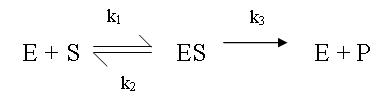Team:Imperial College London/Drylab/Enzyme/Analysis/Detailed
From 2009.igem.org

The behaviour of the system can be described with ODEs. The first 4 equations describe the single-substrate mechanism for an enzyme reaction without Michaelis-Menten assumptions, while equation 5 describes modification with the Michaelis-Menten assumption.
The Enzyme Mechanism
k1, k2 ,k3 are the kinetic rate constants for each step.
In substrate binding, 1 molecule of Enzyme (E) binds with 1 molecule of Substrate (S) in a reversible reaction to form the Enzyme-Substrate complex (ES). We are unable to measure this step.
In catalysis, the Enzyme-Substrate complex (ES) produces the unchanged enzyme (E) and the product (P) in an irreversible reaction. This step can be measured.
When [S] becomes high enough, the second step of the enzyme kinetics becomes the rate determining step, controlled by k3.
This enzyme mechanism scheme can be described by the following differential equations describing rate of change of each component. Each component is described by a simple rate term for either its production or its decomposition.
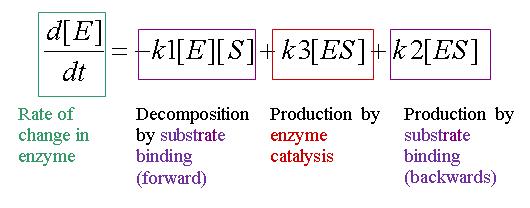
Equation 1: Rate of change of Enzyme
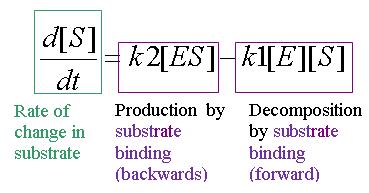
Equation 2: Rate of change of substrate
Equation 3: Rate of change of enzyme-substrate complex

Equation 4: Rate of change of product
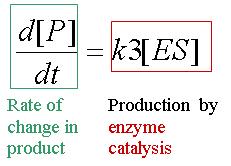
With Michaelis-Menten Assumptions
Consider the enzyme mechanism again:
From the model, we want to derive an equation that describes the rate of enzyme activity (amount of product formed per time interval) as a function of substrate concentration.
Consider equation 4: the rate of product formation. This equation is essentially useless as we cannot measure [ES] experimentally, therefore, to correctly determine d[P]/dt, we need to solve for ES in terms of the other quantities.
Therefore, we need to make some approximations as mentioned previously. (hyperlink to assumptions part)
At steady state, equation 3 is reduced to:
![]() When this result is used together with equation 4,
a new differential equation for rate of change of product is obtained:
When this result is used together with equation 4,
a new differential equation for rate of change of product is obtained:
Equation 4-MM: New rate of change of product
Michaelis-Menten assumption:
- The Michaelis-Menten (MM) assumption is a simplifying assumption that is commonly made with enzymatic reactions. It says that since substrate binding is very fast compared to catalysis, the enzyme complex ES is always at quasi-steady state, ie:
 , during the time of the experiment.
, during the time of the experiment.
- It is straightforward to prove that combining the MM assumption
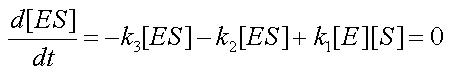 with the conservation equation [E0]=[E]+[ES] yields a new form for the fourth equation
with the conservation equation [E0]=[E]+[ES] yields a new form for the fourth equation 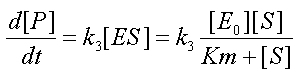 , where KM is the Michaelis-Menten constant.
, where KM is the Michaelis-Menten constant.
Equation (4-MM) seems more complex but is in practice more useful exploitable than Equation (4). Indeed, [S] can often be measured during an experiment, whereas [ES] cannot. Furthermore when the initial concentration of substrate [S0] is large compared to KM, then [S] remains large compared to KM over a certain range of time (that includes the start of the assay) and (4-MM) simplifies to  over this range of time. Simply put, for these times P is produced at a constant rate at the start of the assay. Because of its close association to one of the possible behaviours of the system, the KM is one of the important pieces of information that can be given about an enzymatic reaction.
over this range of time. Simply put, for these times P is produced at a constant rate at the start of the assay. Because of its close association to one of the possible behaviours of the system, the KM is one of the important pieces of information that can be given about an enzymatic reaction.
 "
"