Team:Duke
From 2009.igem.org
| Home | Project | Team |
The Duke University's iGEM team consists of 5 undergraduates, 3 graduates, and 2 professors. Due to the high costs and inefficiency of the process of cloning a gene, the DUKE iGEM team has invented a new procedure which lowers costs and also increases efficiency. This method, Circular Polymerase Extension Cloning (CPEC), saves time as well, since this method does not involve ligation or restriction enzymes. The rising costs of the current method of producing biodegradable plastics has hindered its widespread use; however, this year's IGEM team has discovered a more efficient pathway to produce these biodegradable plastics. With this team's determination and motivation, they would like to present their two projects:
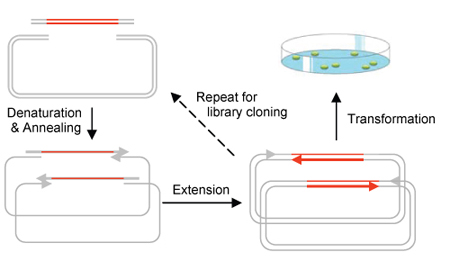
What is CPEC?
Circular Polymerase Extension Cloning (CPEC) is the development of a much simplified
sequence-independent cloning technology based entirely on the polymerase extension
mechanism. This method extends overlapping regions between the insert and vector
fragments to form a complete circular plasmid. An extremely simple theory, CPEC
piggybacks PCR in splicing genes. The gene insert is modified to have ends that
overlap with the ends of the linearized vector and both have similar melting temperatures.
The insert and vector are placed within a PCR machine in the absence of primers.
Denaturation separates the double-stranded insert and vector and the overlapping
ends anneal. Polymerase extension mechanism is then used to complete the plasmid.
What are benefits?
The process of high-throughput cloning is bottlenecked at the restriction and ligation stages. A combination of high costs, requirements for restriction site specific enzymes and general inefficiency of the process makes cloning on a large combinatorial gene library inviable. Circular Polymerase Extension Cloning (CPEC) addresses this issue by eliminating the need for restriction and ligation enzymes and thereby streamlining and condensing the procedure into the duration of 5 minutes.Advantages/Disadvantages of BioBricks
- BioBrick parts can be incorporated in E. coli , due to its common interphase.
- BioBrick parts are not easily made since its
Standardized CPEC
We will apply CPEC in the construction of a multi-component plasmid containing biobricks. Previous Duke iGEM projects have yielded the genes in a metabolic pathway that synthesizes poly(3HB-co-4HB), a biodegradable plastic, in E. coli. We will transform those genes into biobricks, with sticky ends, and efficiently combine them in a vector using CPEC.Biodegradable Plastic Synthesis Pathway in E. coli
Polyhydroxyalkanoic acids (PHA),
naturally occurring storage polymers found in a variety of bacteria, have received
increased attention for their potential use as bioplastics that are both biodegradable
and reduce reliance on petroleum-based plastics. In particular, the copolymer
poly(3-hyroxybutyrate-co-4-hydroxybutyrate), or poly(3HB-co-4HB), which combines
the 3HB and 4HB polymers from different bacteria, has elastic properties ideal
for a wide range of thermoplastic applications. The high cost of PHA, however,
is the biggest impediment to widespread use of bioplastics. Moreover, poly(3HB-co-4HB)
pathways developed so far in /E. coli/ have yielded undesirably low and unpredictable
4HB-to-3HB ratios.
Thus, this project aims to develop a more efficient
biopathway for poly(3HB-co-4HB) while increasing the 4HB monomer composition predictably.
It was hypothesized that optimizing codon permutations of the phaC gene would
greatly increase affinity of PHA synthase to the 4HB monomer. To date, the phaCAB
and cat2 operons have been cloned into pUC19 and PCR Blunt II-TOPO vectors for
successful independent production of the 3HB and 4HB polymers. Ligation and transformation
into /E. coli/ as six different recombinant constructs will soon be completed
and allow for engineering of the poly(3HB-co4HB) biopathway. Future directions
would be to test the hypothesis to see if phaC can be manipulated to increase
4HB-to-3HB composition in poly(3HB-co-4HB) and to increase efficient production
of the bioplastic by engineering the FtsZ cell division protein to allow for cells
to accumulate larger quantities of PHA granules before dividing. Ultimately, once
an optimal biopathway is found, the goal would be to explore a model for mass
production of PHA bioplastics so that novel applications of bioplastics can be
feasible economically.
| Duke University iGEM Calendar | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Below are several important milestones:
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Advisors
| Dr. Jingdong Tian | jtian@duke.edu |
| Faisal Reza | faisal.reza@duke.edu |
Students
 "
"