Back To Top
Analysis
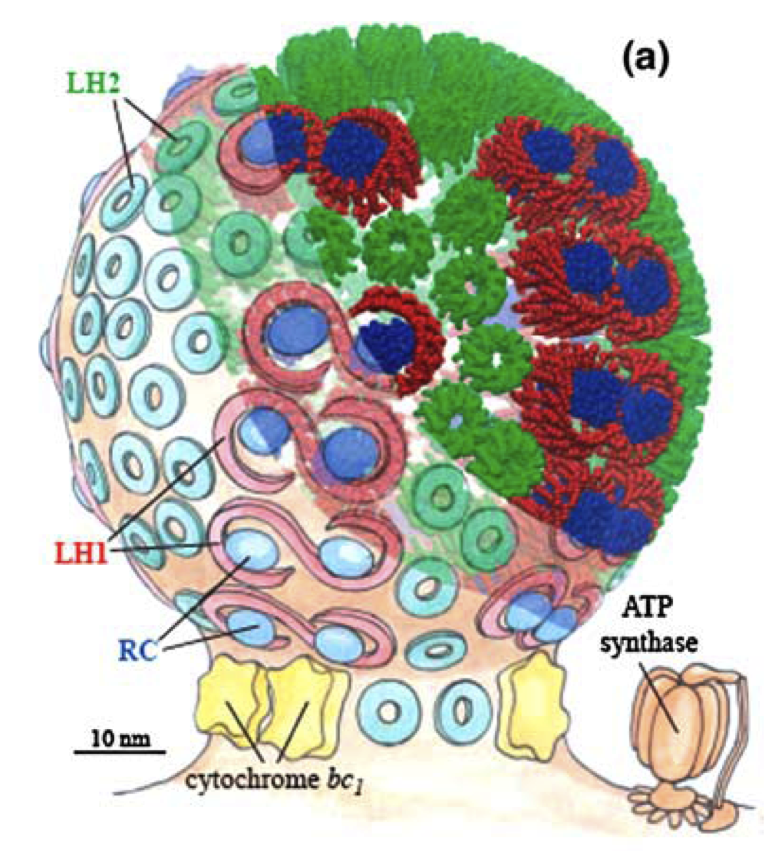
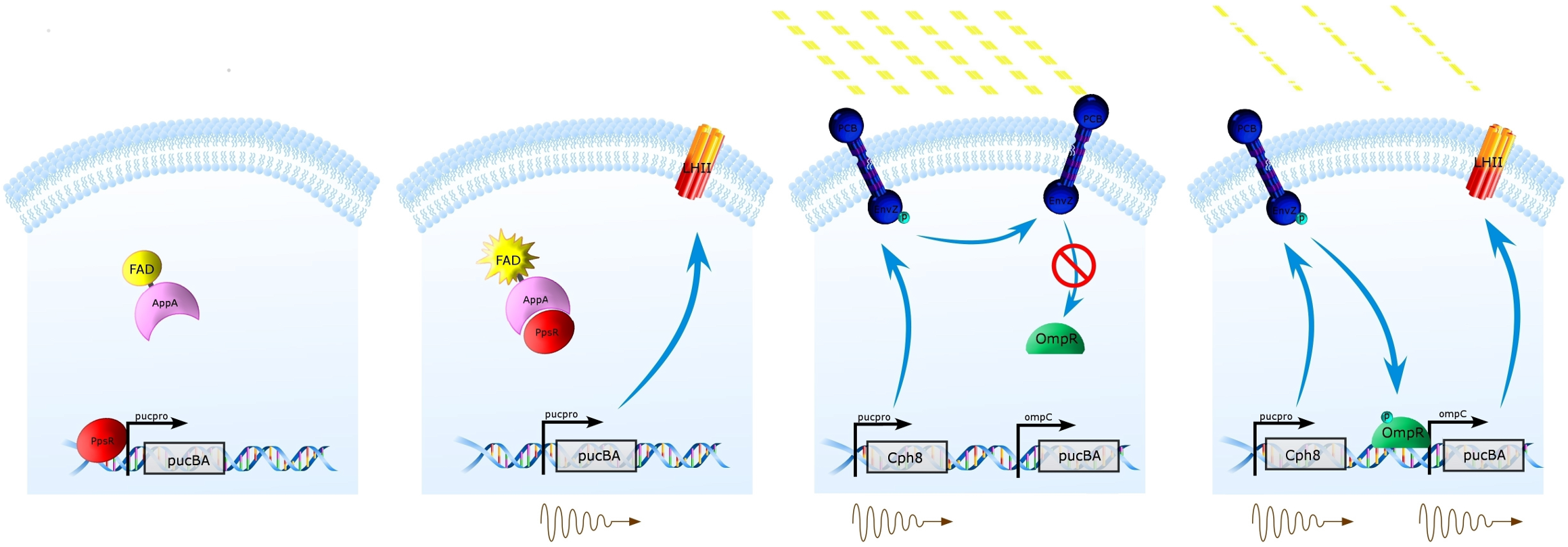
The tissue flask experiment was an interesting way to observe the effects of a full knockout of the Light Harvesting Antenna Complex 2 (LH2) vs the Wild Type on an array of conseuctive photobioreactors. A couple of conclusions can be drawn from this data that can be used in modeling our synthetic regulation system for LH2 and in the future design of systems for the optimization of Light Harvesting Antenna size for photobioreactors .
