Team:Washington
From 2009.igem.org
| Line 1: | Line 1: | ||
__NOTOC__ | __NOTOC__ | ||
{{Template:Team:Washington/Templates/Header}} | {{Template:Team:Washington/Templates/Header}} | ||
| - | |||
<html> | <html> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
<img id="mainimg" src="/wiki/images/4/41/Main_graphic.png" alt="IPP System Overview" usemap="#cellmap" /> | <img id="mainimg" src="/wiki/images/4/41/Main_graphic.png" alt="IPP System Overview" usemap="#cellmap" /> | ||
<map name="cellmap"> | <map name="cellmap"> | ||
| Line 35: | Line 29: | ||
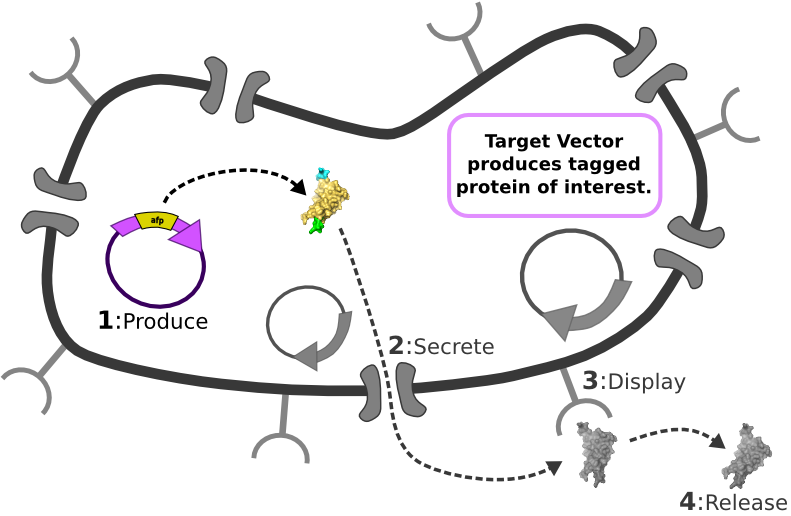
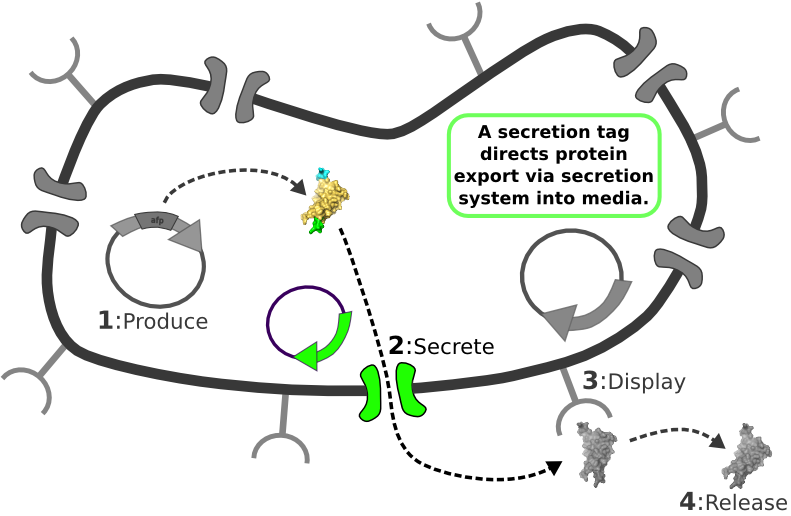
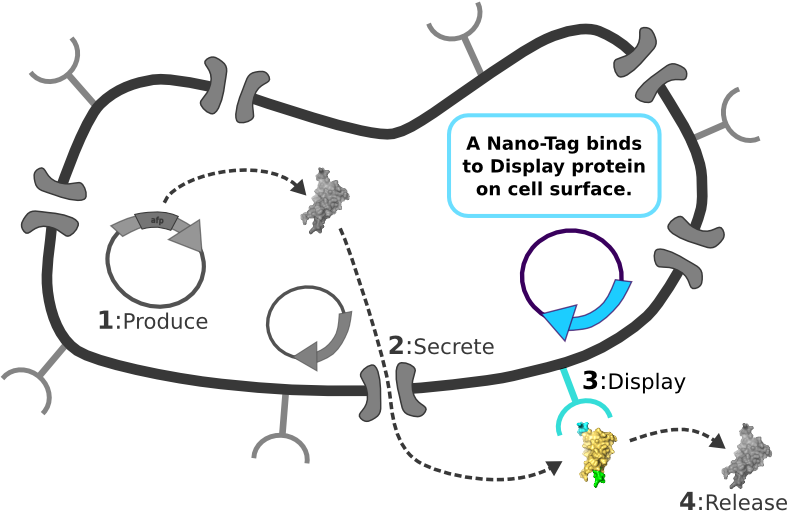
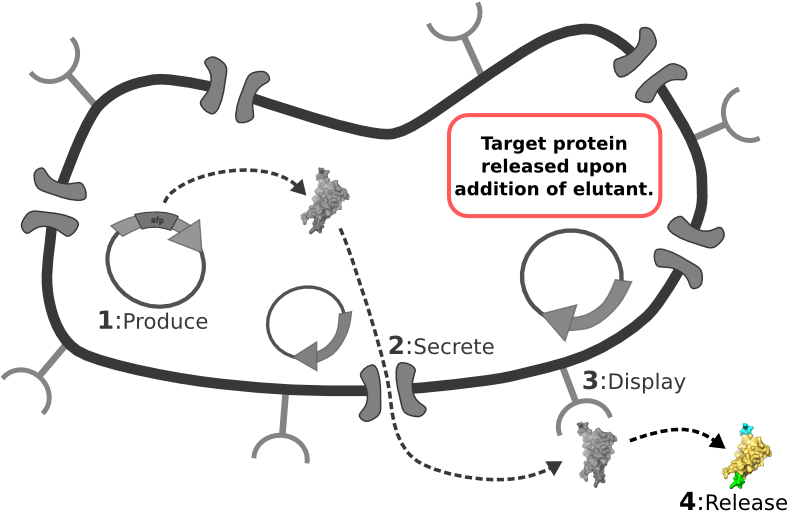
