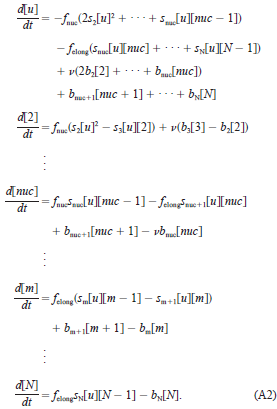Team:TorontoMaRSDiscovery/Modeling
From 2009.igem.org
| Home | The Team | The Project | Parts Submitted to the Registry | Modeling | Bioinformatics | Notebook |
|---|
Contents |
Summary
The objective for this years modelling work was to lay the foundations for future efforts.
A model has been constructed for our ideal construct design. We have used approximate parameter values gathered from online tools to represent all processes up to the production of eCFP and Encapsulin proteins. We have outlined the approaches that we will take for further modelling of eCFP degredation and Encapsulin assembly pathways.
We believe that our simulations may currently give fairly accurate predictions of eCFP and Encapsulin protein levels. In our initial experiments, we will use these predictions to identify a critical concentration of Encapsulin mononomer for Encapuslin assembly. Subsequently, we will incorportate existing models based on virus capsid assembly, and apply methods of parameter scanning to elucidate the mechanisms of Encapsulin microcompartment formation.
Model
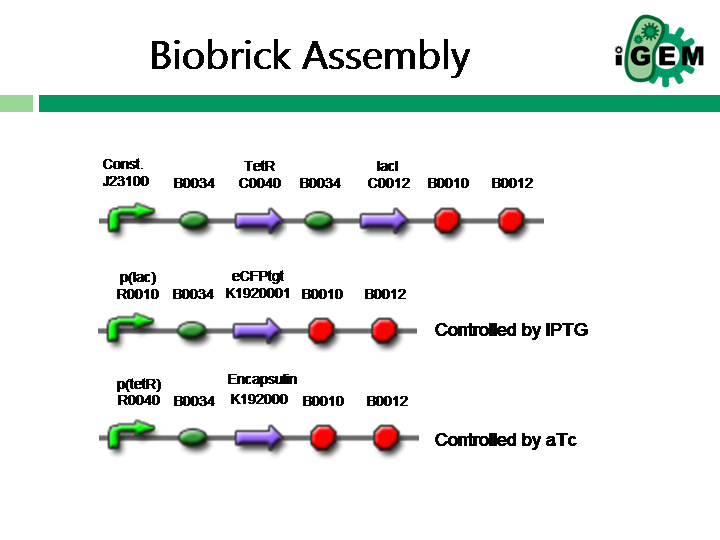
The ideal construct that needs to be modelled is shown below. However, our submission this year will not contain all parts as shown.
The construct is represented by the reactions shown below (reactions.xls). The reaction framework was mainly generated using the [http://synbioss.sourceforge.net/ SynBioSS Designer (a really useful modelling tool for iGEM teams)], which also provides "default" parameter values. These parameter values have been gathered from various literature [http://neptune.cems.umn.edu/designer/designer_defaults.pdf sources], and should be taken as approximations on the appropriate order of magnitude. All units were taken to be (1 / molarity^n-1 * s), where n is the order of the reaction. For example, for the reaction 2 lacI --> lacI2, n = 2, thus the rate constant has the units 1/molality*s.
The pathways for the expressed proteins, eCFPt and Encapsulin, need to be modelled in more detail. For example, in this basic model eCFPt is degraded via a first order reaction and Encapsulin makes a dimer complex. However, in reality fluorescent proteins have been shown to degreade via Micheales-menton kinetics and Encapsulin is thought to make a complex of about 60 monomers. These issues will be addressed in future work.