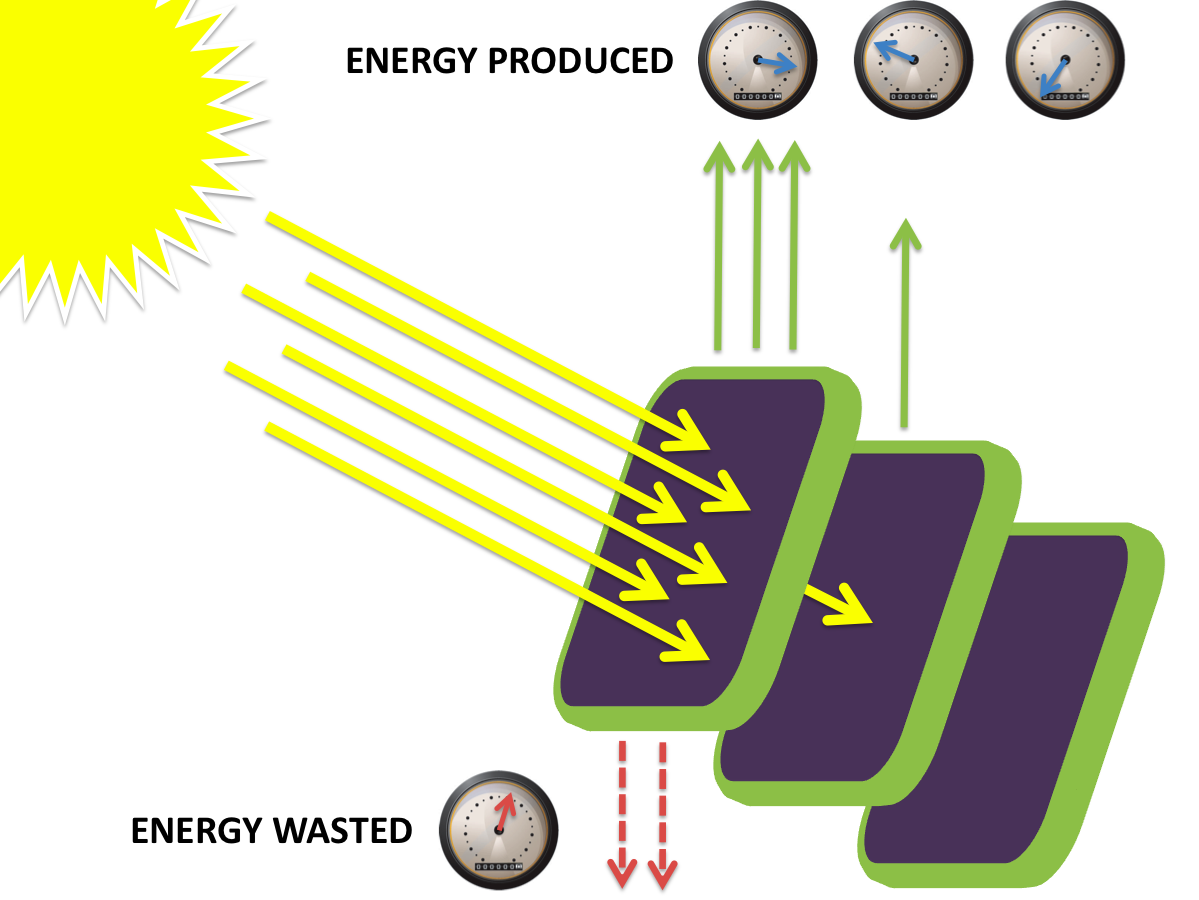
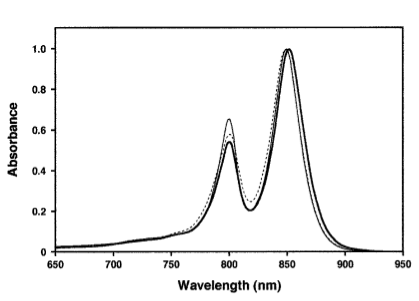
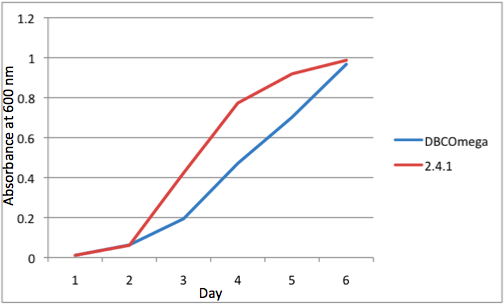
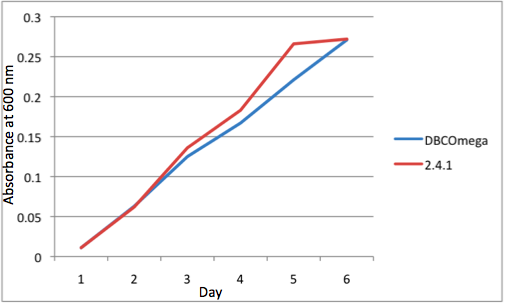
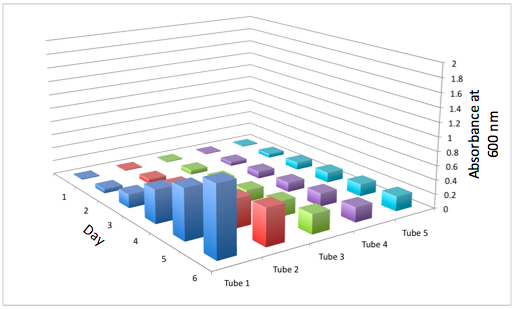
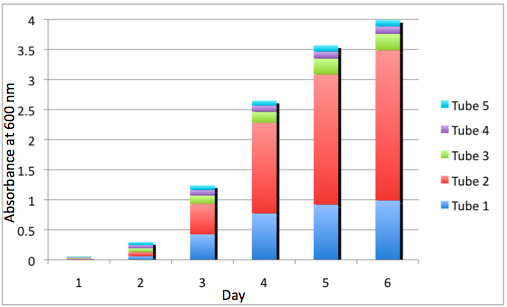
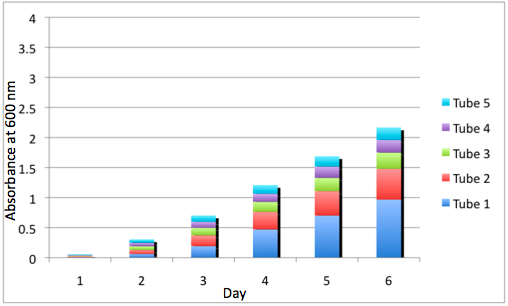
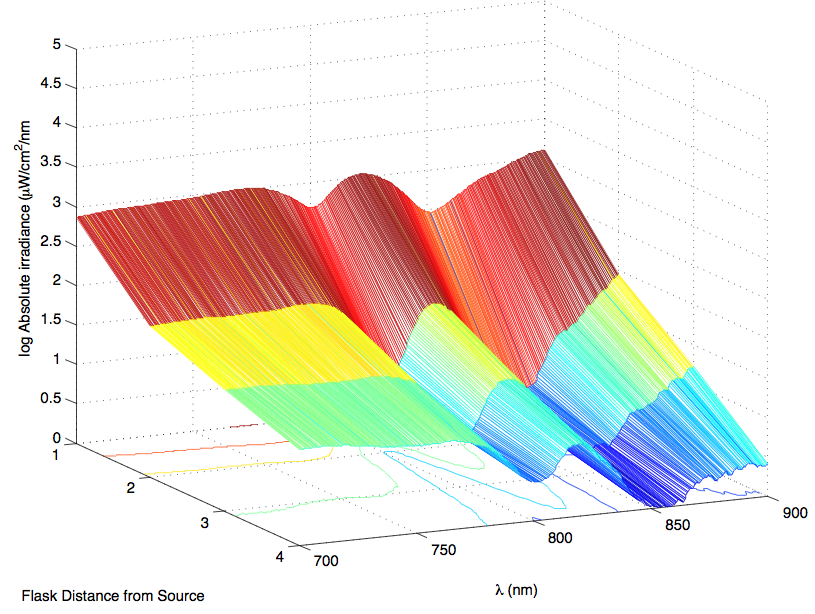
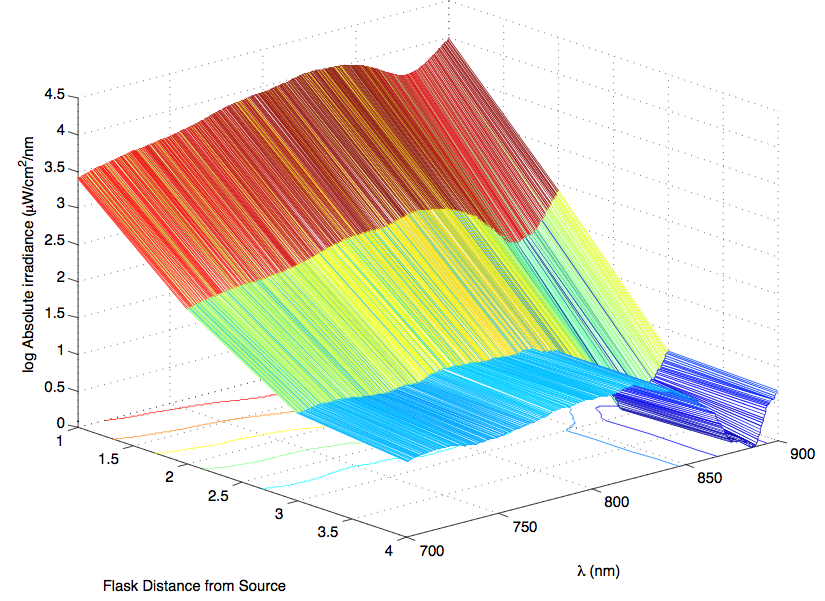
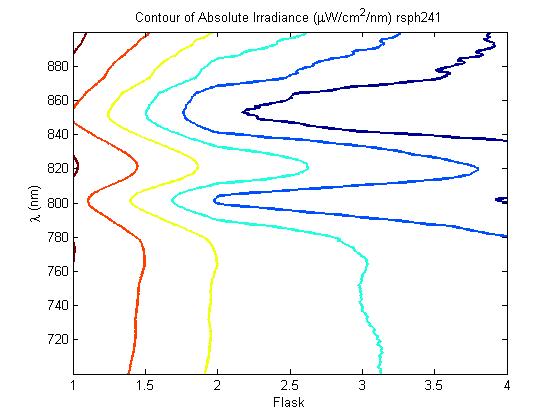
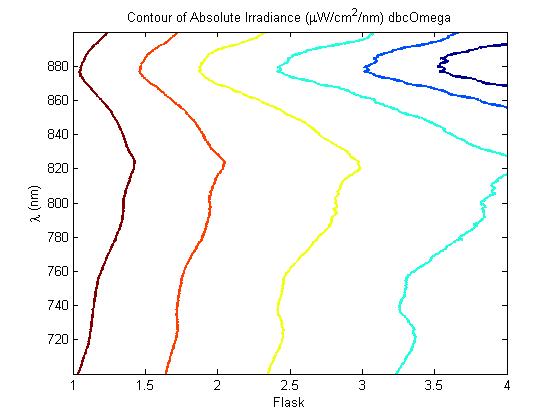
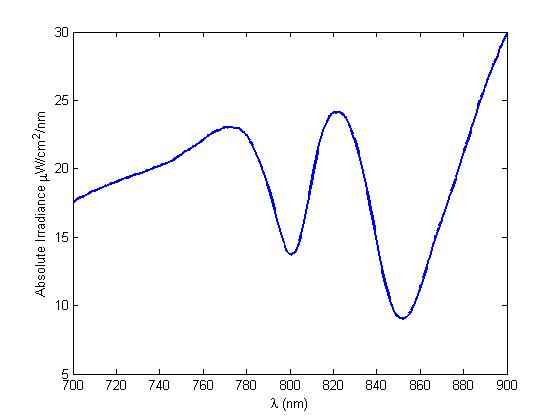
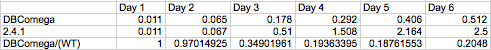
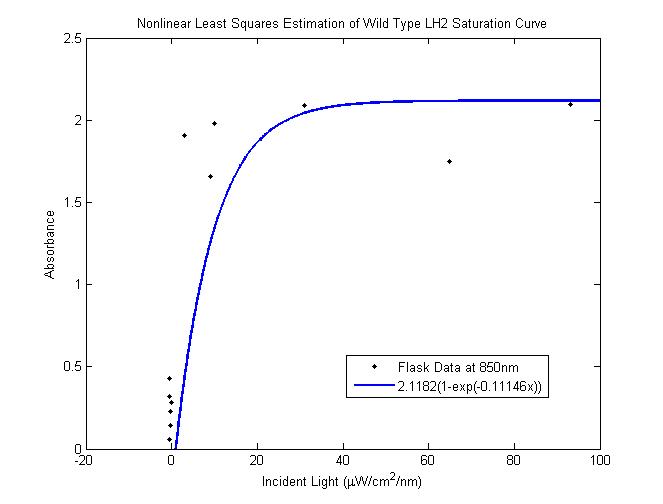
The most striking difference between the Wild Type Rhodobacter Sphaeroides 2.41 and DBCOmega (LH2 knockout) growth patterns is that the first flask in the WT (closest to the light source) grew less than the second flask, while the converse was true in DBCOmega (figure 2a and 2b). This can be attributed to