|
|
Introduction
Implementing a "Timer" Function!
- Our project is to make a sort of "timer", where gene activation is triggered after a certain time.
- Our approach to this goal is to create a series of transcription factors (TFs) that can be activated by the same inducer molecule but with a different sensitivity. In an environment where the inducer concentration gradually levels up, these TFs switches on one by one according to the order of sensitivity.
- We believe such collection of such TF variants would be useful for the timing control in biological function at will, and thereby contribute to the synthetic biology & iGEM community.
- Using a set of TF variants, we aimed to draw an "animated picture": a picture that pop up one by one.
|
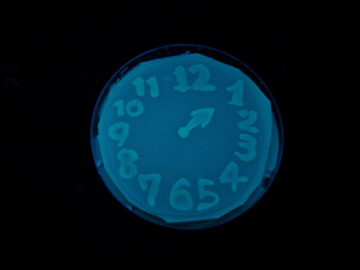
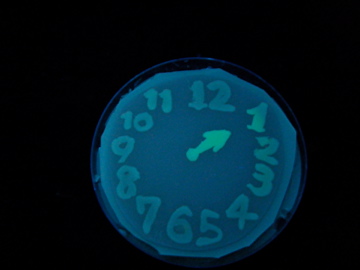
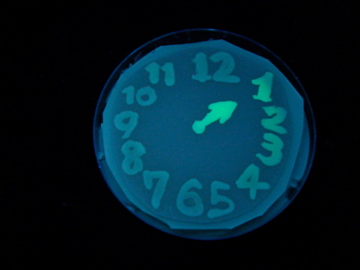
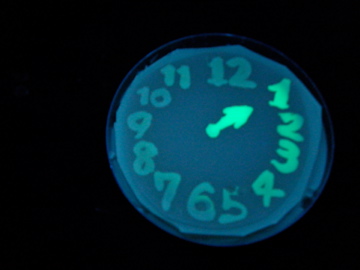
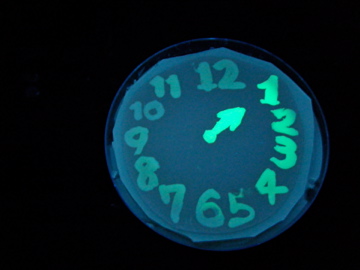
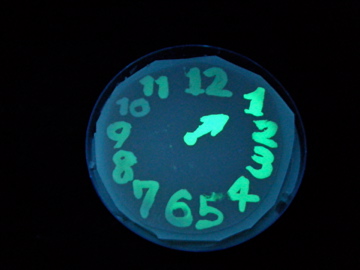
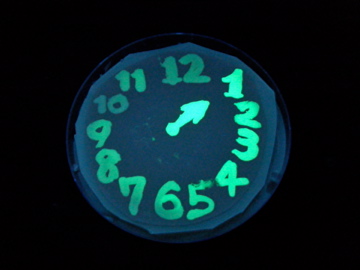
Fig. 1 Completion drawing of our bacterial timer
|
Project Design
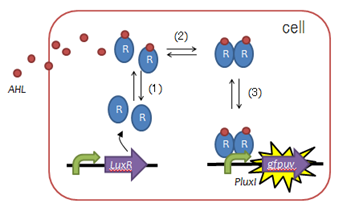 Fig. 2 Function of LuxR :(1)LuxR protein generation, (2)AHL binding domain, (3)dimerization, (4)DNA binding domain - To create such a timer, we utilized LuxR, a protein used to mediate cell-cell communication during quorum sensing, as shown in the figure on the left.
- By inserting point-mutations in the luxR gene, we sought to create LuxR proteins requiring a variety of lengths of time to activate downstream transcription.
Experiments, Results & Discussion
Experiments
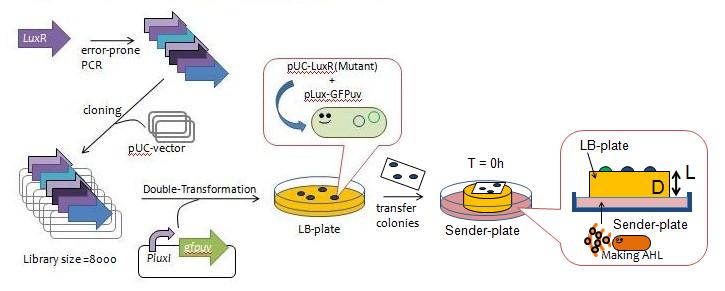 Fig.3 Y Directed evolution to get some delayed-LuxR mutants - We created a mutant library of LuxR (size = 8000) with the use of error-prone PCR and incorporated the resulting coding regions into expression vectors.
- The vectors were then transformed into a strain of E.coli JW1226 cells harboring plux-gfp and grew approximately 200 colonies.
- The colonies were lifted off the agar plate with a nitrocellulose filter and transfered to a plate containing AHL in order to observe the evolution of GFP fluorescence over time (Fig. 4).