| Reaction | Rate Constant |
| Constitutive Expression | |
| --> lacl4 | 1E-10 |
| --> tetR2 | 1E-10 |
| Multimerization | |
| 2 lacI --> lacI2 | 1.00E+09 |
| lacI2 --> 2 lacI | 10 |
| 2 lacI2 --> lacI4 | 1.00E+09 |
| lacI4 --> 2 lacI2 | 10 |
| 2 tetR --> tetR2 | 1.00E+09 |
| tetR2 --> 2 tetR | 10 |
| 2 Enc --> Enc2 | 100 |
| Encapsulation | |
| Enc2 + eCFPt --> Enc2:eCFPt | 100 |
| Transcription | |
| RNAp + BBa_R0010 + LacI_binding_site --> RNAp:BBa_R0010:LacI_binding_site | 10000000 |
| RNAp:BBa_R0010:LacI_binding_site --> RNAp + BBa_R0010 + LacI_binding_site | 0.057 |
| RNAp:BBa_R0010:LacI_binding_site --> RNAp:BBa_R0010:LacI_binding_site* | 0.1 |
| RNAp:BBa_R0010:LacI_binding_site* --> RNAp:DNA_eCFPt + BBa_R0010 + LacI_binding_site | 30 |
| RNAp:DNA_eCFPt --> RNAp + mRNA_eCFPt | 0.035 |
| RNAp + BBa_R0040 + TetR_1 + TetR_2 --> RNAp:BBa_R0040:TetR_1:TetR_2 | 10000000 |
| RNAp:BBa_R0040:TetR_1:TetR_2 --> RNAp + BBa_R0040 + TetR_1 + TetR_2 | 0.057 |
| RNAp:BBa_R0040:TetR_1:TetR_2 --> RNAp:BBa_R0040:TetR_1:TetR_2* | 0.1 |
| RNAp:BBa_R0040:TetR_1:TetR_2* --> RNAp:DNA_Enc + BBa_R0040 + TetR_1 + TetR_2 | 30 |
| RNAp:DNA_Enc --> RNAp + mRNA_Enc | 0.0375 |
| Translation | |
| rib + mRNA_eCFPt --> rib:mRNA_eCFPt | 100000 |
| rib:mRNA_eCFPt --> rib:mRNA_eCFPt_1 + mRNA_eCFPt | 33 |
| rib:mRNA_eCFPt_1 --> rib + eCFPt | 0.1154 |
| rib + mRNA_Enc --> rib:mRNA_Enc | 100000 |
| rib:mRNA_Enc --> rib:mRNA_Enc_1 + mRNA_Enc | 33 |
| rib:mRNA_Enc_1 --> rib + Enc | 0.1234 |
| Protein-DNA | |
| lacI4 + nsDNA --> lacI4:nsDNA | 1000 |
| lacI4:nsDNA --> lacI4 + nsDNA | 1.6225 |
| lacI4 + LacI_binding_site --> lacI4:LacI_binding_site | 1.00E+09 |
| lacI4:LacI_binding_site --> lacI4 + LacI_binding_site | 0.005 |
| tetR2 + nsDNA --> tetR2:nsDNA | 1000 |
| tetR2:nsDNA --> tetR2 + nsDNA | 1.6225 |
| tetR2 + TetR_1 --> tetR2:TetR_1 | 1.00E+09 |
| tetR2:TetR_1 --> tetR2 + TetR_1 | 0.005 |