the photosystem saturation curves for the respective cultures. The Wild Type has an LH2 complex, meaning that their antenna size is inherently larger and that their photosystem will be saturated at lower light intensities than the LH2 deficient mutant. We observed that the incident light intensity used in this experiment led to oversaturation for the first WT tissue flask and resulted in growth-inhibiting photodamage, as is evidenced by its lesser growth relative to the second culture. In contrast, photodamage was not observed in DBCOmega as is evidenced by the first tissue flask that grew at the fastest respective rate. Furthermore, it appears that this photodamage slowed the growth of the first WT tissue flask to the point that the OD of this tissue flask was nearly equal to that of DBCOmega after 5 days of growth(.987 vs. .967) (figure 1a). As such, the contribution of LH2 to the growth of the WT in flask 1 can be assessed to be little after day 3 when the growth curve slows- though, this light (at 800 and 850 nm) was still greatly absorbed by flask 1 (see irradiance figure for wt flask 1 day 6). It is likely that much of this light was then wasted through Non-Photochemical Quenching (NPQ), depleting the flasks behind flask 1 of photons at the LH2 absorbtion peaks and decreasing the cumulative productivity of flasks 1-5. This effect of wasting photons though NPQ under high light intensities is one that we sought to minimize in the design of our synthetic regulation for the pucB/A genes.
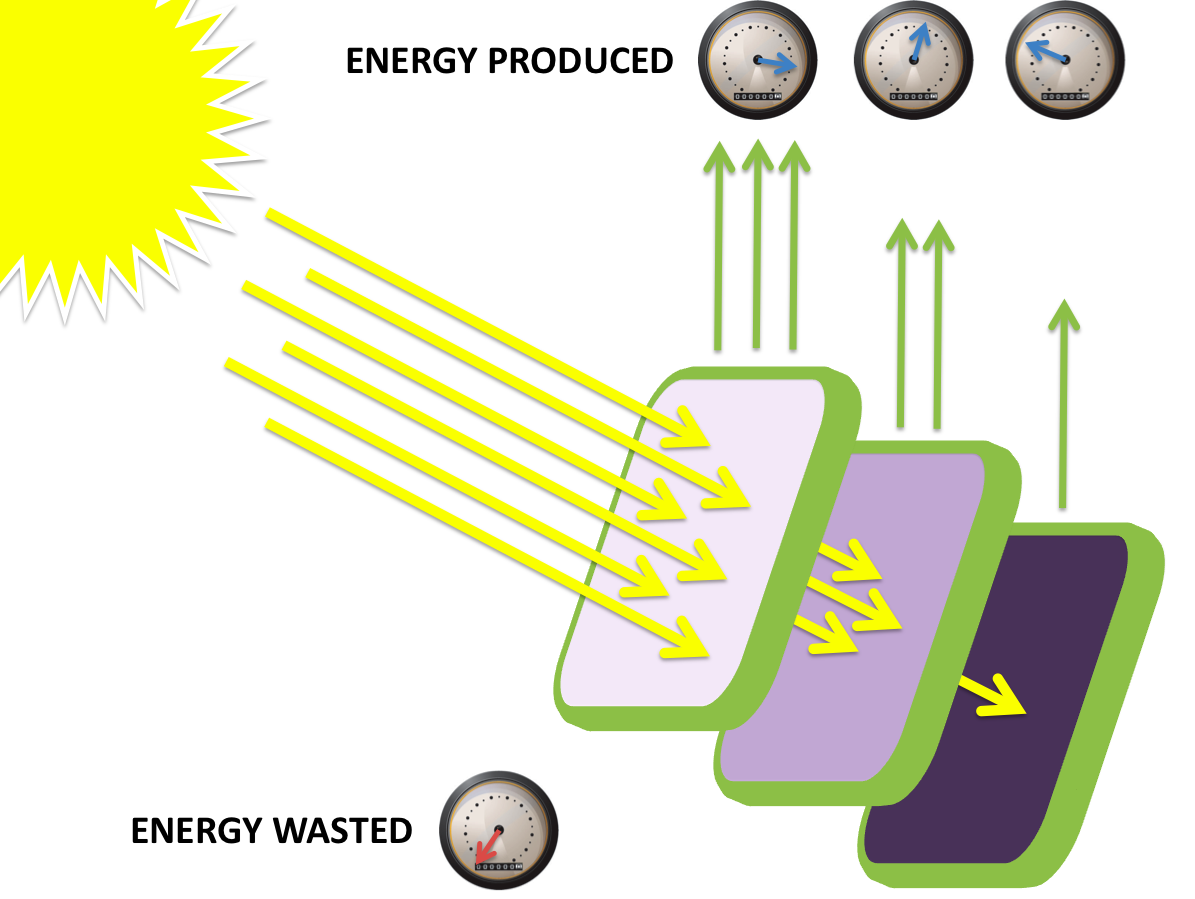
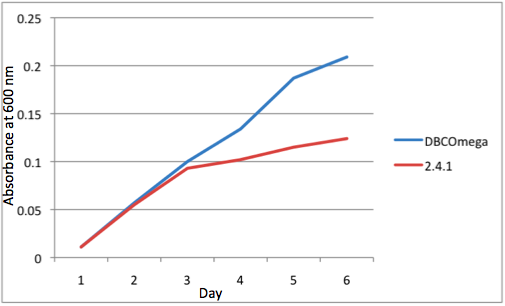
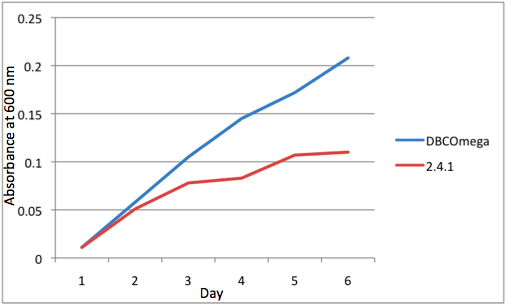
