Team:Warsaw/Project/endosome
From 2009.igem.org
| Line 2: | Line 2: | ||
==Escape from the phagosome== | ==Escape from the phagosome== | ||
| + | |||
| + | Although the induction of phagocytosis and entrance into the eukaryotic cell seems to be simple, this is not the final step of the bacteria's invasion. Majority of the enteropathogenic bacteria are incapable of dividing within the [https://2009.igem.org/Team:Warsaw/Glossary#endosome phagosome], but subsequently to leaving the [https://2009.igem.org/Team:Warsaw/Glossary#endosome phagosome] they can proliferate rapidly within the cytoplasm. | ||
==Theoretical basis== | ==Theoretical basis== | ||
[[Image:Endosome_escape.png|thumb|180px]] | [[Image:Endosome_escape.png|thumb|180px]] | ||
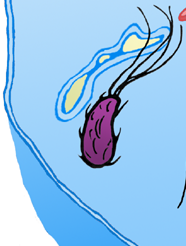
| - | + | Escape from the [https://2009.igem.org/Team:Warsaw/Glossary#endosome phagosome] is possible due to the [https://2009.igem.org/Team:Warsaw/Glossary#listeriolysin hemolysin], which causes membrane permeabilisation and [https://2009.igem.org/Team:Warsaw/Glossary#endosome phagosome] disruption [16]. It was previously demonstrated that transfection with hemolysin-encoding gene is sufficient to enable the escape from phagosomal vesicle of both ''Bacillus subtilis'' and ''Escherichia coli''[1]. <br/> | |
| - | + | The process of escaping from the phagosome is usually strictly regulated. An interesting example is the [https://2009.igem.org/Team:Warsaw/Glossary#phoP.2FPhoQ PhoP/PhoQ regulon], which exists in ''Salmonella enterica'' ser. ''typhimurium'' LT2 (and some other bacteria). It's a two-component regulatory system which replies to intramacrophage conditions like low pH or low metal ions concentration, leading to the activation of PhoP-dependent genes responsible for virulence of bacteria, like the [https://2009.igem.org/Team:Warsaw/Glossary#mgtc_gene_promoter MgtC] gene. | |
===Endosome detection operon=== | ===Endosome detection operon=== | ||
| Line 12: | Line 14: | ||
Fig 3. Overview of endosomal detection operon. It is composed of cro – antirepressor from phage λ, tetR – repressor of tetracycline resistance operon, cI – repressor from phage λ and cfp – Cyan Fluorescent Protein. | Fig 3. Overview of endosomal detection operon. It is composed of cro – antirepressor from phage λ, tetR – repressor of tetracycline resistance operon, cI – repressor from phage λ and cfp – Cyan Fluorescent Protein. | ||
| - | Low endosomal pH and low concentration of metal ions | + | Low endosomal pH and low concentration of metal ions leads to the decreasing likelihood of the transmembrane receptor kinase phoQ binding its ligands – divalent cations. This enables the phosphorylation of phoP by phoQ. Phosphorylated phoP activates the gene expression from [https://2009.igem.org/Team:Warsaw/Glossary#mgtc_gene_promoter MgtC promoter] leading to the production of proteins encoded by the endosome detection operon. The [https://2009.igem.org/Team:Warsaw/Glossary#cI cI] protein inactivates the invasion operon and the TetR protein activates cytoplasmic operon. Not all genes encoded by cytoplasmic operon are expressed in the endosome, due to the presence of the cro box sequence, which binds the cro protein thus stopping the trancription. The cro protein is encoded by the endosome detection operon. After the bacterium escapes into the cytoplasm the endosome detection operon genes are no longer expressed since phoP is not phosphorylated in cytoplasmic conditions. The process can be monitored by observing the CFP fluorescence protein. |
{{WarFoot1}} | {{WarFoot1}} | ||
Revision as of 18:33, 21 October 2009
Escape from the phagosome
Although the induction of phagocytosis and entrance into the eukaryotic cell seems to be simple, this is not the final step of the bacteria's invasion. Majority of the enteropathogenic bacteria are incapable of dividing within the phagosome, but subsequently to leaving the phagosome they can proliferate rapidly within the cytoplasm.
Theoretical basis
Escape from the phagosome is possible due to the hemolysin, which causes membrane permeabilisation and phagosome disruption [16]. It was previously demonstrated that transfection with hemolysin-encoding gene is sufficient to enable the escape from phagosomal vesicle of both Bacillus subtilis and Escherichia coli[1].
The process of escaping from the phagosome is usually strictly regulated. An interesting example is the PhoP/PhoQ regulon, which exists in Salmonella enterica ser. typhimurium LT2 (and some other bacteria). It's a two-component regulatory system which replies to intramacrophage conditions like low pH or low metal ions concentration, leading to the activation of PhoP-dependent genes responsible for virulence of bacteria, like the MgtC gene.
Endosome detection operon
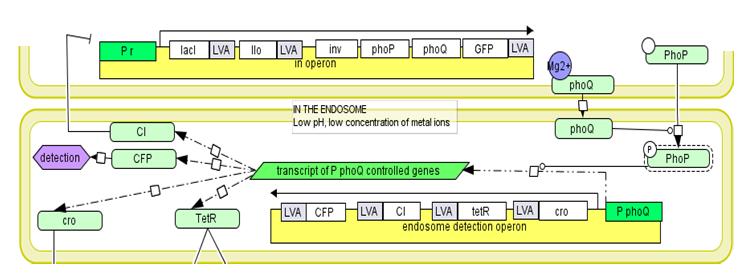
Fig 3. Overview of endosomal detection operon. It is composed of cro – antirepressor from phage λ, tetR – repressor of tetracycline resistance operon, cI – repressor from phage λ and cfp – Cyan Fluorescent Protein.
Low endosomal pH and low concentration of metal ions leads to the decreasing likelihood of the transmembrane receptor kinase phoQ binding its ligands – divalent cations. This enables the phosphorylation of phoP by phoQ. Phosphorylated phoP activates the gene expression from MgtC promoter leading to the production of proteins encoded by the endosome detection operon. The cI protein inactivates the invasion operon and the TetR protein activates cytoplasmic operon. Not all genes encoded by cytoplasmic operon are expressed in the endosome, due to the presence of the cro box sequence, which binds the cro protein thus stopping the trancription. The cro protein is encoded by the endosome detection operon. After the bacterium escapes into the cytoplasm the endosome detection operon genes are no longer expressed since phoP is not phosphorylated in cytoplasmic conditions. The process can be monitored by observing the CFP fluorescence protein.
 "
"