| tetR2 + TetR_2 --> tetR2:TetR_2 | 1.00E+09 |
| tetR2:TetR_2 --> tetR2 + TetR_2 | 0.005 |
| Protein - Effector - DNA | |
| lacI4 + IPTG --> lacI4:IPTG | 50000000 |
| lacI4:IPTG --> lacI4 + IPTG | 0.1 |
| lacI4:IPTG + LacI_binding_site --> lacI4:IPTG:LacI_binding_site | 1.00E+09 |
| lacI4:IPTG:LacI_binding_site --> lacI4:IPTG + LacI_binding_site | 0.7 |
| lacI4:LacI_binding_site + IPTG --> lacI4:IPTG:LacI_binding_site | 1000000 |
| lacI4:IPTG:LacI_binding_site --> lacI4:LacI_binding_site + IPTG | 0.4 |
| lacI4:IPTG + nsDNA --> lacI4:IPTG:nsDNA | 1000 |
| lacI4:IPTG:nsDNA --> lacI4:IPTG + nsDNA | 1.6225 |
| lacI4:nsDNA + IPTG --> lacI4:IPTG:nsDNA | 1000 |
| lacI4:IPTG:nsDNA --> lacI4:nsDNA + IPTG | 1.6225 |
| tetR2 + aTc --> tetR2:aTc | 50000000 |
| tetR2:aTc --> tetR2 + aTc | 0.1 |
| tetR2:aTc + TetR_1 --> tetR2:aTc:TetR_1 | 1.00E+09 |
| tetR2:aTc:TetR_1 --> tetR2:aTc + TetR_1 | 0.7 |
| tetR2:TetR_1 + aTc --> tetR2:aTc:TetR_1 | 1000000 |
| tetR2:aTc:TetR_1 --> tetR2:TetR_1 + aTc | 0.4 |
| tetR2:aTc + TetR_2 --> tetR2:aTc:TetR_2 | 1.00E+09 |
| tetR2:aTc:TetR_2 --> tetR2:aTc + TetR_2 | 0.7 |
| tetR2:TetR_2 + aTc --> tetR2:aTc:TetR_2 | 1000000 |
| tetR2:aTc:TetR_2 --> tetR2:TetR_2 + aTc | 0.4 |
| tetR2:aTc + nsDNA --> tetR2:aTc:nsDNA | 1000 |
| tetR2:aTc:nsDNA --> tetR2:aTc + nsDNA | 1.6225 |
| tetR2:nsDNA + aTc --> tetR2:aTc:nsDNA | 1000 |
| tetR2:aTc:nsDNA --> tetR2:nsDNA + aTc | 1.6225 |
| Degradation | |
| lacI4 --> | 0.000289 |
| tetR2 --> | 0.000289 |
| mRNA_eCFPt --> | 0.0015 |
| mRNA_Enc --> | 0.0015 |
| eCFPt --> | 0.000289 |
| Enc2 --> | 0.000289 |
| Enc2:eCFPt --> | 0.000289 |
| Dilution | |
| lacI4:nsDNA --> nsDNA | 0.000193 |
| tetR2:nsDNA --> nsDNA | 0.000193 |
| lacI4:IPTG --> IPTG | 0.000289 |
| tetR2:aTc --> aTc | 0.000289 |
| lacI4:IPTG:nsDNA --> IPTG + nsDNA | 0.000193 |
| tetR2:aTc:nsDNA --> aTc + nsDNA | 0.000193 |
Simulations
