Team:Washington/Project
From 2009.igem.org
Protein Purification
Purified proteins are an absolute necessity for aspiring synthetic biologists and iGEM participants alike, being used in many key laboratory processes. Though commonly used proteins like EcoRI, XbaI, SpeI, and PstI can be bought, those who research proteins and protein design, must purify their own proteins. Many of us have personal experience dully purifying protein, which makes the field as a whole slower and more tedious that it could be. For these reasons, the UW 2009 iGEM team has decided to design an Idealized Protein Purification (IPP) scheme that will speed up protein purification and possibly make previously difficult to purify proteins easier to work with, in the hopes to make synthetic biology a little faster, and a little more accessible.
Traditional Protein Purification
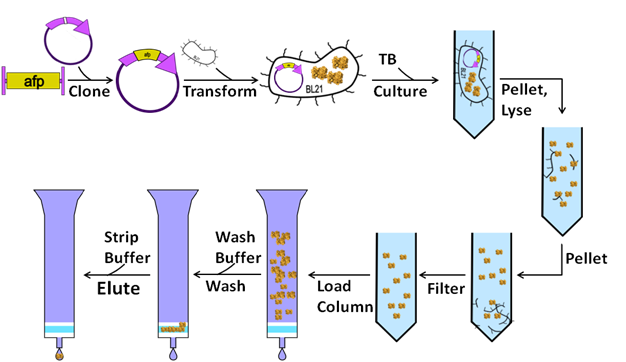
Traditional protein purification is a long and tedious process which often consists of the following 10 steps:
- The first step of traditional protein purification schemes is cloning in a favorite protein (referenced here as afp) into an expression plasmid designed to generate lots of protein.
- Transform this plasmid into an expression strain of bacteria streamlined for protein production
- Culture cells and induce protein expression allowing cells to grow to high density
- Pellet Cells
- Re-suspended in a lysis buffer and incubate for 30 minutes to ensure the full cell lysis, releasing proteins trapped within the cell into the media.
- Spin media down in an ultra-centrifuge for 1hr to remove insoluble matter (membrane, insoluble proteins, etc) and leave soluble proteins in the supernatant.
- Filter the supernatant to remove any lingering debris before being added to an affinity column. This ensure that the columns does not get clogged.
- Run the filtered supernatent over an affinity column several times to ensure thorough binding.
- Flow wash buffer over column to eliminate proteins that non-specifically bind to the column.
- Collect afp by running an elution buffer over the column to release afp.
Though not necessarily hard, this process is tedious and time consuming, taking us about three and a half hours to complete on purification day. With this in mind, we created an idealized protein purification scheme that makes protein purification facile, that takes a fraction of the time older methods require.
Idealized Protein Purification
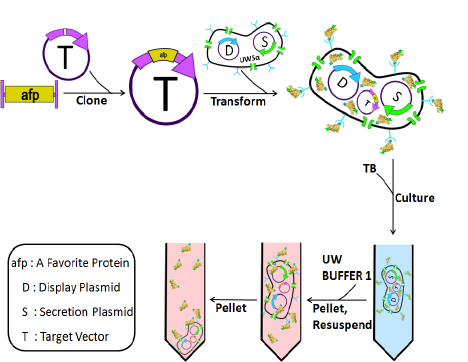
To avoid all the tedious steps one runs into during traditional protein purification we designed a system in which the cell does each of these steps for you making recombinant protein product take only 6 simple steps:
- Clone afp into a "target vector". This creates a fusion protein in which a "display tag" to the N-terminus of AFP, and a "secretion tag" to the C-terminus of AFP.
- Transform the target vector with the AFP insert into cells already containing two additional plasmids. The first is a "Secretion plasmid", which contains all the parts of a type 1 secretion system and specifically recognizes the secretion tag. The second is a "Display plasmid" which expresses a protein that is directed to the cell surface and can bind to the "display tag", and then subsequently release the display tag when a specific small molecule is introduced to the system.
- Grow up cells and induce the expression of afp, just as in the tradiontal protein purification method. The addition of the specific protein tags on afp and additional plasmids in the cell line make it so that as the protein is produced it gets secreted to the media via the secretion system, and then binds to the outside of the cells via the display system. Now that the protein of interest is non-covalently attached to the outside of the cell.
- Spin down the cells and re-suspend in an elution buffer containg a small molecule which disrupts the interaction between the display tag and the protein it was binding to on the surface of the cells. This causes afp to fall off of the cell.
- Collect afp by pelleting the cells again and keeping the supernatant. which contains your purified protein.
This process should take about 10 minutes, quick enough to do between classes! In addition to saving time, our idealized protein purification method has other potential advantages over older methods. By keeping the cells intact, contaminants are not released into the media, which should result in purer protein product. A host of proteins toxic to E. coli could still be grown by secreting them before reaching high intra-cellular concentrations. Alternately, some proteins can’t properly fold in E. coli, but by secreting these proteins into the appropriate media, they might fold correctly. Finally, there are some applications, such as detoxification, where it is desirable to secrete protein into the environment, which our system could be easily modified to do.
 "
"