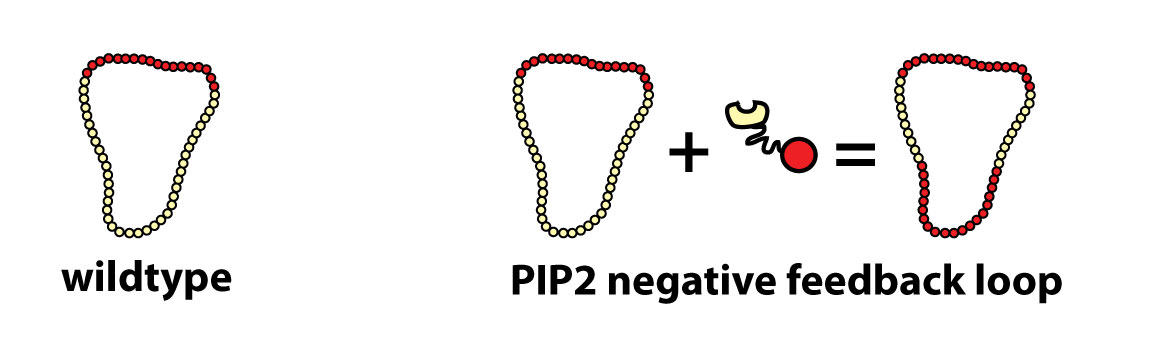
When a receptor binds chemoattractant, it induces the conversion of PhosphatidylInositol(4,5)bisphosphate (PIP2) to PhosphatidylInositol(3,4,5)trisphosphate (PIP3) (two signaling lipids in the plasma membrane) at the front of our cells. In a positive feedback loop PIP3 triggers the formation of more PIP3 at the front while similarly PIP2 leads to more PIP2 production at the sides and rear of the cell. This system sets the axis of polarity of the cell. The PIP3 patch at the front aligns the actin network and accordingly functions as a ‘turbo boost’ pushing the cell forward.
Approach:
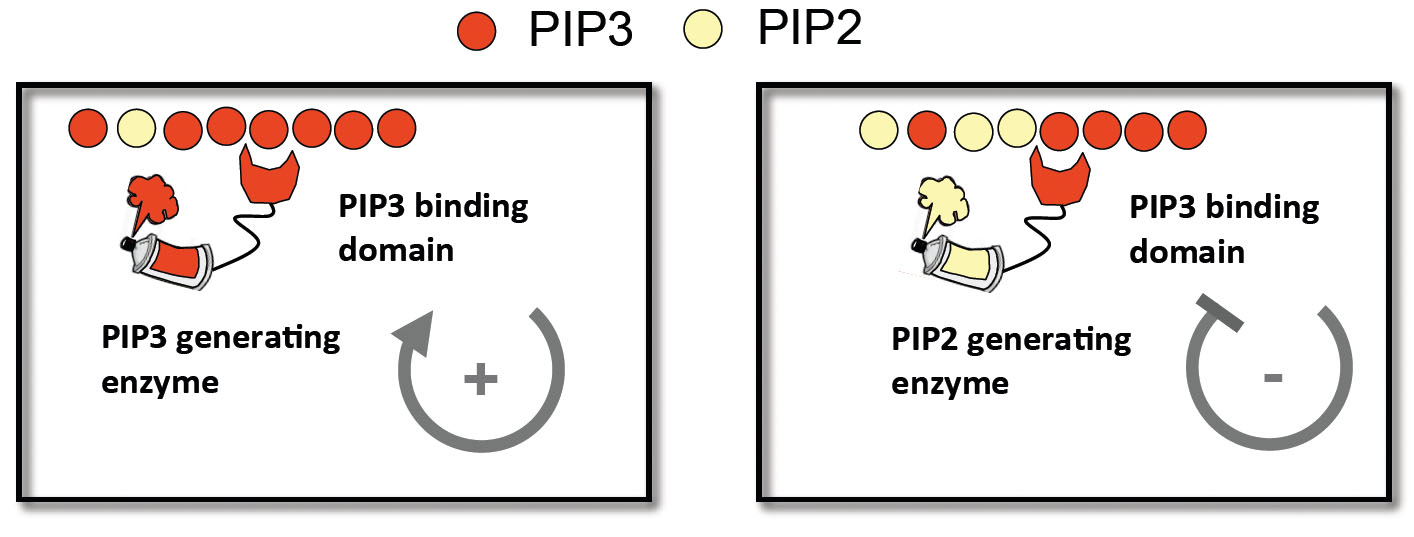
Inspired by nature we tried to build accelerators and brakes by introducing our own synthetic protein based feedback loops. We designed feedback elements by fusing localization and catalytic domains involved in PIP3 production and degradation to modulate localization and level of PIP3 and PIP2 in the cell.