All simulations were carried out using the SimBiology Matlab Toolbox, which was freely available to iGEM teams.
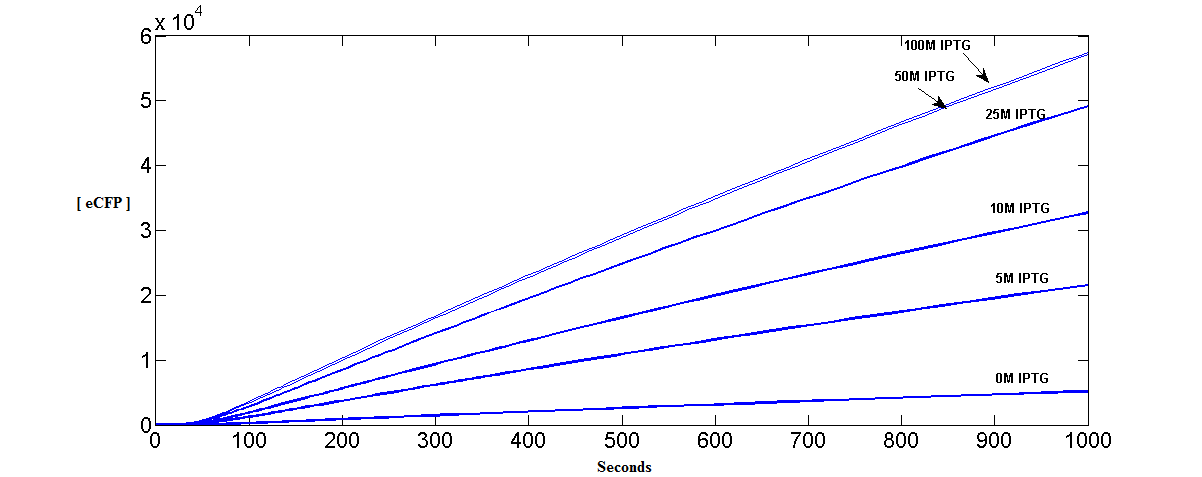
Accumulation of eCFP at Different Initial Concentrations of IPTG
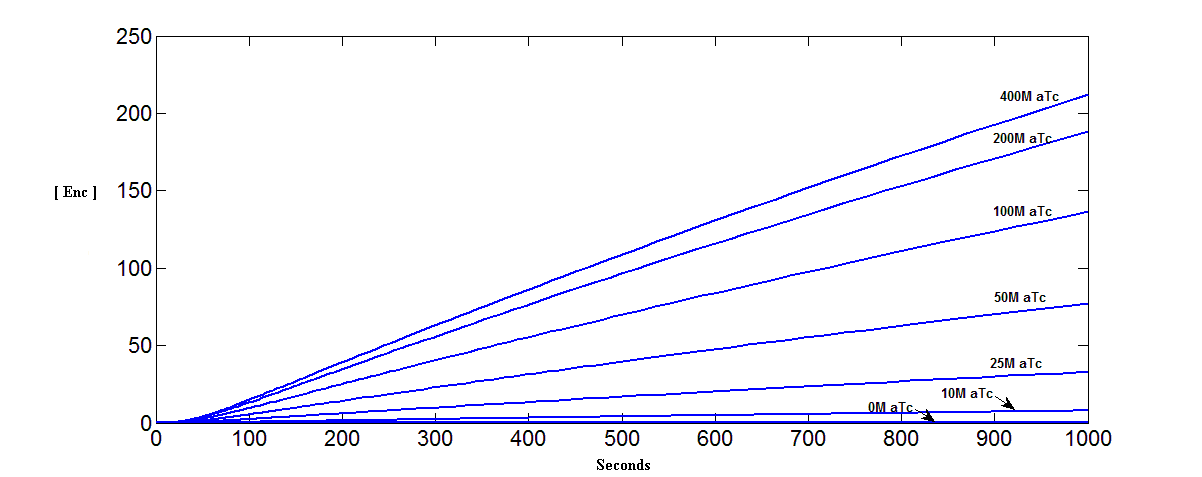
Accumulation of Encapsulin monomer at Different Initial Concentrations of aTc
Sensitivity Analysis
It is interesting to note which parameters have the greatest effect on the dynamics of the system. Since the parameters in our model are approximations from literature sources, the most sensitive parameters would be leading candidates to be experimentally determined.
(Currently, the SimBiology Toolbox reports an "OUT OF MEMORY" error when running a sensitivity analyis on our system).
Future Work
Improvements to the basic model presented above can be separated into three areas; 1) performing experiments to determine parameter values that specific to our system, 2) more detailed modeling of eCFPt behavior, 3) more detailed modeling of Encapsulin assembly.
The experiments required to determine the general transcription and translation parameters need to be further researched, so this will be skipped over for now. Here we will discuss the more interesting areas – the detailed modeling of eCFP and Encapsulin pathways. This will be accomplished by incorporating previous modeling efforts that are applicable to the eCFP and Encapsulin proteins.
Degredation of eCFPt
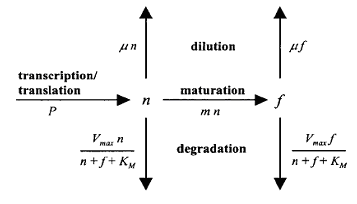
The expression and degradation of [http://www.pubmedcentral.nih.gov/picrender.fcgi?artid=95514&blobtype=pdf Green Fluorescent Protein (GFP) was modeled previously]. This paper noted that GFP must mature before going into fluorescent phase, and GFP degradation was shown to follow M-M kinetics, as depicted below. This paper also outlined how the degradation parameters could be derived for any expression system using only simple fluorescence and optical density measurements.
