Team:Valencia/Modelling
From 2009.igem.org
| Line 33: | Line 33: | ||
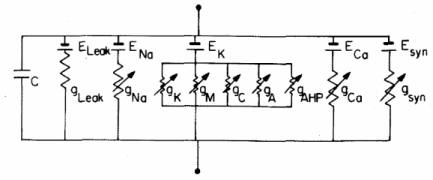
Now, we have to model how transmembrane potential changes in time. To do this, we can consider the following membrane-equivalent electrial circuit, where all ionic currents involved in initiation and propagation of the action potential are represented: | Now, we have to model how transmembrane potential changes in time. To do this, we can consider the following membrane-equivalent electrial circuit, where all ionic currents involved in initiation and propagation of the action potential are represented: | ||
| + | |||
| + | [[Image:V_circuit.gif|center]] | ||
</span> | </span> | ||
Revision as of 19:40, 12 August 2009
Modelling
Our aim in this part of the Project is the development of a model which describes how intracellular calcium concentration changes in time when we apply electrical stimulation, this is, a potential difference across the plasma membrane. We have considered very interesting to make different approaches to this problem: on the one hand, a deterministic model of the calcium influx through the voltage-dependent calcium channels (VDCCs) of excitable cells (neurons and muscle cells) and yeasts -based on the Hodgkin-Huxley model modified by Koch et. al.-, and, on the other hand, we have included stochastic methods for a further study of these gates, particularly of its activation/inactivation. We are working hard to offer you this model in an easy and clear way, also trying to allow you to interact with the system.
- What are VDCCs?
Living cells are surrounded by impermeable membranes containing specialized proteins providing for exchange of various atoms and molecules between extracellular and intracellular spaces. Two basic mechanisms of transmembrane transport have been recognized: carriers and channels.
Carriers, such as the Ca2+ pump, Na+-Ca2+ exchanger, or Na+-K+ pump, transport ions against concentration and/or electrical gradients are coupled to metabolic energy consumption. Membrane channels are viewed as pores, which, when opened, allow passive transport downhill the electric and/or concentration gradients. Opening of a channel can be accomplished in two ways:
- by binding of a specific ligand either directly to the channel or to another membrane protein coupled to the channel
- by a change in transmembrane voltage.
The first pathway is characteristic for ligand-gated channels, such as the glutamate or acetylcholine receptors. The second pathway activates the so-called voltage-gated channels. The foundation of biophysical analyses of voltage-gated ion channels was laid in the pioneering works of Hodgkin and Huxley in the 1930’s and culminated in the 1950’s by formulating the Hodgkin-Huxley model of action potential (Hodgkin and Huxley 1952). Initially, the Na+ and K+ selective voltage-gated channels and a leakage channel were recognized. Later, the family of voltage-gated ion channels was extended to include Ca2+ and Cl- channels.
Voltage-gated calcium channels were first identified by Fatt and Katz (1953) in crustacean muscle. Then it was discovered that there are different channel subtypes in excitable cells and, some years later, it was accepted that there are analog calcium channels in yeast plasma membrane.
- Modelling Ionic Current Flow through VDCCs
If we assume that the whole calcium currents occur through this calcium channels and that the instantaneous current-voltage relation is linear, we can describe the ionic current I by the Ohm's law:
Where g is the conductance associated with the channel, V is the transmembrane potential and E is the Nerst potential, related to the different ionic concentration inside and outside the cell.
Considering that these channels are only permeable to calcium and have two states -open or closed-, the total conductance associated with the population of VDCCs can be expressed as the maximal conductance g' times the fraction of all channels that are open. This fraction is determined by hypothetical activation and inactivation variables m and h, which depend on voltage and time:
Now, we have to model how transmembrane potential changes in time. To do this, we can consider the following membrane-equivalent electrial circuit, where all ionic currents involved in initiation and propagation of the action potential are represented:
 "
"