Team:Groningen/Project/Accumulation
From 2009.igem.org
m (→Arsenic - ArsR) |
m (OpG as a fraction.) |
||
| Line 166: | Line 166: | ||
<dt>Unbound substances</dt> | <dt>Unbound substances</dt> | ||
<dd> | <dd> | ||
| - | OpG = <span id=" | + | OpG / OpG<sub>total</sub> = <span id="OpGFraction"></span><br/> |
ArsR = <span id="ArsRConcentration"></span> µM<br/> | ArsR = <span id="ArsRConcentration"></span> µM<br/> | ||
</dd> | </dd> | ||
| Line 200: | Line 200: | ||
// Intermediates (mostly useful for debugging) | // Intermediates (mostly useful for debugging) | ||
| - | var | + | var OpGFractionNode = document.getElementById("OpGFraction"); |
var ArsRNode = document.getElementById("ArsRConcentration"); | var ArsRNode = document.getElementById("ArsRConcentration"); | ||
//var ArsRAs3Node = document.getElementById("ArsRAs3Concentration"); | //var ArsRAs3Node = document.getElementById("ArsRAs3Concentration"); | ||
| Line 245: | Line 245: | ||
// Set intermediates if they exist | // Set intermediates if they exist | ||
| - | if ( | + | if (OpGFractionNode) setOutput(OpGFractionNode, OpG/OpGT); |
if (ArsRNode) setOutput(ArsRNode, ArsR* 1e6); | if (ArsRNode) setOutput(ArsRNode, ArsR* 1e6); | ||
//if (ArsRAs3Node) setOutput(ArsRAs3Node, ArsRAs3* 1e6); | //if (ArsRAs3Node) setOutput(ArsRAs3Node, ArsRAs3* 1e6); | ||
Revision as of 13:36, 19 August 2009
[http://2009.igem.org/Team:Groningen http://2009.igem.org/wiki/images/f/f1/Igemhomelogo.png]
|
|---|
- Transport
- Accumulation
- Metal-sensitive Promoters
- Gas Vesicles
Introduction
Once heavy metals have entered the cell it is key to keep them there. As these metals are toxic to cell survival in critical amounts evolution has provided us with biological detoxicification proteins such as [http://en.wikipedia.org/wiki/Metallothionein metallothioneins]. These proteins can aid us in our quest to accumulate a variaty of heavy metals as they bind to a wide range of metals including cadmium, zinc, mercury, copper, arsenic, silver, etc..
Metallothioneins
Metallothioneins are a class of low molecular-weight metal-binding proteins rich in cysteines residues. They are capable of binding a variety of heavy metals. And they have readily been used to create cell based systems for purification of contaminated water Chen1998, Brady1994. In addition to their wide application possibilities they also have the capacity to carry multiple metal ions at one time, in contrast to some other metalloproteins that carry them one-on-one Chang1998. Many forms of metallothioneins are known and their affinity for different metals has been investigated on several occasions, such as for cadmium Deng2007, arsenic Ngu2006, Kostal2004, Singh2008, mercury Chen1998, Chen1997-2, Deng2008, nickel Deng2003 or a combination of metals Chang1998, Kao2008. Metal-protein complexes can be quantified using a fluorescent molecule Cadosch2008.
Metals
- As--> ordered pP-1 with rh-MT (human MT gene for accumulation of As3+) Ngu2006
- Vector properties unknown, the vector was found to be produced or so by GeneArt.
- Cu--> pBG68 with mymT (M. tuberculosis MT gene for Cu(I) accumulation) Gold2008
- Vector properties: pMB1 ori(20 copy nr.), M13 ori (? copy nr.), tagged with mxe-gyrA intein and chitin binding domain, produced from IPTG inducible T7 promoter (LacI also present).
- Zn--> pMHNR1.1 (a pET29a vector) with smtA (Cyanobacterial MT gene for Zn accumulation) Blindauer2001
Alternatives
Inclusion bodies Fowler1987
(Bacterio)Ferritins
Phytochelatins
[http://www.wiley.com/legacy/products/subject/reference/messerschmidt_toc.html A list of opportunities]
Inhibitory characteristics?
Modelling
Arsenic - ArsR
Below you can calculate how many grams of arsenic will be taken out of the water per cubic meter of cells. This extra weight raises the density of the cell and therefore lowers its capacity for buoyancy. Our preliminary results look very promising. Even under the assumption that the weight of the metal is added to the weight of the cells, without increasing their volume, we could add upto a hundred times the currently computed weight without having a large effect on the required fraction of gas vesicles (it will only go up from about 12.2% to 12.7%).
At this moment we use four different variables:
- Molecular weight of arsenic. Source: [http://en.wikipedia.org/wiki/Arsenic Arsenic page on Wikipedia]
- Millimol arsenic per kg of cell dryweight (note that this is equivalent to nmol/mg). Source: Kostal2004
- The proportion between the weight of a dry cell and a wet cell. Source: [http://redpoll.pharmacy.ualberta.ca/CCDB/cgi-bin/STAT_NEW.cgi CCDB Database]
- Cell density. Source: see our gas vesicle page.
| As per cell volume = awAs * nAs(III) / Mcell(dry) * Mcelldrywet * rhocell mol As per cell volume = nAs(III) / Mcell(dry) * Mcelldrywet * rhocell |
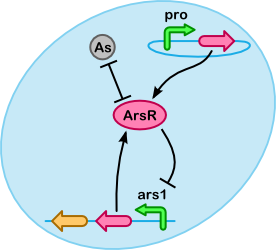
At a lower level arsenic accumulation can be described using reactions between ArsR, As(III) and the ars promoter. As shown in the figure on the right a number of different substances(/complexes) are involved. For our purposes it is especially important to determine what fraction of As(III) is unbound, if more As(III) is bound we can accumulate more.
The calculator below tries to compute the ratio between bound and unbound arsenic, specifically As(III), in the cell. See our Modelling page for detailed information on the constants/variables used and a derivation of the formulas. Note that the computations currently involve slightly more variables/constants than strictly necessary. TODO: The half-lifes were guesses based on cell-division, but since we have "resting" cells which we assume do not divide this seems like a very bad guess, so we need a new guess?
|
In conclusion:
- Even at the accumulation levels of Koster et al. the amount of arsenic accumulated in E. coli is so little that it shouldn't matter much for the buoyant density (which normally is about 1100kg/m3).
- If you substitute constitutive promotors for ars promotors you can see that it is clearly advantageous to use constitutive promotors (they give a much higher increase in accumulation).
- The model is not very sensitive to different values for K3d (with K3d=1M the accumulation factor is 7905.0 and with K3d=10-12M it is 7188.0).
Planning and requirements:
- Modelling
- Speed
- Metaliotheines concentration
- How often does the ArsR sensitive operator/operon occur in our E. coli?
- Lab
- Measurements
- Transport Assays
- Measure accumulation. By measuring before/after concentration metal with and without accumulation protein.
- Determine the dissociation constant of ArsR and As(III). (By measuring the ratio between bound and unbound ArsR?)
- It might be possible to do this with (tryptophan related) fluorescence (that is how it is done for ArsD in Chen1997). In the paper ArsD is purified, but if that's not feasible for us we might try to simply do it in living cells (and hope that ArsR both fluoresces enough and is produced enough to be measurable).
- Production rate of ArsR?
- Biobrick Bba_K129004
- Rest
- Measurements
 "
"