The GFP maturation and degradation parameters found in this paper were:
| m = 0.0004279 1/s |
| Vmax = 2E-10 molality/s |
| Km = 9E-13 molality/s |
Assuming eCFPt would behave similarly to eGFP, the following additional reactions would be included in the model:
| Maturation | |
| eCFPt --> F_eCFPT | 0.0004279 |
| Enc2:eCFPt --> Enc2:F_eCFPt | 0.0004279 |
| Encapsulation | |
| Enc2 + F_eCFPt --> Enc2:F_eCFPt | 100 |
| Degradation | |
| eCFPt --> | Vmax = 2E-10 |
| Km = 9E-13 | |
| F_eCFPt --> | Vmax = 2E-10 |
| Km = 9E-13 |
Assembly of Encapsulin microcompartments
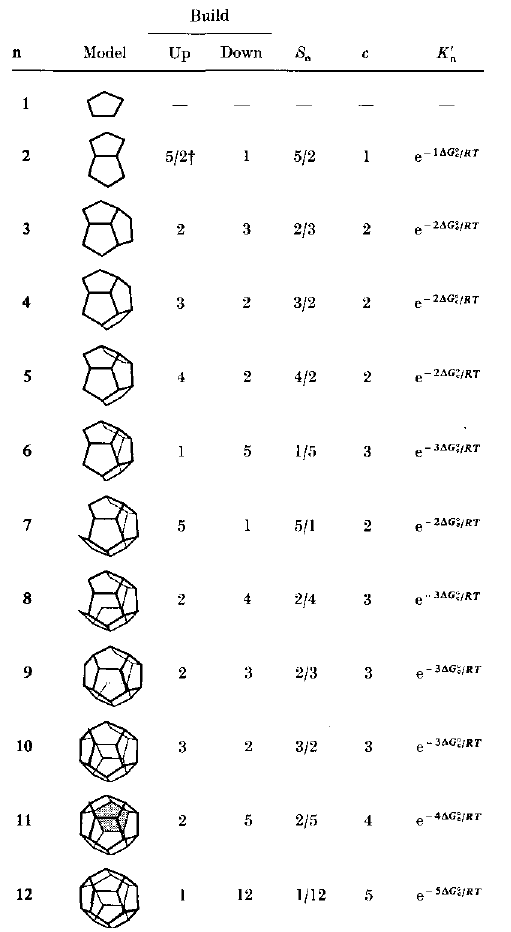
Though Encapsulin microcompartment assembly is not well-studied, it has been theorized to resemble the assembly of viral capsids. Thus, we could look to the modeling of viral capsid assembly, a relatively well-studied procedure, as a representation of Encapsulin assembly.
Adam Zlotnick has published models of viral capsid assembly using several different approaches, the most recent of which is [http://www.pubmedcentral.nih.gov/picrender.fcgi?artid=1302223&blobtype=pdf based on viral capsid assembly but is general enough to be applied to other examples of spherical polymerization.]
Model consists of reactions from an initial monomer subunit leading to a final capsid product. The sequential reactions take into account DeltaG for contact (Kn'), and parameters describing path degeneracy (varies for different intermediates).
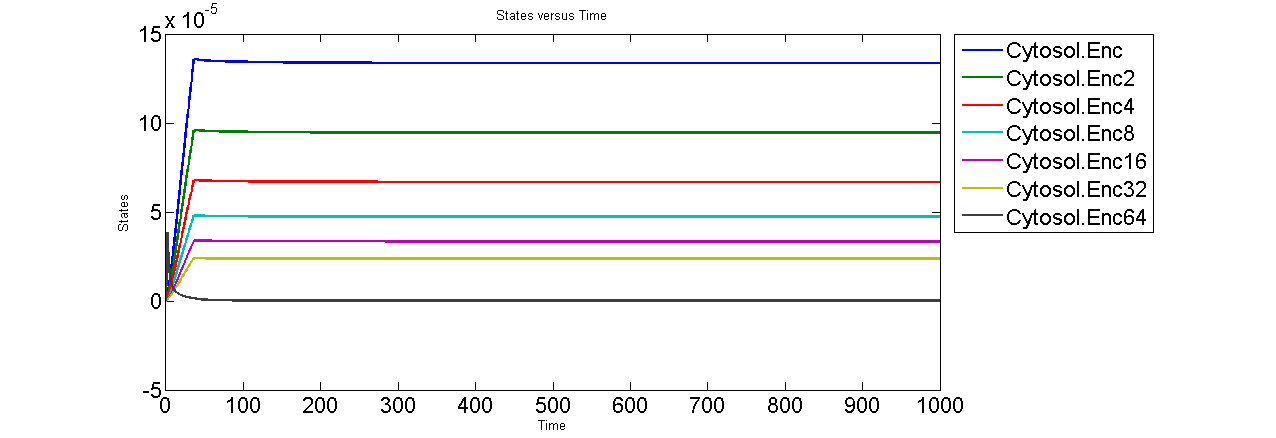
Simulations from a preliminary incorporation of this approach are shown below. In this version of our basic model, Encapsulin assembles in a series of first order reactions. However, for simplicity, the rate constants are kept identical and do not take path degeneracy into account.
