|
Introduction
File:Modeling scheme.jpg
SynGenome Based GenSniper Production Modeling
In this part, we will use mathematical methods(by solve a set of ODEs with proper parameters) to predict the amount of GenSnipers we can get in a given time(the zero point for time is set as the time when IPTG is added in). This model can inform us whether our system is efficient in industry production.
Bottom-Up Strategy
diagram of dynamic process
In a simple model, the assembly of proteins are considered negligible compared with the time of transcription and translation of these proteins precursors. The amount of product is the smaller one of the amount of package proteins of phage and the amount of targeted DNA ( to be packaged by phage) per bacterial cell.
results
Beginning with 50 plasmid per cell, the protein amount comes into equilibrium between synthesis and degradation about 10 hours later. The steady state amount of package proteins can reach the amount of about 10 in the orders of 7, given the parameters as showed in the figure, which means it is enough for packaging target DNAs.
Since there are 420 major protein D and 420 major protein E on the surface of bacteriophage lambda viroin, the number of the theoretically packaged GenSniper viroin is 5.0 in the orders of 4. The wildtype bacteriophage lambda requires a lytic cycle of 40-45 min with a production rate of 100 viroins per cell. In contrast, the production model suggests that the yield of our synthesized gene therapy vector overweighs the wildtype lambda viroin production by 2 orders, while the lytic cycle seems to be longer and less violent. This point can be reflected in our wet lab work, where the actual IPTG induction time was about 10h.
Moreover, the accepted high efficiency gene therapy vector production system requires a titer of 1×10^10-1×10^12 genome/ml. Assuming that OD600≈2 means about 10^8 cells per ml in the liquid culture, and that very viroin should contain one genome, our Bottom-Up project can give rise to a titer of 5.0×10^12 Therapeutic DNA/ml, which meets the criteria for industrialization.
paratmeters
Protein tranaslation rate: r_Translation 0.0833/second
T7promoter transcription rate: T7_trsc 5.0/second
protein degradation rate: d_protein 1.93E-4/second
mRNA degradation rate: d_mRNA 0.00578/second
Top-Down Strategy
Q protein is a native λ phage protein which regulates λ phage's late gene transcription. In our system, Q protein is transcripted under regulation of T7 promomter then regulates the transcription of λ phage package protein through its anti-termination property to Pr' promoter. In this modeling section, we'll predict how much package proteins we will get using Pr'-Q regulatioin system.
reaction kinetics
the production of Q protein
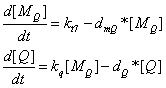
the production of target package proteins when Pr' is fully activated
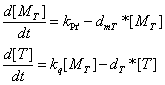
Q protein's influence on Pr's promoter activity
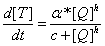
parameters
T7 promoter transcription rate kt7 = 5 molecule/second
Pr' promoter transcription rate kpr'= 20 molecule/second (estimated according to the fact that it is faster than T7)
mRNA degradation rate dm = 0.05 molecule/second
protein translation rate kq = 0.1 molecule/second
protein degradation rate dQ = 0.003 molecule/second
results
This is the Q protein level changing with time. When time= 1000s , 90% Q protein versus maximum amount haven been produced. At this time point, we assume that Pr' promoter is fully activated.
This figure is the Q protein level versus time, notice the zero point of time axis is set to the time point that Pr' promoter is fully activated.
Defining the time of adding IPTG as t=0, the simulated Q concentration increases with the structral proteins. When t≈2000s, the desired proteins reach a maximum and their growth rate decrease, when the lytic apparatus is designated to open. The entire lytic cycle lasts 35 min, which is much shorter than the Bottom-Up strategy and in the same order with the wildtype bacteriophage lambda production.
This modeling result matches our project design, where Top-Down approach uses the same transcription strategy (promoter R') as the wildtype lambda genome and Bottom-Up approach uses a engineered one (promoter T7).
At the steady state, the concentration of the structral proteins reach at 1.3×10^4. Assuming that there are 420 D or E major structral protein on the surface of one GenSniper viroin, the packaged viroin number is 3.0×10^1 per cell, which is near to the wildtype bacteriophage lambda packaging. When the density of the cells is 10 in the orders of 9, the titer of synthetic GenSniper will be 3.0×10^10 Therapeutic DNA/ml, which also meets the standard for industrialization.
Plasmid GenSniper Production Modeling
One of our subproject is the use nakes plasmid bound with a specified protein as the vector of gene therapy DNA. The normal replication process of plasmid will be disrupted due to the binding of specified protein(in our model,lambda repressor). If the protein has a high affinity with the plasmid we will have more fraction of plasmid protein complex however with less plasmid substrates. Considering production of the plasmid protein complex, we want to maxize the amounts of plasmid protein complex, so a balance between the amounts of plasmid and protein is needed. In this model, we want to find out the optimized parameters for this plasmid vector production process.
Reaction kinetics
Dimmer formation and dissociation between λ rep-fiber
parameters:
kdon =0.05/(M*sec) [1,6]
kdoff =0.5/sec
DNA protein complex’s binding and dissociation is a dynamical process
parameters:
kon =0.021/(mole*sec) binding rate of dimers to DNA
koff1 =0.04/sec unbinding rate of cI dimer from Or1
koff1 =1.026/sec unbinding rate of cI dimer from Or2
koff1 =5.197/sec unbinding rate of cI dimer from Or3
Notes:we get these values from [2]. Cao.etc estimate these values
from experimentally measured protein diffusion coefficient in e.coli and
Gibbs free energy. We give all the three unbinding rate of cI dimer from three kinds of binding sites to find out the best site we want to use in the experiment.