A further a comparison of the WT and DBCOmega experiments reveals that the 3rd tissue flask in each had almost identical growth rates (figure 1c), while the DBCOmega 4th and 5th tissue flask outperformed that of the WT (figure 1d and 1e). This can simply be attributed to the higher density of cells in the 2nd flask of the WT that led to an overall lesser quantity of photons passing through to the 4th and 5th flasks in the WT experiment and likely resulting in heterotrophic growth (though this will need to be confirmed with a "dark control" experiment that should be completed in time for the Jamboree). This decrease in available photons for the WT can be seen by viewing the absolute irradiance data (incident light intensity at a given wavelength) after the third culture for the WT (after flask 2 WT Irradiance day 3). This decrease in irradiance is the most pronounced for the LH2 absorption bands at 800 and 850 nm, as was expected. Our synthetic regulation system for pucB/A was designed with the intention of allowing greater penetration of light through the first couple of flasks at the LH2 wavelengths, as expression of pucB/A is inversely correlated to incident light intensity.
Thomas, add in some further comments about the sprectroradiometer stuff. Specifically about how it changes in the WT vs. that of DBCOmega using some specific examples on the graphs
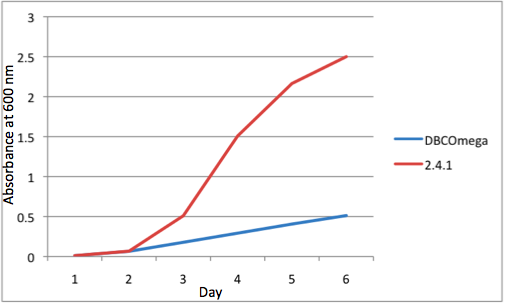
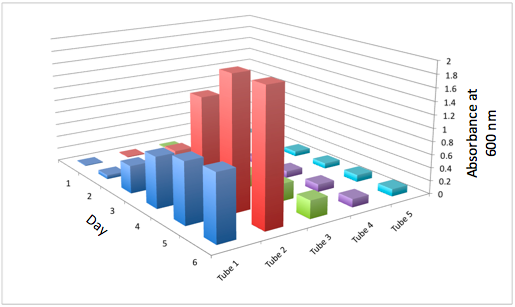
Overall, the cumulative culture growth of the WT exceeded that of DBCOmega (figure 3) due to the performance of the second tissue flask of the WT relative to that of DBCOmega (figure 1b) despite the photodamage that occurred in tissue flask 1 for the WT. This cumulative culture growth can be considered photosynthetic productivty and is proportional to the amount of energy/desired product that can be obtained from these cultures.
