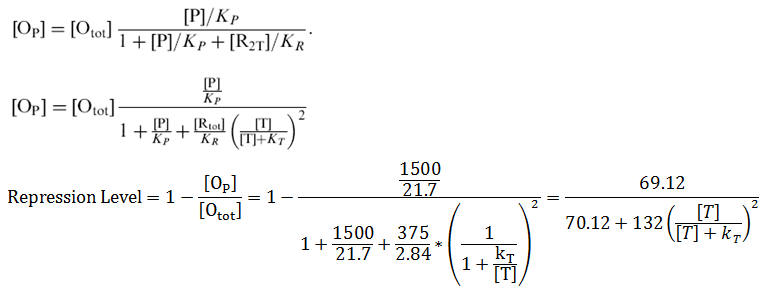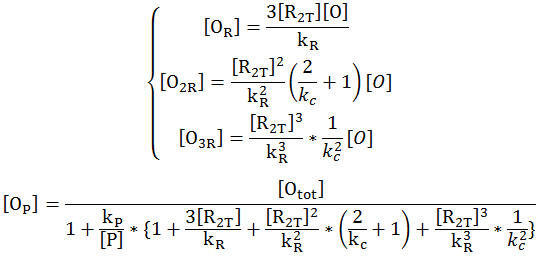Team:Stanford/ModelingPage
From 2009.igem.org

| Home | Project | Modeling | Parts | Notebook | Team | SBOL-V |
Our modeling team consisted of Leon Lin and Mary Yang.
Contents |
Goal
- To model and optimize the kinetics of these devices.
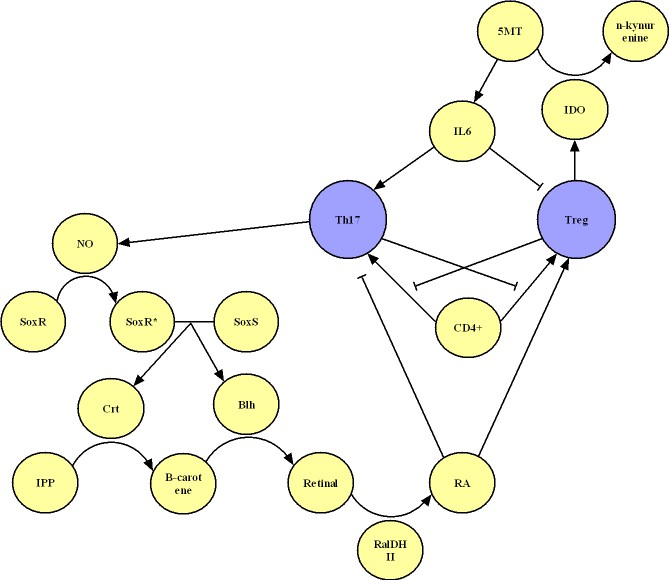
- Population dynamics: analyze the conditions to switch between Th17 and Tregs.
- To predict the results of experiments which we could not perform in the lab
- Below is a chart of all the individual processes in our respective devices. This page contains mathematical models many of those processes and provides a quantitative description for how our devices work.
Anti-Inflammatory Device
The IPP-->B-carotene-->RA Model
The process above can be devided into two procedures:
- IPP*8-->B-carotene
- B-carotene(+Blh)-->Retinal(+RalDH)-->Retinoic Acid
Modeling on the production of B-carotene
The process of IPP producing B-carotene is quite complex, as shown in the Fig 3.2.
In this process, we basically care about two main issues:
- Yield output of B-carotene ([B-carotene]/[All Caronoids]).