- We selected 13 colonies that were slow to display fluorescence, which we designated as delyaed-LuxR mutants.
 Fig. 4 display fluorescence
- The following lists where point-mutations were incorporated into the luxR gene.
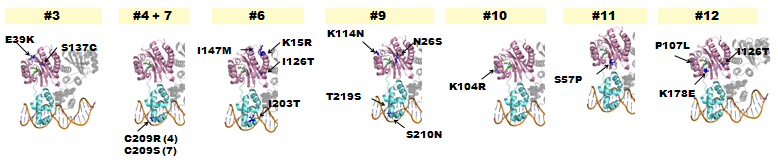 Fig. 5 - A delayed GFP expression (in comparison to wild-type LuxR) phenotype was observed from colonies housing genes in which either the AHL-binding domain or the DNA binding domain had been mutated.
Characterization of LuxR Mutants
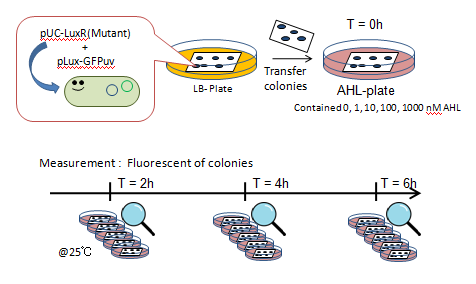 Fig. Characterization of LuxR Mutants Experiments
- JW1226 bacterial strains co-transformed with plasmids containing either wild-type or mutant (13 variants) LuxR and pLux-GFP were cultured at 37℃ for 12h.
- Using a pin, the culture was transfered to a nitrocellulose filter, placed on an agar plate, cultured at 37℃ for an additional 12h.
- The nitrocellulose filter was then transfered to a solid medium containing 0, 1, 10, 100, or 1000 nM AHL.
- Designating this the start point, we observed fluorescence through the naked eye at 30 minute intervals for 4 hours under UV light of 365nm (Funakoshi UVGL-58 at long wave mode).
Results and Discussion
- Upon further characterization of the 13 LuxR mutants above, we found that several of these had similar response times to AHL. We therefore selected 8 (Mutant No. 1, 3, 4, 5, 7, 11, 12, 13) out of the 13.
Results on 100 nM AHL medium plate
 Fig.時間依存 - Among 0 ~ 1000 nM AHL media examined, we found that the 100nM plate gave the larger time difference of response of GFP expression with mutated LuxR.
- The mutated LuxR was caracterized as follows.
- The timing of response is delayed from wt.
- The increasing ratio of GFP fluorescence after the response is less than that of wt.
最初はDNA結合ドメインが
変になってるのがダラダラあがるんだろうという想をしていたのですが結果はそうではない。
- 4と7はAHL応答濃度の閾値は変化してるみたいなんですが
- 応答が生じてからの蛍光の上がりかたはだらだらしているわけではない。
AHL濃度を振った場合の、6h後の蛍光強度の差についての結果
 Fig.濃度依存 - スイッチングが起こる濃度がWTと異なるMutantを得ることができた。
- 変異がはいっている部分がほとんど同じである#4と#7の間に、以下のような差が見られた。
- 4 : 10 nM AHLで蛍光がみられる。
- 7 : 100 nM AHLで蛍光がみられる。
- DNA結合ドメインに変異が入っている6と9の表現系をみると応答濃度にさがでている。
- 結合ドメインは応答濃度を左右しているのではないかと考えた。
Demonstration
- Double-Transformed (pUC-LuxR Mutants series-ColE1 and pLux-GFPuv-P15A) and cultured on LB-Amp-Cm-plate. (37°C,12h)
- Pick and cultured in 10mL of LB-Amp-Cm (37°C,12h).
- Drawing picture with those liquid culture on nitrocellulose Filter.
- Those nitrocellulose Filter placed on LB-Amp-Cm-plate.
- Cultured at 37°C, 12h.
- Transfer pictures on special plate.
- Exposed to UV (385nm) light once every 30 minutes to observe GFP fluorescence.
- For the determination of best condition for E coli painting, Click here!
Conclusions
- By using error-prone PCR, we have created a LuxR library.
- With a simple and convenient screening method, we have isolated various LuxR mutants which confer delayed switching behavior in GFP signals.
- By introducing these LuxR variants together with reporter genes (such as GFP) under the control of Lux promoter, we created bacteria 'ink's that develop their color with unique delay-time.
- We conducted painting with these bacteria inks thereby created animated pictures.
- Characterization of LuxR mutants is ongoing. But our preliminary data showed that some of the variants turned out to be the one with less sensitivity to AHLs, and others seemed to be as sensitive as wild-type LuxR but seemed less efficient somewhere in the downstream process.
- We created variety of Biobricks during the course of this projects. Some of them are characterized and sent to the HQ, and many more are almost ready for shipping!
|
 "
"