Here is an example of a positive feedback loop: a PIP3 binding localization domain fused to a PIP3 producing catalytic domain could produce more PIP3 where there is already PIP3- at the front. This might strengthen polarity and accelerate a cell.
Results:
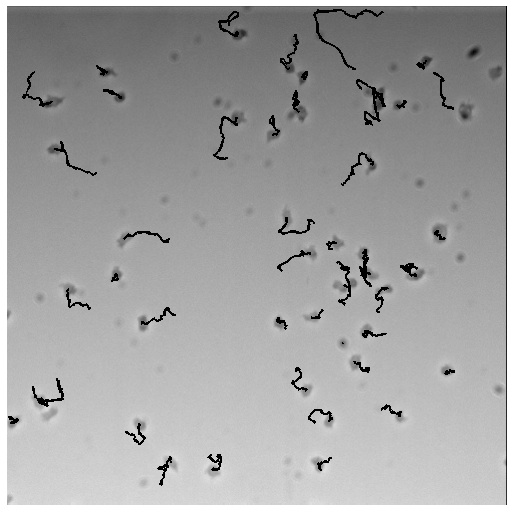
Over the summer we assembled more than 100 fusions of localization and catalytic domains and screened whether they work. How? We measured the effect our constructs have on motility of Dictyostelium cells: stronger polarity should make cells faster while weaker polarity ought to slow them down!
Here is an overview of all feedback loops we screened and the effect they had on the speed of cells. We used automated cell tracking on more than 196 hours worth of movies (note: one movie is 10 minutes!) and identified strains that moved faster or slower at a very stringent statistical cutoff (p<0.0001).
This way we were able to identify 7 brakes and 1 accelerator!
Check out the movie of one of our strong brakes (PTEN-RasC dominant active (da))[http://www.youtube.com/watch?v=_9od33Nx06Y] compared to wildtype [http://www.youtube.com/watch?v=Vtdtf8-zSRs].
This is indeed an effect of fusing the particular localization to the catalytic domain as neither of them alone has such a strong effect (see details).
We hypothesize that this construct acts as a negative feedback loop on PIP2 - (generating PIP3 where PIP2 should be) thereby confusing the cell with multiple fronts:
Summary and outlook:
We have screened more than 100 synthetic feedback elements for their ability to accelerate or slow down speed of cell motility. We have isolated a hand full of functional elements. Now we need to confirm the mechanism of action of these elements. In the future we would like to make them inducible by a signal from outside – like a stoplight!
Part 4: PAYLOAD: Harnessing the cell to deliver cargo
Motivation: Why is this useful?
Once a cellular robot has detected and traveled to a site of disease in the human body, it should be able to perform a useful therapeutic task. One simple but very useful task that we envision our cells performing in the body is the targeted delivery of drugs or signaling molecules to diseased areas. This would be useful in the delivery of all sorts of substances to targeted areas in the body. Some examples include: 1) cases where a drug is toxic and off-target side effects are dose-limiting (e.g. chemotherapeutics used for cancer), 2) cases where precise delivery of a contrast agent to diseased areas would aid in diagnosis (e.g. sites of cancer metastasis), 3) cases where localized delivery of a specific bioactive molecule to a site would be therapeutically beneficial (e.g. delivery of specific growth factors to areas of wound healing and regeneration).
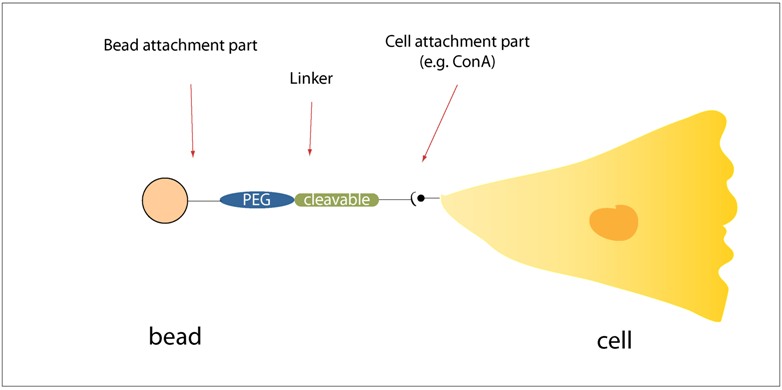
Toward the end of engineering our cellular robots to carry useful cargos, we designed and began testing a generalizable scheme for tethering fluorescent polymer microspheres ("beads") to the outside of our cells.
Approach:
We designed a modular and generalizable scheme for attaching fluorescent polymer microspheres to the outside of our cells. To make the approach modular, we thought of our strategy as having three main interchangeable parts: 1) Bead attachment part, 2) Linker, 3) Cell attachment part. By ensuring that we make the parts interchangeable, we made the approach generalizable. For example, changing out the cell attachment part for a different one would allow us to attach beads to a different type of cell. Likewise, we could use different types of linkers including photocleavable or enzymatically cleavable ones. Changing out the bead attachment part could allow us to attach not only beads but other cargos to cells.
