Team:Bologna/Project
From 2009.igem.org
| Line 41: | Line 41: | ||
|width=110| <font size="3">'''Suffix''' | |width=110| <font size="3">'''Suffix''' | ||
|- style="background:#99BADD; color:black; text-align: center" | |- style="background:#99BADD; color:black; text-align: center" | ||
| - | | <font size="2" color="#228b22"> | + | | <font size="2" color="#228b22">GAATTCGCGGCCGCTTCTAGAG |
| - | | <font size="2" color="#000080"> | + | | <font size="2" color="#000080">AACACAAACTATCACTTTAACAACACATTACATATACATTAAAATATTAC |
| - | | <font size="2" color="#FF6600"> | + | | <font size="2" color="#FF6600">AAAGAGGAGAAA |
| - | | <font size="2" color="#228b22"> | + | | <font size="2" color="#228b22">TACTAGTAGCGGCCGCTGCAG |
|} | |} | ||
<br> | <br> | ||
Revision as of 11:33, 21 October 2009
| HOME | TEAM | PROJECT | SOFTWARE | MODELING | WET LAB | PARTS | HUMAN PRACTICE | JUDGING CRITERIA |
|---|
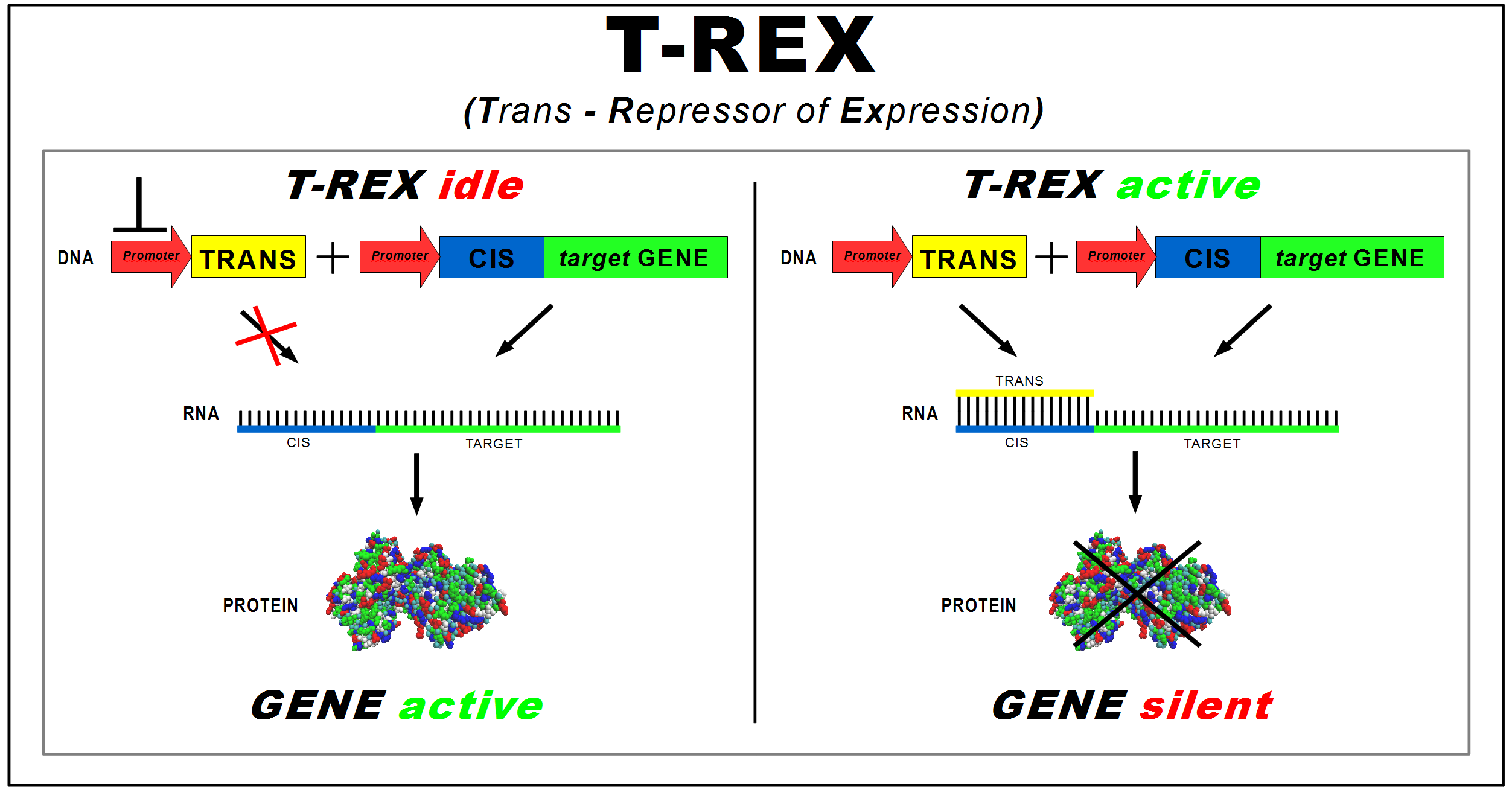
(Trans-Repressor of Expression)
The project aims to implement a protein synthesis regulation system in Escherichia coli that acts at translational level, regardless of the target gene. This "general-purpose" device allows a faster control of protein expression.