- Velocity of the whole process
Yield output of B-carotene
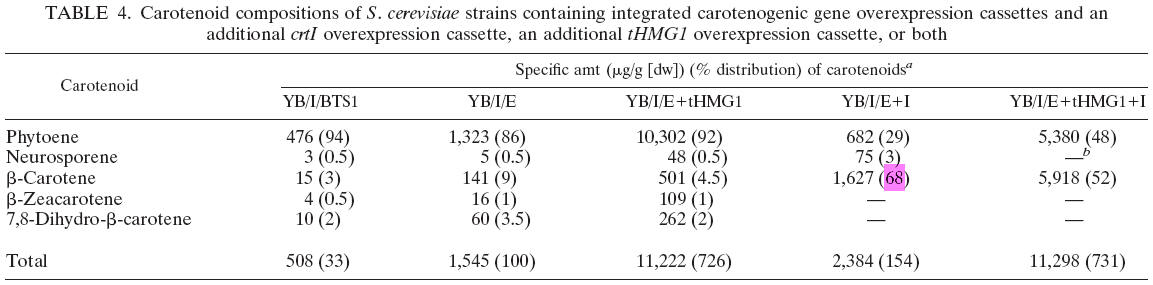
We found the B-carotene distribution in yeast in [1]. Below is an important form as to the issue.
Form 3.1 B-carotene distribution [1]
Basically, using the cluster of "YB/I/E I", in the final product of caronoids, we get 68% of B-carotene, highest percentage in the paper. (Also 29% Phytoene, and 3% Neurosporene.) "YB/I/E tHMG1 I", producing 52% of B-carotene, might be another choice.
Production rate of B-carotene
