Team:Berkeley Wetlab/Cell Surface Display Parts
From 2009.igem.org
| Line 75: | Line 75: | ||
The precise role of spacers in the functionality of proteins has not been extensively characterized; however, as illustrated in the Hag protein, multiple spacer elements are typically present in natural display systems suggesting their importance. We hoped to characterize the effects of the inclusion of spacer elements within our passenger-display design. There were five spacer domains introduced to our surface display system: INP-repeats, beta roll, bet helix, Gly-Ser repeats and GFP-LVA. These elements are further discussed below. <br> | The precise role of spacers in the functionality of proteins has not been extensively characterized; however, as illustrated in the Hag protein, multiple spacer elements are typically present in natural display systems suggesting their importance. We hoped to characterize the effects of the inclusion of spacer elements within our passenger-display design. There were five spacer domains introduced to our surface display system: INP-repeats, beta roll, bet helix, Gly-Ser repeats and GFP-LVA. These elements are further discussed below. <br> | ||
| - | INP-Repeats-repeated portion of ice nucleation protein (INP) sequence. <br> | + | INP - Repeats-repeated portion of ice nucleation protein (INP) sequence. <br> |
| - | Beta Roll- | + | Beta Roll - protein tertiary structure that is natively found in autotransporters and other outermembrane proteins. |
| - | Beta Helix-protein helical structures stabilized by hydrogen bonds and protein-protein interactions. The resulting structure contains two to three faces formed by the association of parallel beta strands. | + | Beta Helix - protein helical structures stabilized by hydrogen bonds and protein-protein interactions. The resulting structure contains two to three faces formed by the association of parallel beta strands. |
| - | Gly-Ser Repeats | + | Gly-Ser Repeats - a flexible 14-mer amino acid sequence of glycine and serine residues repeated. |
| - | GFP-LVA | + | GFP-LVA - |
==Displayers== | ==Displayers== | ||
Revision as of 00:16, 20 October 2009
Contents |
Passengers
Follow any of the links below to see assay information for each of the passengers we made.
|
Streptavidin |
Leucine Zippers |
Ag4 Peptide |
|
MGFP-5 |
Cellulases |
TypeIII Needle scFv |
Spacers
Spacer elements occur in natural membrane protein systems. This is exemplified in the Hag protein, autotransporter-containing system containing two spacer beta roll domains, shown below:
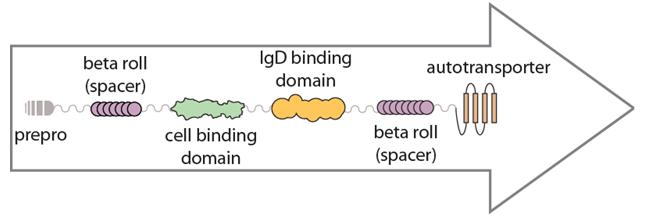
The precise role of spacers in the functionality of proteins has not been extensively characterized; however, as illustrated in the Hag protein, multiple spacer elements are typically present in natural display systems suggesting their importance. We hoped to characterize the effects of the inclusion of spacer elements within our passenger-display design. There were five spacer domains introduced to our surface display system: INP-repeats, beta roll, bet helix, Gly-Ser repeats and GFP-LVA. These elements are further discussed below.
INP - Repeats-repeated portion of ice nucleation protein (INP) sequence.
Beta Roll - protein tertiary structure that is natively found in autotransporters and other outermembrane proteins.
Beta Helix - protein helical structures stabilized by hydrogen bonds and protein-protein interactions. The resulting structure contains two to three faces formed by the association of parallel beta strands.
Gly-Ser Repeats - a flexible 14-mer amino acid sequence of glycine and serine residues repeated.
GFP-LVA -
Displayers
An outmembrane protein that carries another protein through the outermembrane
For successful cell surface display of proteins, there must be an effective protein localization mechanism. Gram-negative bacteria such as E. Coli have two membranes, which present a problem for transporting proteins synthesized in the cytoplasm to the outside of the cell. Various transport schemes exist in gram-negative bacteria to effectively localize proteins to the outermembrane. The most common schemes are TypeI, TypeIII, and TypeV secretion.