| Encapsulin Assembly | |
| 2 Enc --> Enc2 | 1.00E9 |
| 2 Enc2 --> Enc4 | 1.00E9 |
| 2 En4 --> Enc8 | 1.00E9 |
| 2 Enc8 --> Enc16 | 1.00E9 |
| 2 Enc16 --> Enc32 | 1.00E9 |
| 2 Enc16 --> Enc64 | 1.00E9 |
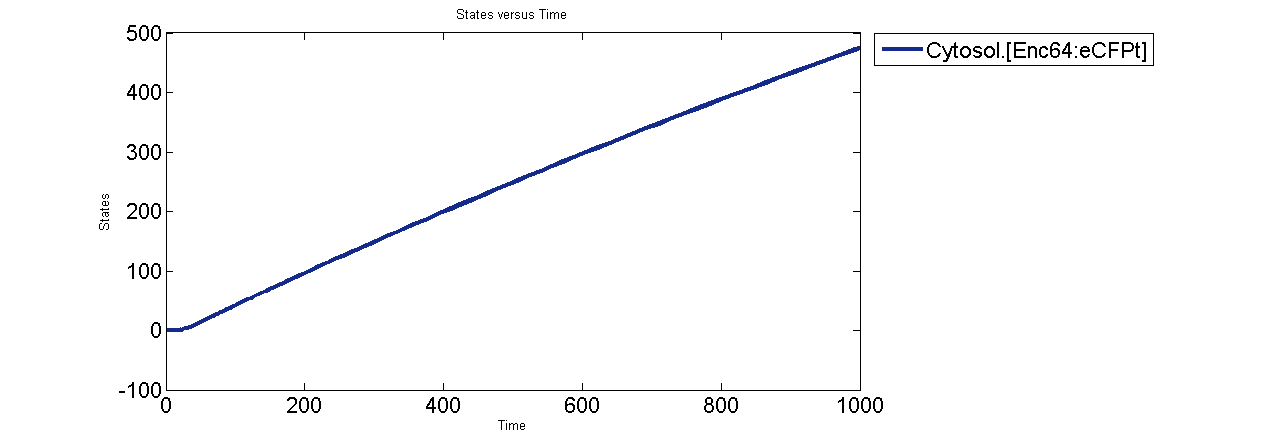
Encapsulin assembly intermediates are observed to quickly come to a steady state. The last intermediate Enc64 is the most depleted probably because of the formation of Enc64:eCFPt complex. Formation of Enc64:eCFPt then mirrors the production of eCFPt.
The Zlotnick model also describes two important features of spherical polymerization that we should also expect to observe in Encapsulin assembly, kinetic trapping and nucleation. Kinetic trap occurs when the deltaG or initial concentration of monomer subunit is too high, which results in over-initiation of assembly and quickly the reactions become starved for subunits. This is prevented by the inclusion of nucleation, where the initial reactions occur at a slower rate because the first few intermediates in a series of reactions are less stable than downstream products. Essentially, nucleation serves as a “slow first step” that regulates the assembly pathway.
It will be interesting to note how the translational dynamics of Encapsulin interplay with its assembly dynamics. Since our system does not start with any initial concentration of Encapsulin, there will be an optimum rate of production that essentially serves the role of nucleation – avoiding kinetic traps while still maintaining a maximum production of full Encapsulin assemblies.
The general model is presented as:
Where u is the initial subunit concentration (Enc in our case), m is the "mth" intermediate species, and N is the number of monomers in the final assembly.
All of the included parameters can be calculated from the four basic parameters; the microscopic, per-contact equilibrium constant (KAcon), the nucleus size (nuc), the nucleation on-rate (fnuc), and the elongation on-rate (felong). The paper describes methods to aquire these parameters from kinetic experiments, and provides examples for N = 12 and 30 unit viral capsid assemblies.
 "
"