As this is a really long process....Basically, the velocity is mainly dependent on the most time-consuming reaction in the whole chain. Thus, Leon and I looked for the kcat values of different enzymes in the procedure, as shown in the cart below:
Apparently, cyclization of Lycopene is the slowest reaction, as the concentration of enzymes are approximately in the same level.
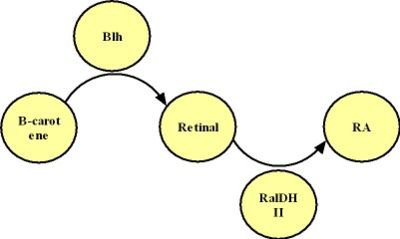
Modeling on the B-carotene-->RA process
The production of RA is mainly based on a chain of two catalyzed reactions, as shown in the graph below:
<center>Fig 2.3 The production of RA</center>
Neither Retinal nor RA has any other degrading process in E.coli. Degrading rate of B-carotene is 9.769e-9 s^-1. Other paremeters we use in this model are shown in the form below:
<center>Form 2.3 Parameters used in the B-carotene-->RA model</center>
Simulation and Analysis
- Equations for the IPP-->RA process:
References
[1] High-Level Production of Beta-Carotene in Saccharomyces cerevisiae by Successive Transformation with Carotenogenic Genes from Xanthophyllomyces dendrorhous. APPLIED AND ENVIRONMENTAL MICROBIOLOGY, July 2007, p. 4342–4350