The device was named T-Rex (Trans Repressor of Expression). It consists of two new BioBricks:
- CIS-repressing, to be assembled upstream of the target protein coding sequence; it contains a ribosomal binding site; (RBS)
- TRANS-repressor, to be placed under the control of a different promoter; it is complementary to the CIS-repressing, and contains a RBS cover in two versions of different length (4 and 7 nucleotides).
The longer version covers also 3 nucleotides of the Shine-Dalgarno sequence.
Transcription of the target gene produces a mRNA strand, starting with the Cis element, which is translated into proteins by ribosome. Trans’ promoter induction produces a transcript that binds with the Cis part. The RNA duplex prevents ribosome from binding to RBS, repressing protein synthesis. Thus, the TRANS-repressor amount regulates the gene mRNA translation rate.
The Parts
We choose to use 50bp non-coding sequences as CIS-repressing and TRANS-repressor complementary parts, and we developed a bioinformatic tool (BASER) to design them, in order to minimize unwanted interactions with genomic mRNA, reduce the probability of intra-molecular annealing and of partial/shifted hybridization.
| CIS-repressing | |||
| Prefix | non-coding TRANS target | RBS | Suffix |
| GAATTCGCGGCCGCTTCTAGAG | AACACAAACTATCACTTTAACAACACATTACATATACATTAAAATATTAC | AAAGAGGAGAAA | TACTAGTAGCGGCCGCTGCAG |
| TRANS-repressor (4) | |||
| Prefix | Cover | non-coding TRANS | Suffix |
| TACTAGAG | CCTCTTT | GTAATATTTTAATGTATATGTAATGTGTTGTTAAAGTGATAGTTTGTGTT | TACTAGAG |
| TRANS-repressor (7) | |||
| Prefix | Cover | non-coding TRANS | Suffix |
| TACTAGAG | CTTT | GTAATATTTTAATGTATATGTAATGTGTTGTTAAAGTGATAGTTTGTGTT | TACTAGAG |
- CIS-repressing, to be assembled upstream of the target protein coding sequence; it contains a ribosomal binding site (RBS):
