Team:ULB-Brussels/Project/Properties
From 2009.igem.org
Contents |
Physical and chemical properties of our glue
In order to characterize the glue, a series of tests have been carried out such as:
Phenotypic description of the transformed bacteria
Transformed bacterial colonies showed a filamentous aspect when put back in a liquid culture. Because of the glue, the whole colony is taken as they stick together. We let the bacteria grow at 37°C overnight in a shaking incubator. A strand originating from the bottom of the tube was observed.
The first tests were done to see if our glue actually stuck. A colony from a Petri dish was isolated and spread on a sheet of paper. After that, the paper has been placed on a glass substrate during a short period of time (1 to 2 hours) under a 2N pressure. We noticed a slight adherence and a bright spot where the bacteria had been spread on the sheet of paper.
A second phenotypic test has been carried out in a liquid culture with glass beads. The beads have been dipped into the liquid culture and placed on a plastic medium. After 7 hours of incubation, we observed a perfect adherence to the plastic medium (even after a huge shock).
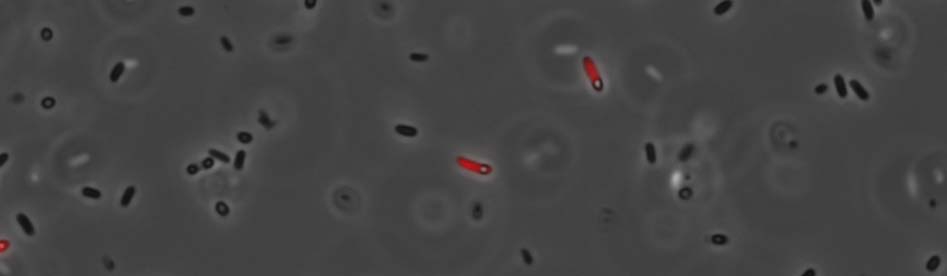
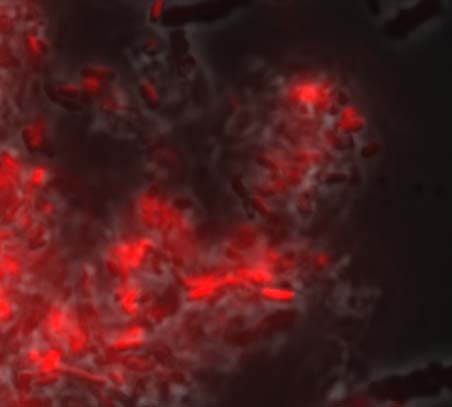
Fluorescence microscopy
Only a few bacteria showed expected fluorescence properties (due to the RFP sequence).
Furthermore, it seems that the glue is synthesized in a vacuole. Indeed vacuoles were visible in transformed bacteria. (see figure below)
This is a significant feature of our modified strain of E. coli. We also observed a higher bacteria aggregation at 27°C in comparison to 37°C (see figure below).
Biofilm-forming abilities
A test on a 96-well microtiter was carried out to highlight biofilm-forming abilities. Unfortunately, this test was not conclusive; the bacteria did not stick to each other. Caulobacter crescentus bacteria might stick to nearly all substrates but they also synthesize a matrix of extracellular polymeric substance to adhere to each other.
Wet strength
Water and "growth medium (LB)" inhibit the adhesive effect. Moreover, water separated glass beads. This result was not what we expected given what is observed for C. crescentus living in aquatic environments. It would be advisable to specify that glass beads stick together again when water is removed.
First it is possible that the similar genes found in E.coli (hfsE, hfsF, hfsC, hsfI, hfsA, hfsB and hfsD genes) do not work in an identical way than in C. crescentus.
Secondly the two exogenes (named hfsG and hfsH) are overexpressed compared to the other holdfast biosynthesis and export genes. Indeed, we used a high copy plasmid with a strong promoter. A failure during sugar polymerization might explain the ineffectivineness in water environment.
Thirdly, C. crescentus synthesizes its complex holdfast structure at the tip of a stalk. The anchoring proteins might be required to "protect" the adhesive against the water.
Finally, apart from the polysaccharides, the composition of the adhesive remains unknown. Other essential components might be implicated in the water and solvent strengths.
Complementary approaches are foreseen to differentiate these assumptions:
- The other genes (hfsE hsfF, hsfC hsfI, hsfD hsfA and hsfB) should also be transferred to E. coli with the same transcription rate.
- As the anchoring genes may play a role in the glue composition, hfaA, hfaB and hfaD genes should be inserted in E.coli too.
- We need to have more information about the adhesive composition. A proteomic approach with C.crescentus mutants that do not produce the glue might be undertaken.
Tensile strength
Two different tests have been carried out in order to characterize the tensile strength of the glue. It is important to note that the glue used in these experiments has not been purified. It consists of bacteria lysate.
In the first test, liquid cultures of transformed bacteria are tested on different materials. To enhance the adhesion of these materials, different attempts have been made to minimize the impact of the "wet part" of the liquid culture. Indeed, previous experiments showed the adhesive to be ineffective in water.
On one hand different materials stuck together have been placed in a box filled up with salt. On the other hand, a sample of 300 μ of liquid culture has been spread on LB solid medium (Petri dish). The bacteria then grow on the surface while the LB solid medium absorbs the remaining liquid. To collect the glue on a specific material, it has been rubbed against the Petri dish. In fact, this is how we first showed our glue was functional.
Unfortunately none of these experiments were successful. Two hypotheses can be put forth. Firstly the glue is still too liquid and because of that ineffective. Secondly there are proportionately more bacteria than glue. So there is not enough glue to stick different materials together.
In the second test, different "GluColi" colonies have been taken from solid cultures and directly put on the materials. This experiment showed better results than the previous one. Several materials such as plastic, wood, cork, paper and CD-ROM have been successfully stuck.
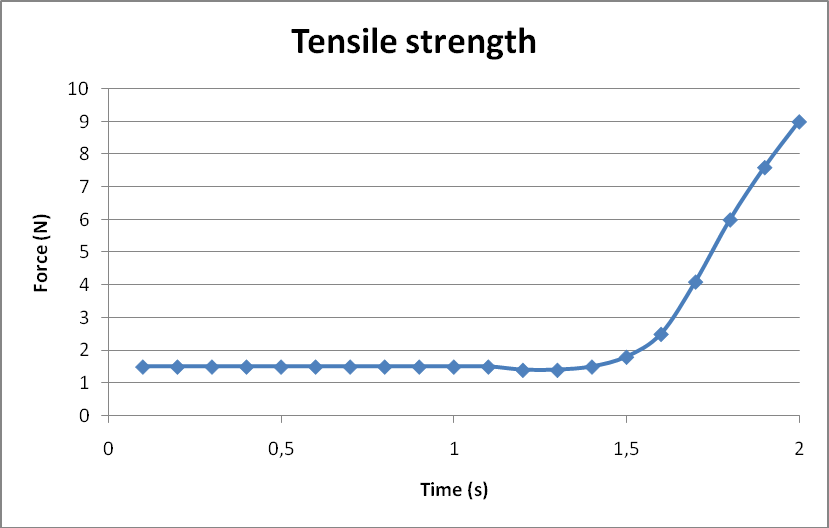
In order to measure the tensile strength, two plastic materials have been stuck together on a surface of 1.5 cm2. The dynamometer (Force sensor, Pasport, PS-2104) showed a tensile strength of 9N which is 0,06 N/mm2,(see figure above). This result is far less than the one found in other laboratories with C. crescentus (up to 68 N/mm2). One explanation, as previously stated, is that our glue is not purified.
 "
"