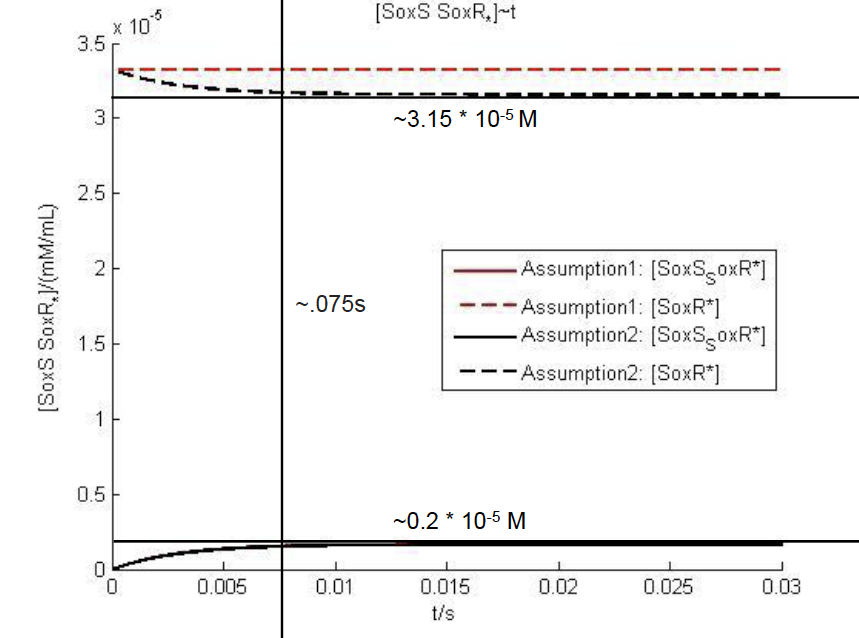
The NO->SoxS-SoxR Model
This model illustrates the level of SoxS-SoxR binding over time
Anti-Immunosuppresion Device
Models of the Tryptophan System
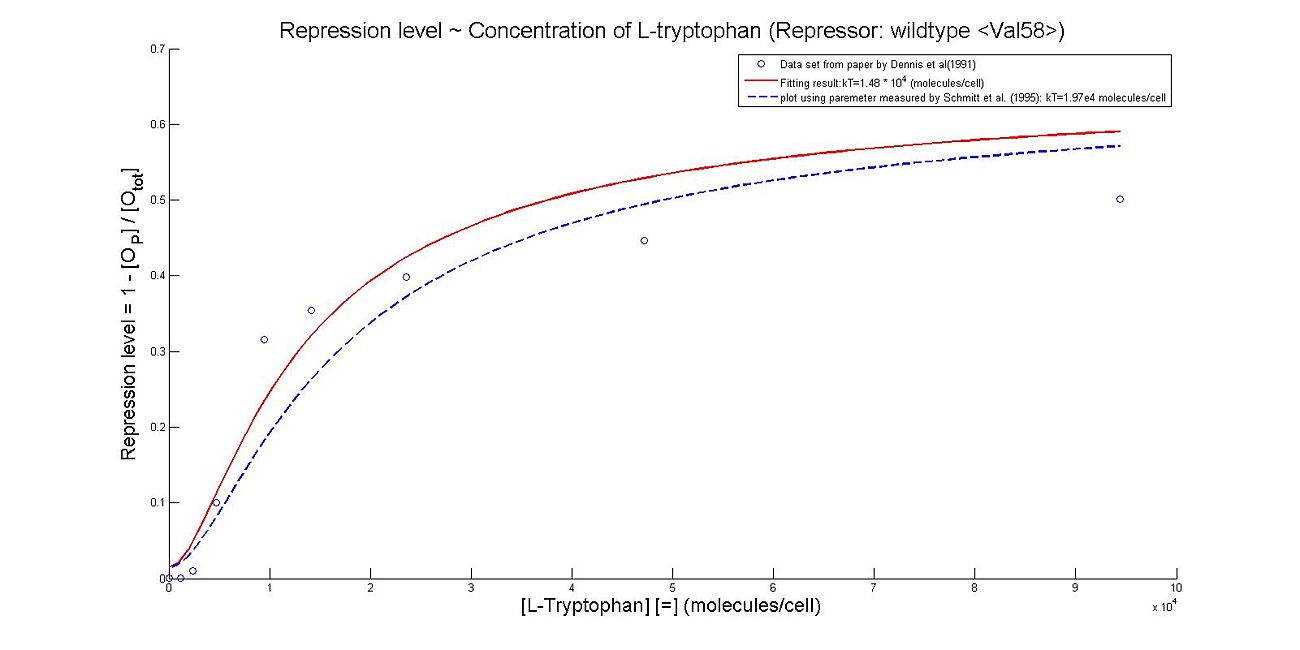
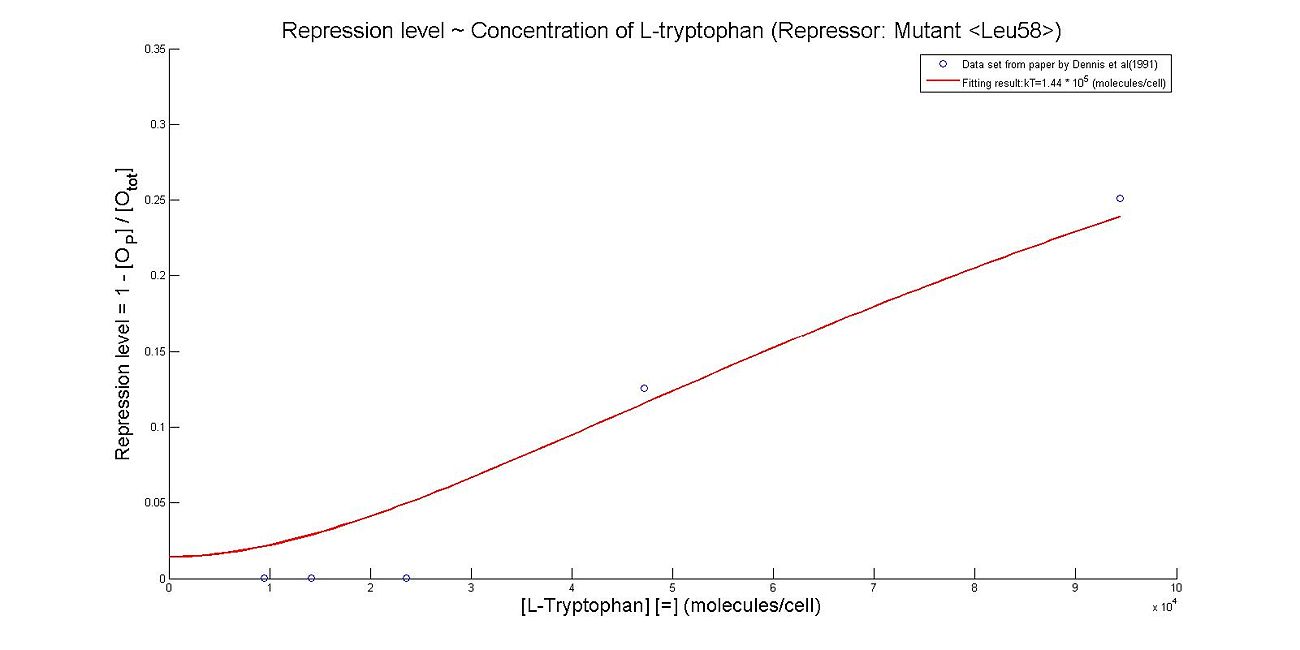
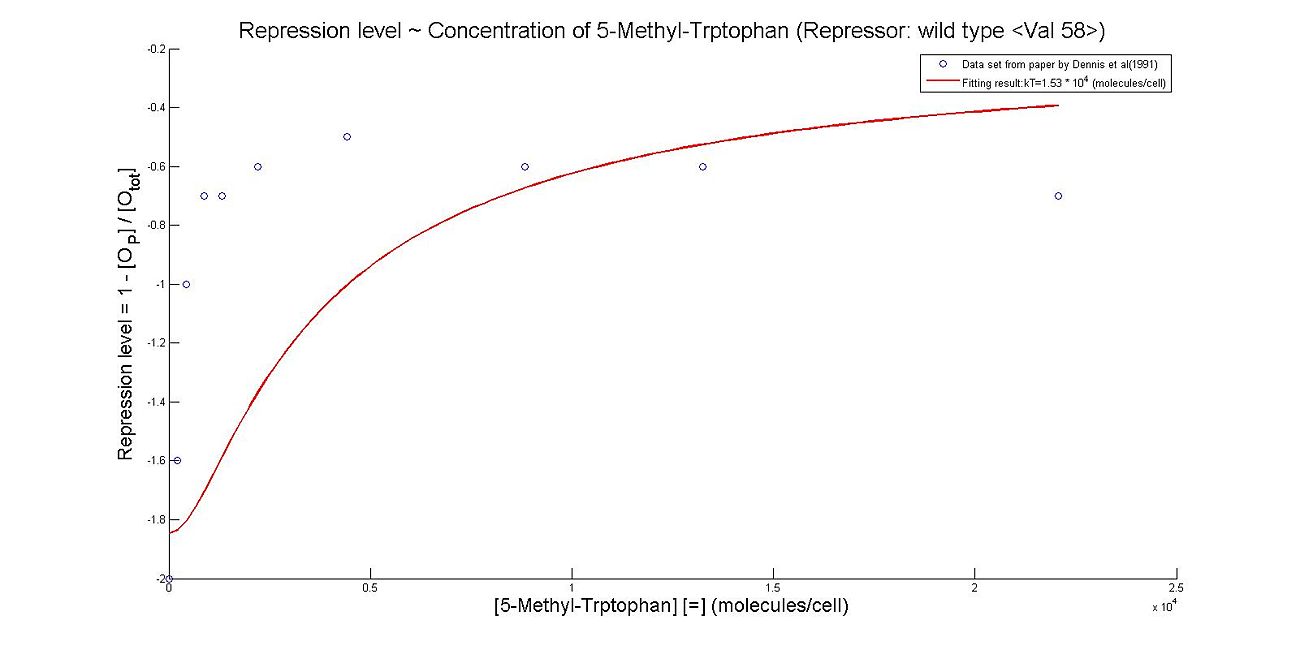
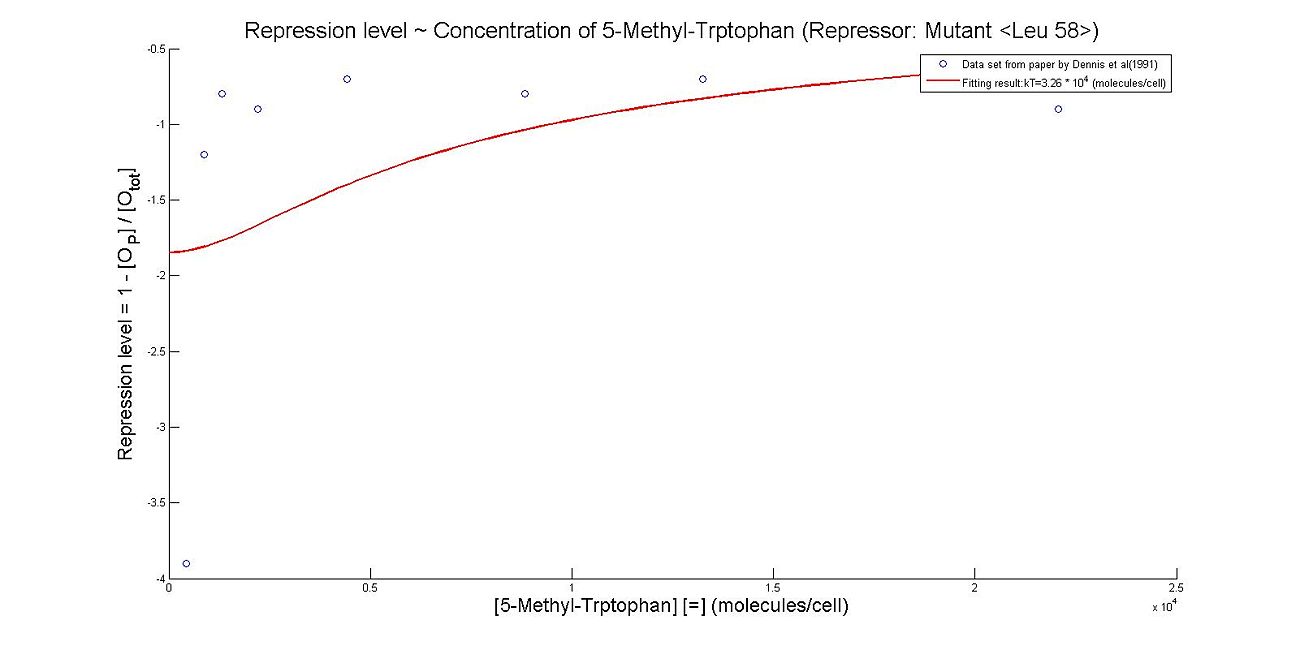
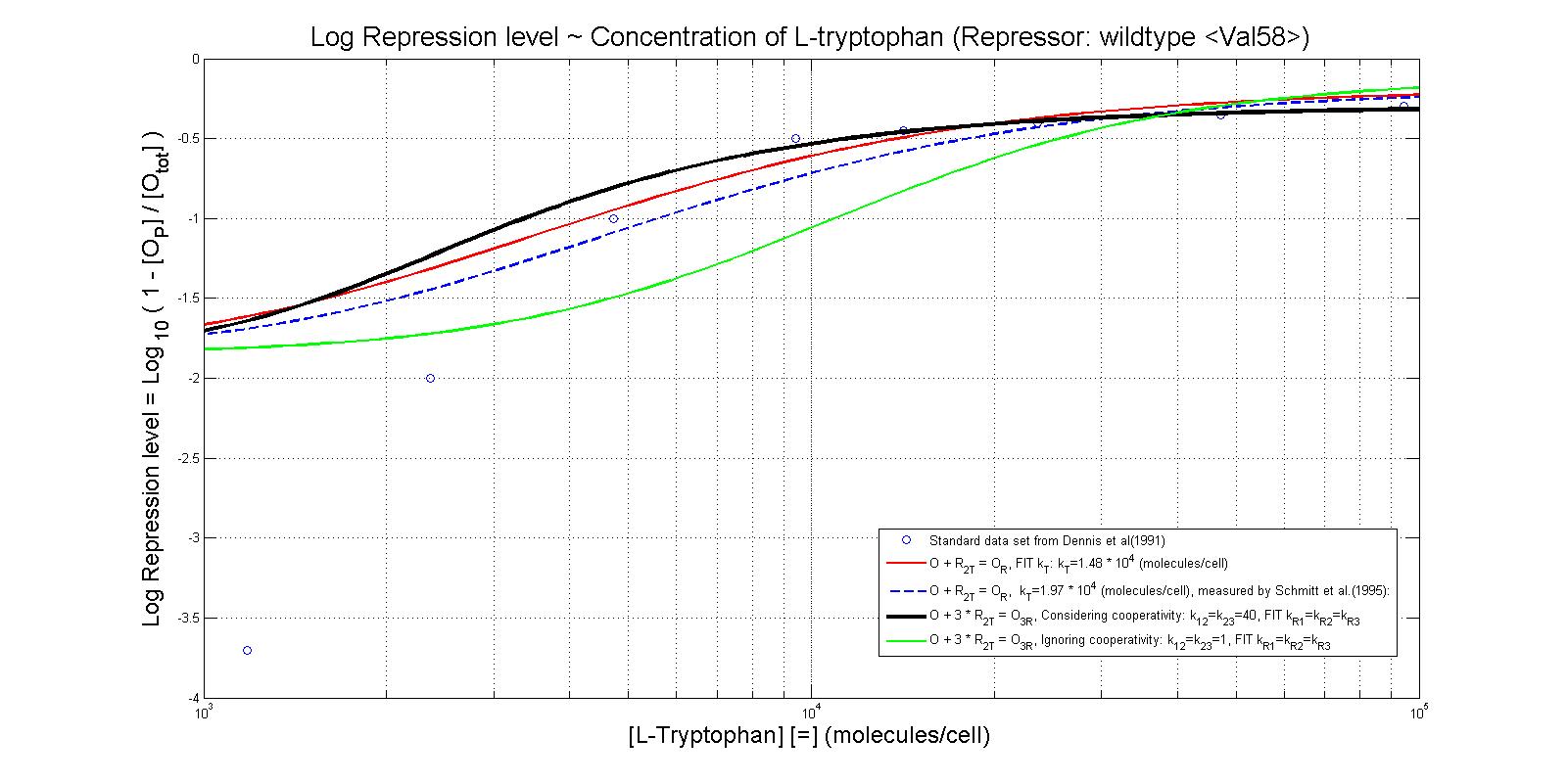
- Perhaps the most unique model we made this summer was the model which illustrates the repression rates of 5MT binding with our mutant repressors. Because there was almost no information containing kinetic or thermodynamic data regarding the particular mutations we made to the system, many of our constants were derived based upon best fit lines that we had created after looking at data. We made many assumptions in our models, which are listed below. However, from closely examining these graphs, it becomes clear that the particular mutations we made to the tryptophan system were justified given the faster rates of repression.