Some important early considerations for us were therefore: 1) Best to use a scheme that does not require much (or any) genetic manipulation, so that we can use our scheme quickly and easily with any cell or cell type, 2) Use well-established chemistries and reagents, 3) Be able to swap and interchange modular parts.
For our pilot experiments this summer, we used the simplest possible parts that we could think of. For cellular attachment, we used the jack bean lectin protein [http://en.wikipedia.org/wiki/Concanavalin_A Concanavalin A] (ConA), which binds mainly to internal and nonreducing terminal alpha-mannosyl groups. We left the linker out in some cases, though in others we used [http://en.wikipedia.org/wiki/Polyethylene_glycol polyethylene glycol (PEG)] linkers. For bead attachment, we used either streptavidin-biotin chemistry, passive adsorption of protein on the surface of polystyrene beads, or [http://en.wikipedia.org/wiki/Carbodiimide carbodiimide chemistry] with the crosslinker EDC.
To observe the cells, we coated glass-bottomed [http://www.nuncbrand.com/en/page.aspx?ID=235 Lab-Tek II Chambered Coverglass] slides with fibronectin, adhered cells to the slide, and then incubated with varying concentrations of modified beads followed by a wash step. We then visualized the cells using time-lapse microscopy.
Results:
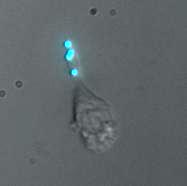
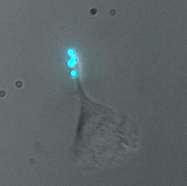
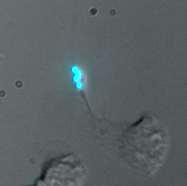
We have been able to observe our cells exhibiting motility while tethered to "cargos" of fluorescent beads. Below are two representative movies, each taken using fluorescent beads with ConA (no linker). Frame rate is 15 seconds (real time) per frame.
Here are some stillshots from the movies.
Summary and outlook:
We have shown that we can tether simple fluorescent bead "cargos" to our cells. We are using a simple, generalizable, and modular scheme to attach these cargos, and we will continue to try different attachment and linker parts to improve on our initial result. Some things we are working on include efficiency and robustness of attachment and internalization of beads by cells.
We would also like to explore the ability of cells to carry larger bead cargos and different types of cargos (non-bead). In addition, we would like to study the physical limitations of these cells in carrying cargo. For example, it would be particular interesting to determine whether the cells can carry carry cargos through complex environments such as three-dimensional hydrogel environments, endothelial cell monolayers, and more.
Methods
Navigation:
- HL-60: Transfection Protocol - here is how we get our constructs into HL-60 cells
- HL-60: Boyden Chamber (Transwell) Protocol - here is how we assay chemotaxis in HL-60 cells
- HL-60: Time-lapse Microscopy Protocol - here is how we film HL-60 cells
Engineering Speed:
- Cloning: Aar1 Shuffle - besides BBFRFC28, here is an actual step by step scheme we used for some of our combinatorial assembly of parts
- Dicty: Transformation Protocol - here is how we get our constructs into Dicty
- Dicty: Motility Assay Protocol - here is how we prepare and film Dicty cells
Payload:
Selected Reading
Navigation (New Sensors):
Receptors induce chemotaxis by releasing the betagamma subunit of Gi, not by activating Gq or Gs. Neptune ER, Bourne HR. Proc Natl Acad Sci U S A. 1997 Dec 23;94(26):14489-94.
Navigation (Sensor Sensitivity):
Engineered protein connectivity to actin mimics PDZ-dependent recycling of G protein-coupled receptors but not its regulation by Hrs. Lauffer BE, Chen S, Melero C, Kortemme T, von Zastrow M, Vargas GA. J Biol Chem. 2009 Jan 23;284(4):2448-58. Epub 2008 Nov 10.
Engineering Speed:
G protein signaling events are activated at the leading edge of chemotactic cells. Parent CA, Blacklock BJ, Froehlich WM, Murphy DB, Devreotes PN. Cell. 1998 Oct 2;95(1):81-91.
PI3-kinase signaling contributes to orientation in shallow gradients and enhances speed in steep chemoattractant gradients. Bosgraaf L, Keizer-Gunnink I, Van Haastert PJ. J Cell Sci. 2008 Nov 1;121(Pt 21):3589-97. Epub 2008 Oct 7.
Payload:
Microoxen: Microorganisms to move microscale loads. Weibel DB, Garstecki P, Ryan D, DiLuzio WR, Mayer M, Seto JE, Whitesides GM. PNAS August 23, 2005 vol. 102 no. 34 11963-11967.
| Home | The Team | The Project | Parts Submitted to the Registry | Our summer experience | Notebook | Human Practices |
|---|
 "
"