Team:HKU-HKBU/polar expression results
From 2009.igem.org
YinanZhang (Talk | contribs) |
YinanZhang (Talk | contribs) |
||
| Line 5: | Line 5: | ||
='''Strain Selecion'''= | ='''Strain Selecion'''= | ||
| - | + | ==Swimming ability assay== | |
| - | + | The swimming plates of ''BL21'', ''NCM3722'' and ''MG1655'' yielded negative results. Although the clouded area of the MG3 plate showed augmentation in diameter, the colony was of the shape of dispersed clouds whereas a positive result would show distinct circles as a result of chemotaxis. We attributed this to contamination by other bacteria. ''Salmonella'' - a bacteria with well known swimming abilities (~4.5mm/hr) - was employed as our positive control. However, surprisingly, ''E.coli 2443'' showed even more impressive performance, with a speed of approximate 5.5mm increase in radius every hour at the end of eight-hour-experiment. | |
| - | The | + | |
| + | ==LPS completeness search== | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
LPS takes vital part in specifically expressing streptavidin, the protein enabling the binding of bacteria to motor in our project. After literature reviews, ''E. coli 2443'' [[#Reference | [1]]] and ''Salmonella'' [[#Reference | [2]]] are identified to possess complete LPS layer. Their ability to express desirable proteins on the head is examined in later experiment. | LPS takes vital part in specifically expressing streptavidin, the protein enabling the binding of bacteria to motor in our project. After literature reviews, ''E. coli 2443'' [[#Reference | [1]]] and ''Salmonella'' [[#Reference | [2]]] are identified to possess complete LPS layer. Their ability to express desirable proteins on the head is examined in later experiment. | ||
| - | |||
| - | |||
==Reference== | ==Reference== | ||
Revision as of 15:00, 14 October 2009
Contents |
Strain Selecion
Swimming ability assay
The swimming plates of BL21, NCM3722 and MG1655 yielded negative results. Although the clouded area of the MG3 plate showed augmentation in diameter, the colony was of the shape of dispersed clouds whereas a positive result would show distinct circles as a result of chemotaxis. We attributed this to contamination by other bacteria. Salmonella - a bacteria with well known swimming abilities (~4.5mm/hr) - was employed as our positive control. However, surprisingly, E.coli 2443 showed even more impressive performance, with a speed of approximate 5.5mm increase in radius every hour at the end of eight-hour-experiment.
LPS completeness search
LPS takes vital part in specifically expressing streptavidin, the protein enabling the binding of bacteria to motor in our project. After literature reviews, E. coli 2443 [1] and Salmonella [2] are identified to possess complete LPS layer. Their ability to express desirable proteins on the head is examined in later experiment.
Reference
- Sumita Jain, Peter van Ulsen, Inga Benz, M. Alexander Schmidt, Rachel Fernandez, Jan Tommassen, and Marcia B. Goldberg, Polar Localization of the Autotransporter Family of Large Bacterial Virulence Proteins, Journal of Bacteriology, July 2006, p. 4841-4850, Vol. 188, No. 13
- Maurien M. A. Olsthoorn, Bent O. Petersen, Siegfried Schlecht, Johan Haverkamp, Klaus Bock, Jane E. Thomas-Oates and Otto Holst, Identification of a Novel Core Type in Salmonella Lipopolysaccharide, The Journal of Biological Chemistry, Vol. 273, No. 7, Issue of February 13, pp. 3817-3829, 1998
Polar Expression
E. coli polar expression system
Fluorescent Microscope
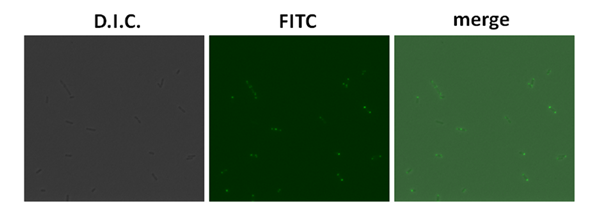
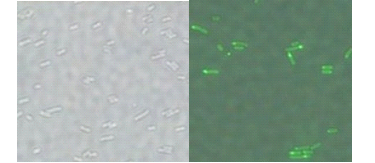
Plasmid in the BL21 strain showed the polar expression of GFP protein under the fluorescent microscope. In the merge picture, the fluorescent proteins were showed at one end of the bacteria.
However, in the E.coli 2443 with the plasmid sample, the polar expression of GFP couldn’t be observerd.
SDS PAGE and Western Blotting
Via SDS PAGE and Western Blotting, the relatively higher GFP protein concentration was observed on the outer membrane, which corresponded with the expected result of the specific expression of proteins on the outer membrane.
When observing the moving strains in the microscope, we could find that all the GFP were expressed at the forehead end of the rod shaped bacteria. Therefore, this system was a perfect choice for binding with biotin-coated motor. It can express streptavidin at pole(s), which allows cells to adhere in the same orientation to a microrotatory motor through biotin-streptavidin interaction.
Salmonella polar expression system
Fluorescent Microscope
plasmids showed the expression of GFP in the bacteria and the polar expression pattern of the GFP protein. The left panel is a scan of salmonella 7207 with Lpp-OmpA-HA, which acts as a control. The right panel can show the successful expression of GFP in the bacteria.
Western Blotting
To verify this hypothesis in a more convincing way, Western Blotting was conducted, which could reflect the surface expression by studying the relative concentrations of expressed proteins. The relative higher concentrations of specific proteins on the outer membrane show that the GFP proteins are particularly expressed on the outer membrane.
Unfortunately, the expression of GFP at the two ends is of equal opportunity, which couldn’t be an effective propelling system to push motor’s directional rotation. It’s better for proteins to specifically express proteins at the head of the bacteria, which ensures the pushing force to the motion of the motor.
 "
"