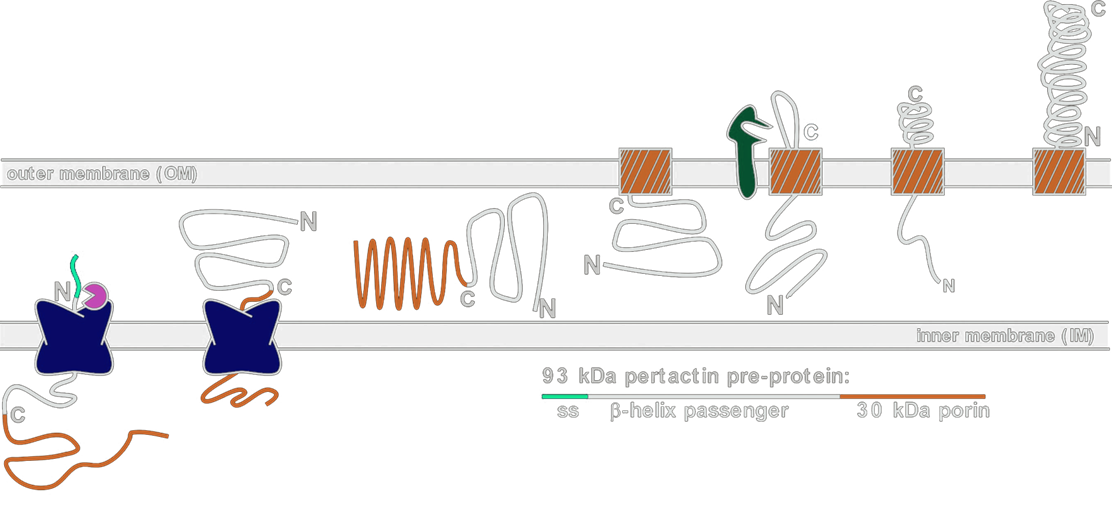
In our display systems, we chose a class of outermembrane proteins called autotransporters that localizes proteins via the TypeV secretion mechanism. Over 700 autotransporters have been sequenced, many of which are used to export virulence factors to the outside of the cell. We decided to harvest this localization system for cell surface display because the outermembrane protein (aka displayer) spontaneously inserts into the outermembrane and pulls the protein it is covalently linked to (aka passenger)into the extracellular space. Moreover, autotransporters are capable of pulling through large proteins, such as enzymes and single-chain variable fragments.
As depicted in the diagram above, the autotransporter localization begins with localization to the periplasm via the Sec secretion pathway. The translocated protein remains unfolded in the periplasm until it inserts into the outermembrane by forming a beta barrel with its C-terminal 250-300 amino acyl residues. The N-terminus of the protein (containing our passenger of interest) is then pulled through the barrel to the outside of the cell. Passengers of displayers are often cleaved for extracellular secretion. In our systems, however, we cleaved the prepro sequence that signals for peptide cleavage so that our passengers would remain attached to the transmembrane displayer protein.
In constructing our parts, we looked into various autotransporters with different attributes conducive to cell surface display.
azo1653 AtD (putative) - organism Azoarcus sp. (strain BH72)
This protein contains one autotransporter domain of the AT-1 family.
OprF AtD - organism Pseudomonas fluorescens
This outermembrane protein is thought to form an 8-stranded beta barrel in the outermembrane.
Cl02365 AtD (putative) - organism Neisseria meningitidis
This protein contains one autotransporter domain of the AT-1 family.
VtaA11 AtD - organism Haemophilus parasuis
This protein contains an autotransporter domain of the AT-2 family.
Hag AtD - organism Moraxella catarrhalis
The protein is a 200kDa outermembrane oligomeric protein with beta barrel structure composing of 10 transmembrane beta helices.
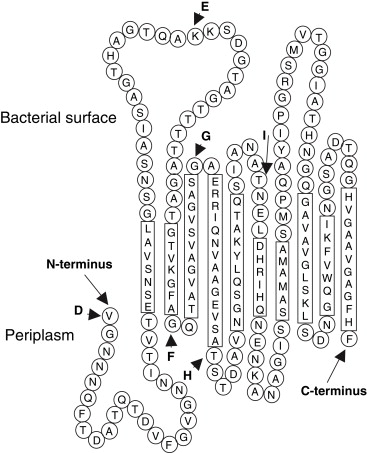
Pcryo_1225AtD - organism Psychrobacter cryohalolentis
Hia AtD - organism Haemophilus influenzae
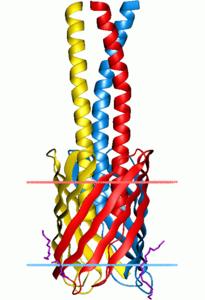
upaG_short - organism Escherichia Coli
espP(beta) - organism Escherichia coli
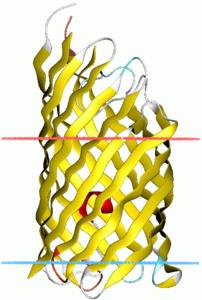
ehaB - organism Escherichia coli
TshA - organism Escherichia coli
subgroup1 of autotransporters
VirG(IcsA) - organism Shigella flexneri
YuaQ AtD - organism Escherichia coli
AIDA-I - organism Escherichia Coli
Ag43_short - organism Escherichia Coli MG1655
eCPX - circularly permuted OmpX(organism Escherichia Coli) This protein is an enhanced CPX variant that is located in the outermembrane that joins the N- and C-termini of OmpX.
CPG_L2 - circularly permuted OmpG(organism Escherichia Coli) This protein is circularly permuted with its backbone opening in loop 2.
CPG_L6 - circularly permuted OmpG(organism Escherichia Coli) This protein is circularly permuted with its backbone opening in loop 6.
Some of these proteins are putative autotransporters that have sequence homology to confirmed autotransporters. We chose these proteins because we wanted to test the functionality of these putative autotransporters and expand the range of displayers available for surface display.
 "
"