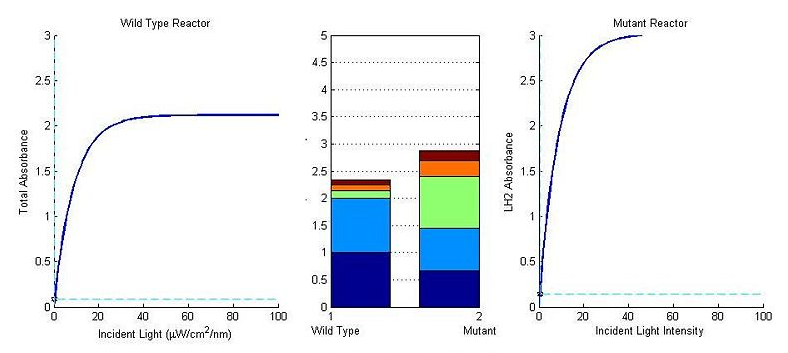
It is an interesting exercise to model how our mutant system would perform under these same conditions: Thomas add in some comments about referencing that diagram and what we took from this tissue flask experiment to construct that (and upload it when you are done with it).
add stuff here about the second flasks and approaching 20% contribution of LH1 to growth
Conclusion and Future Work
Based on model, should have had feedback mechanism- should be something that sets a minimum expression level for LH2, that is...
-tissue flask w prkcbc3 before jamboree and compare/interpret using characterization data
-tissue flask w. full mutant in future- if this doesn't reveal cph8 to responding in an exponentially decaying fashion to light intensity, try w. a system that would
-characterize puc promoter under light conditions and additional oxygen tensions
-complete other characterization work
- What actually happened?
- Ways to improve, redo parts of our experiment differently
- What further should be done to our end product to further its development, related back to what we initially tried to do?
- What are possible biofuel applications and how could our system be used to improve existing biofuels?
Back To Top
References
- "2007-2008 Catalog & Technical Reference." New England BioLabs Inc. (2007).
- Alon, Uri. Introduction to systems biology and the design principles of biological networks. Boca Raton, FL: Chapman & Hall, 2006.
- Ausubel, Fred et al. Short Protocols in Molecular Biology: Third Edition. Canada: John Wiley and Sons, Inc., 1999.
- "BioBrick Assembly Help." Registry of Standard Biological Parts. 2009. 6 July 2009. <http://partsregistry.org/Main_Page>.
- Bower, James M. Computational Modeling of Genetic and Biochemical Networks (Computational Molecular Biology). New York: M.I.T. PRESS, 2001.
- Braatsch, Stephan et al. "A single Flavoprotein AppA, Integrates Both Redox and Light Signals in Rhodobacter sphaeroides." Molecular Microbiology. Vol. 45.3 (2002): 827-836.
- Brown, Stanley B. et al. "Bile Pigment Synthesis in Plants: Incorporation of Haem into Phycocyanobilin and Phycocyanobiliproteins in Cyanidium Caldarium." Biochemistry. Vol. 194 (1981): 137-147.
- Dragnea, Vladimira et al. "Time-Resolved Spectroscopic Studies of the AppA Blue-Light Receptor BLUF Domain from Rhodobacter sphaeroides Biochemistry. Vol. 44 (2005): 15,978-15,985.
- "Entrez Nucleotide Search." National Center for Biotechnology Information. 2009. 6 June 2009 <http://www.ncbi.nlm.nih.gov/nuccore/49175990?from=3533887&to=3534606&report=gbwithparts>.
- Fowler, Gregory J. S. and C. Neil Hunter. "The Synthesis and Assembly of Functional High and Low Light LH2 Antenna Complexes from Rhodopseudomonas palustris in Rhodobacter sphaeroides." The Journal of Biological Chemistry. Vol. 271.23 (1996): 13,356-13,361.
- Gabrielsen, Mads et al. "Peripheral Complexes of Purple Bacteria." The Purple Phototropic Bacteria. (2009): 135-153.
- "GenElute HP Plasmid Miniprep Kit: User Guide." Sigma-Aldrich. (2008).
- Harwood, Caroline S. "Nitrogenase-Catalyzed Hydrogen Production by Purple Nonsulfur Photosynthetic Bacteria." Bioenergy. (2008): 259-271.
- Jager, Andreas et al. "The AppA and PpsR Proteins from Rhodobacter sphaeroides Can Establish a Redox-Dependent Signal Chain but Fail to Transmit Blue-Light Signals in Other Bacteria." Journal of Bacteriology. Vol. 189.6 (2007): 2,274-2,282.