[4] Competing binder for surface protein releases protein of interest into supernatant. | [4] Competing binder for surface protein releases protein of interest into supernatant. | ||
| - | |||
| - | |||
| Line 59: | Line 51: | ||
|- | |- | ||
|} | |} | ||
| + | |||
| + | {{Template:Team:Washington/Templates/Footer}} | ||
Revision as of 07:07, 8 October 2009
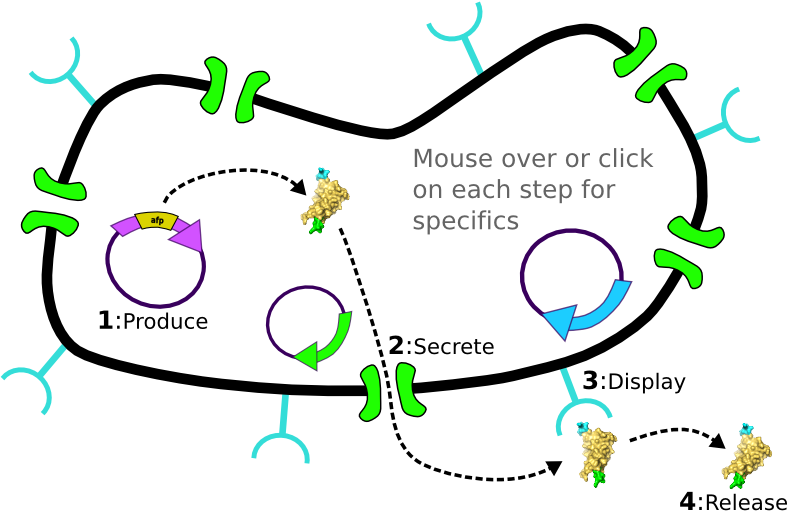
|
[1] Target Expression Vector produces fusion protein of interest. [2] Secretion Tag on fusion protein is recognized by secretion system and transported to extracellular space. [3] Nano-Tag on fusion protein is recognized by protein on cell surface and binds non-covalently to cell. [4] Competing binder for surface protein releases protein of interest into supernatant.
|
 "
"