BioBrick information
From 2009.igem.org
Contents |
IGEM 09 DNA distribution Guide
General Registry Information
• Kit includes high-quality parts from the past as well as new parts from 2008 that were designated by teams as favorites.
• Kits include confirmed quality controlled parts, (3) 384 well plates total, the 2009 kit will be a very effective basis for your synthetic biology projects.
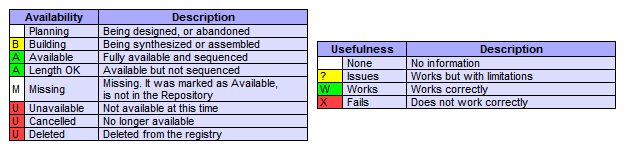
• When searching for Parts use this as a key to determine whether parts are working or not.
• When using DNA from wells the first thing to do is transform it into competent cells. There is not enough DNA to work directly from the parts. Using the Registry:
• Find the desired part’s page on the registry
• Click on “Get This Part” at the top right part of the page. The DNA is accessible only if it reads “Available”.
• The link will be a page outlining the location of the part in the parts kit (Spring 2009 distribution), along with QC information and some other ways of obtaining the part if it is not included in this year's parts kit.
• If you find the part, or parts, that you require are not available in the distribution but available in the Registry, feel free to send us an email (hq@igem.org) in order to request it.
• Include:
--- 1. iGEM team or lab name
--- 2. Part name
--- 3. Plasmid and resistance
--- 4. Source plate and well
--- 5. If there is a change in shipping address from the one listed on your team/lab page, please notify us.
Storage:
• Dried DNA can be stored at room temperature.
• However once 15ul of TE (or water) are added to any of the wells, either store the kit plate with its plastic cover in a -20C freezer, or removing the remainder of the resuspended DNA from the well and store AND LABEL it separately in a -20C freezer. Pipette it into an microcentrifuge tube and label it with the Plate number and well coordinates. Ex:
Planning part usage:
• Parts come as dried DNA found in the plasmid seen below.
• E is the EcoRI restriction site
• X is the XbaI restriction site
• S is the SpeI restriction site
• P is the PtsI restriction site
• Be sure to cut out using only EcoRI and SpeI OR XbaI and PtsI
--- SpeI and XbaI are bad because they will re-ligate to itself because of compatible ends
--- EcoRI and PtsI will also re-ligate to itself because of compatible ends
• Usually the origin is a high copy number so that after transformations DNA can be collected through a simple MiniPrep.
• BioBrick plasmid backbones can have resistance to ampicillin ("Amp" or A), kanamycin ("Kan" or K), chloramphenicol ("Cm" or C) or tetracycline ("Tet" or T).
Using the Parts:
• You will transform the DNA into cells and make your own glycerol stocks of any part that you wish, however it does not contain enough DNA to do assembly. To use the DNA in the Parts Kit:
1. With a pipette tip, punch a hole through the foil cover into the designated well. Do not remove the foil. Make sure you have properly oriented the plate. The two notched corners of the plate should be oriented at the BOTTOM of the plate. Example: Well 13-H
2. Add 15uL of diH2O
3. Take 2uL DNA and transform into your desired competent cells, plate bacteria with the appropriate antibiotic* and grow overnight.
4. Pick a single colony and inoculate in 4-5mL of LB broth (again, with the correct antibiotic) and grow for 18 hours.
5. Use the resulting culture to miniprep the DNA AND make your own glycerol stock (for further instruction on making a glycerol see this page). Registry’s Quality Control:
• Parts sequences are compared with their target sequence through software, and are given the following qualitative values: --- Confirmed --- Partially Confirmed --- Long Part – we don’t know whether the entire part is confirmed, but the sequence ends are --- Inconsistent --- Bad Sequence – usually caused by low DNA concentration or incorrect primers --- [U] - User Confirmed – we manually reevaluate the inconsistent sequences and look at the trace files to see if a simple shift of the sequence will confirm it
• For more in-depth analysis, when reviewing quality control information for a part, just click on the "Sequence" link, right beside the part's Sequence result. Every user can look at all the results we get back from Genewiz: including the trace files, quality scores, and sequence reads.
For more info: http://partsregistry.org/Help:Contents
 "
"