- Jones, M. R. et al. "Mutants of Rhodobacter sphaeroides Lacking One or More Pigment-Protein Complexes and Complementation with Reaction-Centre, LH1, and LH2 Genes." Molecular Microbiology. Vol. 6.9 (1992): 1,173-1,184.
- Kondo, Toshihiko et al. "Enhancement of Hydrogen Production by a Photosynthetic Bacterium Mutant with Reduced Pigment." Journal of Bioscience and Bioengineering. Vol. 93.2 (2002): 145-150.
- Lagarias, J. Clark. PCB from Spirulina. Personal Communication. June 2009.
- Laible, Philip D. "Foreign Gene Expression in Photosynthetic Bacteria." The Purple Phototropic Bacteria. (2009): 839-860.
- Lee, Jeong K. and Samuel Kaplan. "Transcriptional Regulation of puc Operon Expression in Rhodobacter sphaeroides." Journal of Biological Chemistry. Vol. 270.35 (1995): 20,453-20,458.
- Levskaya, Anselmetal et al. "Engineering Escherichia coli to See Light." Nature. Vol. 438 (2005): 441-442.
- "Life Sciences Catalog 2009-2010." National Diagnostics. (2009).
- Melis, Anastasios. "Solar Energy Conversion Efficiencies in Photosynthesis: Minimizing the Chlorophyll Antennae to Maximize Efficiency." Plant Science. (2009).
- Mizuno, Takeshi and Shoji Mizushima. "Characterization by Deletion and Localized Mutagenesis in Vitro of the Promoter Region of the Escherichia coli OmpC Gene and Importance of the Upstream DNA Domain in Positive Regulation by the OmpR Protein." Journal of Bacteriology. Vol. 168.1 (1986): 86-95.
- Moskvin, Oleg V. et al. "Transcriptome Analysis of the Rhodobacter sphaeroides PspR Regulon: PspR as a Master Regulator of Photosystem Development." Journal of Bacteriology. Vol. 187.6 (2005): 2,148-2,156.
- Mussgnug, Jan H. et al. "Engineering photosynthetic light capture: impacts on improved solar energy to biomass conversion." Plant Biotechnology Journal Vol. 5 Issue 6. (2007) pp. 802-814
- "NEBCutter V2.0." New England BioLabs. 2009. 6 June 2009 <http://tools.neb.com/NEBcutter2/index.php>.
- "OligoAnalyzer 3.1." Integrated DNA Technologies. 2009. 6 June 2009. <http://www.idtdna.com/analyzer/Applications/OligoAnalyzer/>
- Sambrook, Joseph and David W. Russell. Molecular Cloning: A Laboratory Manual. Vol. 1-3. New York: Cold Springs Harbor Laboratory Press, 2001.
- Scheuring, Simon and Sturgis, James N. "Chromatic Adaptation of Photosynthetic Membranes" Science Vol. 309 (2005) pp.484-487
- Sener, Melih K. et al. "Atomic-level structural and functional model of a bacterial photosynthetic membrane vesicle" Proceedings of the National Academy of Sciences of the United States of America. Vol. 104, Number 40,pp. 15723-15728
- Shimizu-Sato, Sae et al. "A Light-Switchable Gene Promoter System." Nature Publishing Group. Vol. 20 (2002): 1,041-1044.
- Smith, Harry and M. Geoffrey Holmes. Techniquies in Photomorphogenesis. London: Academic Press, Inc., 1984.
- System modeling in cellular biology from concepts to nuts and bolts. Cambridge, MA: MIT P, 2006.
- Terry, Matthew J. "Biosynthesis and Analysis of Bilins." Heme, Chlorophyll, and Bilins: Methods and Protocols. (2002) 273-291.
- Wang, Wanneng et al. "Heterologous Synthesis and Assembly of Functional LHII Antenna Complexes from Rhodovulum sulfidophilum in Rhodobacter sphaeroides Mutant." Molecular Biology Reports. Vol. 12.3 (2008).
- Walz, Thomas et al. "Projection Structures of Three Photosynthetic Complexes from Rhodobacter sphaeroides: LH2 at 6 A, LH1 and RC-LH1 at 25 A" Journal of Molecular Biology Vol. 282 pp. 833-845
- Yakovlev, A. G. et al. "Light Control Over the Size of an Antenna Unit Building Block as an Efficient Strategy for Light Harvesting in Photosynthesis." Federation of European Biochemical Societies. (2002):129-132.
Back To Top

 "
"