The replication and degration of plasmid
The plasmid is considered to replicate in a constant rate (r) without the interference of binding protein. Here the parameter reff means the actual replicating rate of plasmid taken the binding protein into consideration.
Notes:
plasmid replication is a complex process, here we use constant rate replication and degradation as a simplified model. Three general classes of regulatory mechanisms have been studied in depth, namely those that involve directly repeated sequences (iterons), those that use only antisense RNAs and those that use a mechanism involving an antisense RNA in combination with a protein. For more detailed info about Control of plasmid replication and copy-number: [http://www.sci.sdsu.edu/~smaloy/MicrobialGenetics/topics/plasmids/plasmid-repln.html] . An mathematical model is described in (12)
The transcription of λ rep-fiber, notice that λ rep-fiber is coded in the plasmid, so the copy number of plasmid will affect the amount of λ rep-fiber.
parameters:
υm = 0.033/s
kdm = 0.006/s
Notes:
The rate limiting step in ranscription including transcription initiation and elongation. The rate of transcript initiation reaction is 9.46*10^5 for cI (8,9). Average elongaration rate is 30 nt/sec. λ repressor is about 720bp, so υm = 24/720=0.033/s . The half lifetime of mRNA in e.coli is 2 min(7), kdm=ln2/2*60=0.006/s
The translation of λ repressor
parameters:
υp = 0.0288/sec
kd_cI_w = 0.0002/s (widetype)
kd_cI_tag = 0.003/s (with degradation tag)
Notes: An average ten copies of proteins are assumed to be produced per transcript. Given the half lifetime of mRNA of cI to be 4 min, υp = 10/(4*60/ln2)=0.0288/sec
The half-life of widetype λ repressor is 60 mins (10), however, with degradation tag, it could be as short as 4 min (11).
Plasmid-protein complex affect plasmid replication
If a replication is already initiated but when the replication forks meet a lamda protein on the way, this replication will be aborted. So only when replication forks pass the protein binding point without lamda protein there, the replication processes are effective. This is a simplified model on how plasmid-protein complex affect plasmid replication, we will try to find out some more detailed mechanisms in the future.
The probability of a plasmid to become a protein-DNA complex is:
Because of the DNA-protein complex, the effective replication rate of plasmid is
Bacterial DNA is replicated at a rate of about one million base pairs per minute,and the plasmid DNA we use is 8000+x bp(x is the length of theraputic DNA), so if x=2000bp, the average replication rate for plasmid is about 1.67/(mole*sec).
The amount of plasmid protein complex for a set of parameters
Molecular Docking for Targeted Biobrick
We used ZDOCK software to conduct the molecular docking day experiment. In the generated docking graph, the ligand (we used the crystalized fiber protein to substitute the targeted biobrick) and the receptor (integrin) are both in a stretched confirmation in which the spacial clash is relatively small. Moreover, in the fiber confirmation, the positions for both RGD domain and cell-specific peptide are exposed to the surface to faciliate the specific interaction with the cell specific receptor and integrin.
References
[1] Burz, D.S., et al., Self-assembly of bacteriophage lambda cI repressor: effects of single-site mutations on the monomer-dimer equilibrium. Biochemistry, 1994. 33(28): p. 8399-405.
[2] Cao, Y., H.M. Lu, and J. Liang, Stochastic probability landscape model for switching efficiency, robustness, and differential threshold for induction of genetic circuit in phage lambda. Conf Proc IEEE Eng Med Biol Soc, 2008. 2008: p. 611-4.
[3] Elowitz, M.B. and S. Leibler, A synthetic oscillatory network of transcriptional regulators. Nature, 2000. 403(6767): p. 335-8.
[4] Koblan, K.S. and G.K. Ackers, Cooperative protein-DNA interactions: effects of KCl on lambda cI binding to OR. Biochemistry, 1991. 30(31): p. 7822-7.
[5] Merabet, E. and G.K. Ackers, Calorimetric analysis of lambda cI repressor binding to DNA operator sites. Biochemistry, 1995. 34(27): p. 8554-63.
[6] Shea, M.A. and G.K. Ackers, The OR control system of bacteriophage lambda. A physical-chemical model for gene regulation. J Mol Biol, 1985. 181(2): p. 211-30.
[7] Kushner, S. R. in Escherichia Coli and Salmonella: Cellular and Molecular Biology (ed. Neidhardt, F. C.) (ASM, Washington DC, 1996).
[8] Li M, McClure W, Susskind M. Changing the mechanism of transcriptional activation by phage lambda repressor. Proc Natl Acad Sci U S A 1997;94(8):3691–3696. [PubMed: 9108039]
[9] Hawley D, McClure W. In vitro comparison of initiation properties of bacteriophage lambda wildtype pr and x3 mutant promoters. Proc Natl Acad Sci U S A 1980;77(11):6381–6385. [PubMed:
6450417]
[10] Keiler, K. C., Waller, P. R. & Sauer, R. T. Role of a peptide tagging system in degradation of proteins synthesized from damaged messenger RNA. Science 271, 990±993 (1996).
[11] Andersen, J. B. et al. New unstable variants of green ¯uorescent protein for studies of transient gene expression in bacteria. Appl. Environ. Microbiol. 64, 2240±2246 (1998).
[12] Brendel, V. and A.S. Perelson, Quantitative model of ColE1 plasmid copy number control. J Mol Biol, 1993. 229(4): p. 860-72.
|