Team:MIT
From 2009.igem.org
| Line 25: | Line 25: | ||
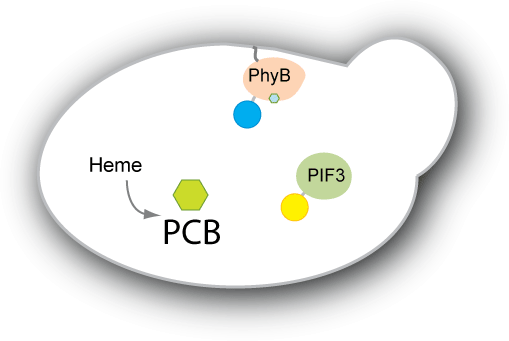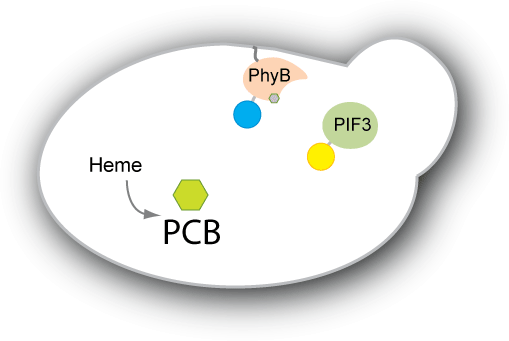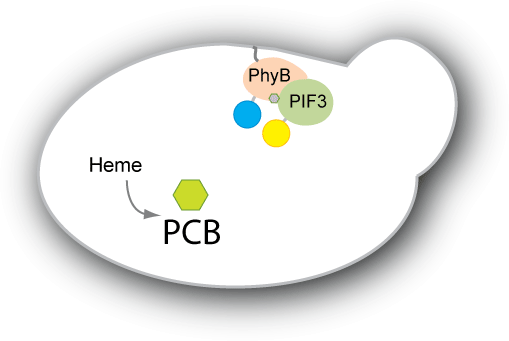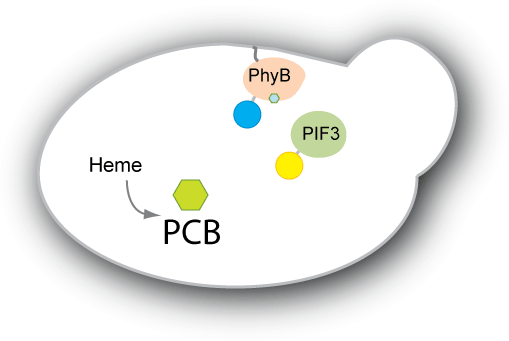
| - | We have two main goals for our project. Our first goal is to be able to engineer yeast to produce the chromophore PCB endogenously. Right now, PCB has to be extracted from plants or cyanobacteria or from strains of E. coli that have been designed to produce PCB. We would like the switch to be self-contained in the strain we engineer, so we would like to engineer a strain of yeast to produce PCB. Our second goal is to engineer a system that adopts the PhyB-PIF3 switch to control protein localization within the cell. We plan to have either PhyB or PIF3 constitutively anchored to the target | + | We have two main goals for our project. Our first goal is to be able to engineer yeast to produce the chromophore PCB endogenously. Right now, PCB has to be extracted from plants or cyanobacteria or from strains of E. coli that have been designed to produce PCB. We would like the switch to be self-contained in the strain we engineer, so we would like to engineer a strain of yeast to produce PCB. Our second goal is to engineer a system that adopts the PhyB-PIF3 switch to control protein localization within the cell. We plan to have either PhyB or PIF3 constitutively anchored to the target location (e.g. mitochondrial membrane, nuclear membrane, vacuole etc). The other will then be bound to our protein of interest or diffuse within the cell depending on the PhyB-PIF3 interaction. When pulsed with red light, the PIF3 and PhyB will bind, causing the protein of interest to localize to the target. |
| - | Such a system would be useful in creating a reversible switch for | + | Such a system would be useful in creating a reversible switch for many purposes. For example, one could synchronize a culture of cells in one part of the cell cycle without having to deal with temperature sensitive mutants or adding chemicals externally to arrest the cell cycle. The system would also be useful to study the kinetics of localization and delocalization, as well as provide an easy on-off switch for expression of essential genes. Light switchable transcriptional regulation has shown to be an effective way of increasing or decreasing gene expression quickly. By applying the concept of light switching to post-translational control as well, we aim to have greater control not only over gene expression, but also how the protein functions within the cell. |
|width="30%" class="green2" | | |width="30%" class="green2" | | ||
Revision as of 22:17, 21 October 2009
We are the 2009 MIT iGEM page.
An aesthetics makeover of this page is due sometime between now and the jamboree.
Project OverviewTo maximize control over a biological system, it would beneficial to have quick, reversible control over each step in gene expression, from transcription to translation to post-translational processing. Much work has been done to create switchable promoters, toggled by pulses of light, to control rates of transcription for genes of interest. The MIT iGEM team aims to take this concept and apply it to post-translational control, more specifically protein targeting in yeast. Our goal is to make a system in which a pulse of light causes a protein of interest to localize to one part of the cell. When pulsed with another wavelength of light, the protein will diffuse. In this way, a user can easily control both localization and delocalization of a protein of interest.
|
MembersMIT Department of Biological Engineering Undergraduates
Graduate
Professors
|
 "
"