- The first set of graphs illustrate the theoretical levels of repression assuming that there is no cooperativity between the binding sites while the second set of models illustrate cooperativity. We decided to include both because many wild type tryptophan model elect to discount cooperativity, however, despite the difficulties we had in calculating the cooperativity constants, we found that our model was better able to fit data when assuming the presence of cooperativity.
Wild Type Model Ignoring Cooperativity
Base on the fitting to Fig 1 from Reference [2], we came to the result kT=1.62e4 molecules/cell. While the kT measured by Schmitt et al. (1995) is 1.97e4 molecules/cell. The figure below shows the result of our fitting, and the comparison to standard kT value.
Thus, it is reasonable to calculate k_T values for trp with mutant repressor, 5MT with wildtype repressor, and MT with mutant repressor.
Wild Type Model Including Cooperativity
IDO Catalyzed Reactions
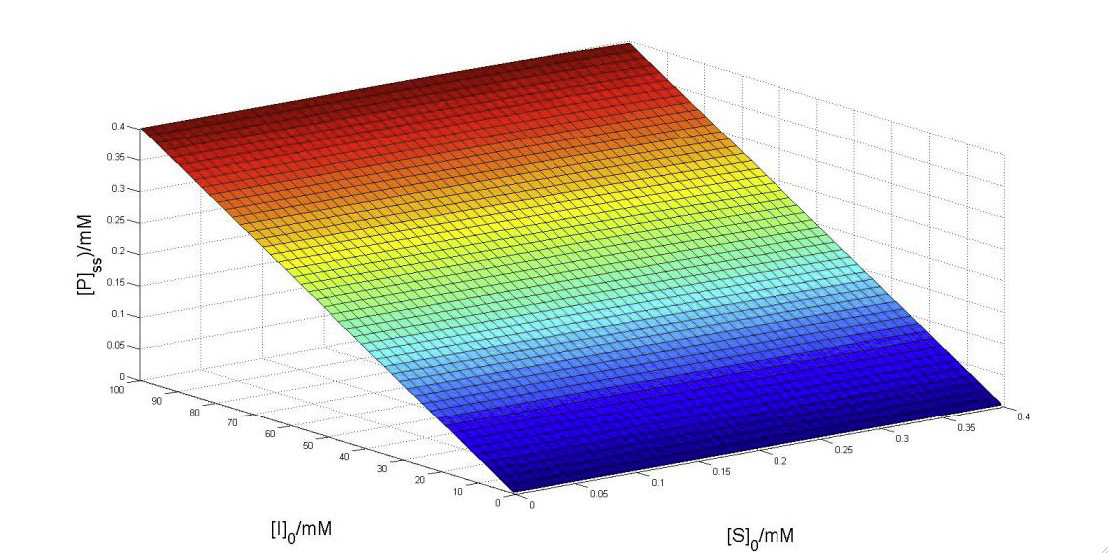
- Below is a graph which illustrates the interaction between IDO, 5MT and Trp. This model illustrates the competitive inhibition which results from both 5MT and wild type Trp attempting to bind with IDO. In the model below, the product between wild type Trp and IDO is represented by P, wild type Trp is represented by S, 5MT is represented by I and the product between IDO and 5MT is represented by J.
- Additional graphs, parameters used, and our derivation of the model can be found in the pdf document below.
 "
"