- TRANS-repressor, to be placed under the control of a different promoter; it is complementary to the CIS-repressing, and contains a RBS cover in two versions of different length (4 and 7 nucleotides):
5' Prefix - CCTCTTTGTAATATTTTAATGTATATGTAATGTGTTGTTAAAGTGATAGTTTGTGTT - 3' Suffix
5' Prefix - CCTCTTTGTAATATTTTAATGTATATGTAATGTGTTGTTAAAGTGATAGTTTGTGTT - 3' Suffix
The BioBricks
</html>
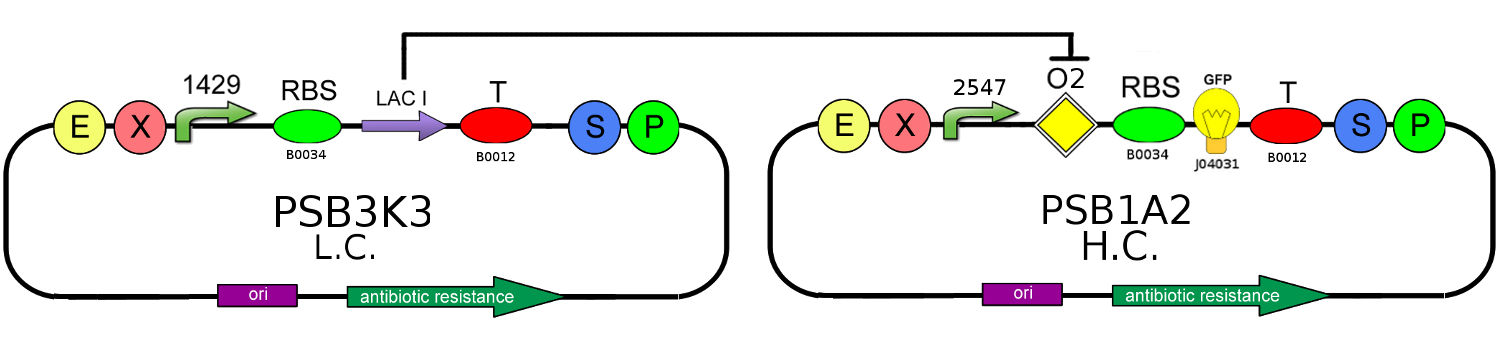
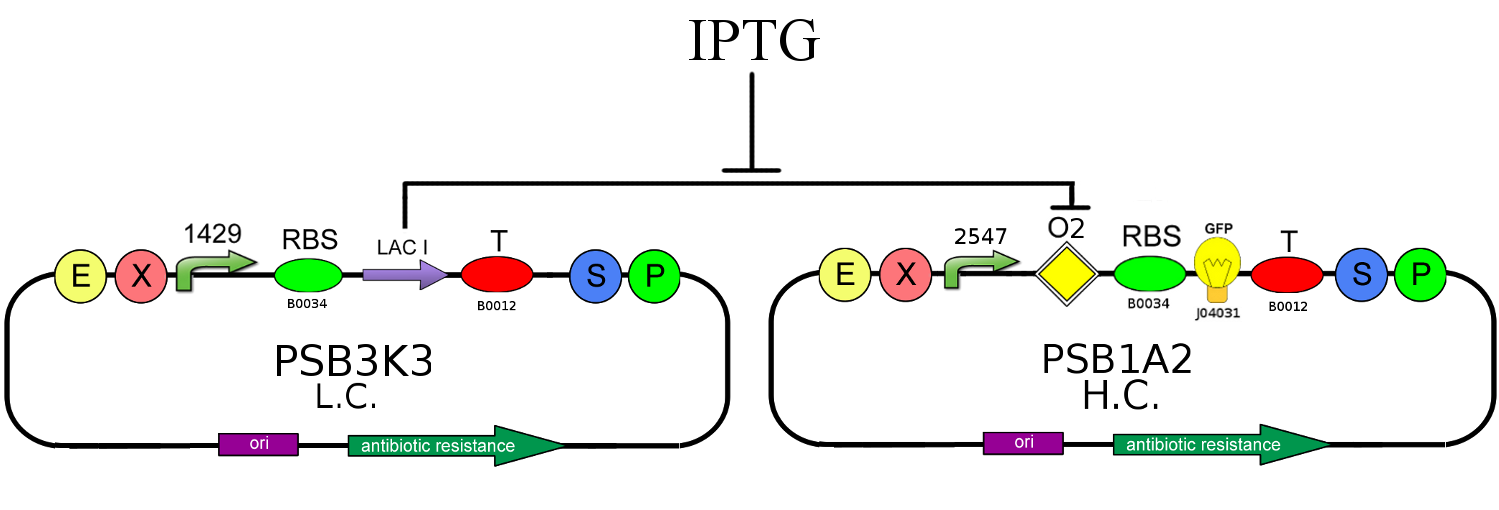
We decided to realize the above circuit, using Cis-repressing and Trans-repressor in order to verify the functionality of our project. The used plasmids are a low copy number one (pSB3K3) and an high copy number one (PSB1A2); the first contains Cis-repressing and the LAC I gene downstream of the promoter J23118, the second, instead, carries a BioBrick composed by the operator Ox (k079019) and the fluorescent protein GFP (J04031) under the control of J23100 promoter and Trans-repressor downstream of the same promoter.
This circuit represent the ON state: Trans-repressor is constitutively expressed, so it's mRNA binds Cis-repressing's one blocking the LAC I production. The absence of the LAC I protein allows the GFP production. In this configuration if you also add IPTG you can further strengthen the repression of the production of LAC I.
We realized also an OFF Configuration in which TRANS-repressor is absent: LAC I protein is produced, so it binds to the operator Ox blocking the GFP generation.
Before realizing the whole device we decided to perform some preliminary tests designed for characterize every part of the final device in order to obtain some information and values concerning the processes we were analyzing. All studies conducted by us can be found in detail in the Wetlab section.
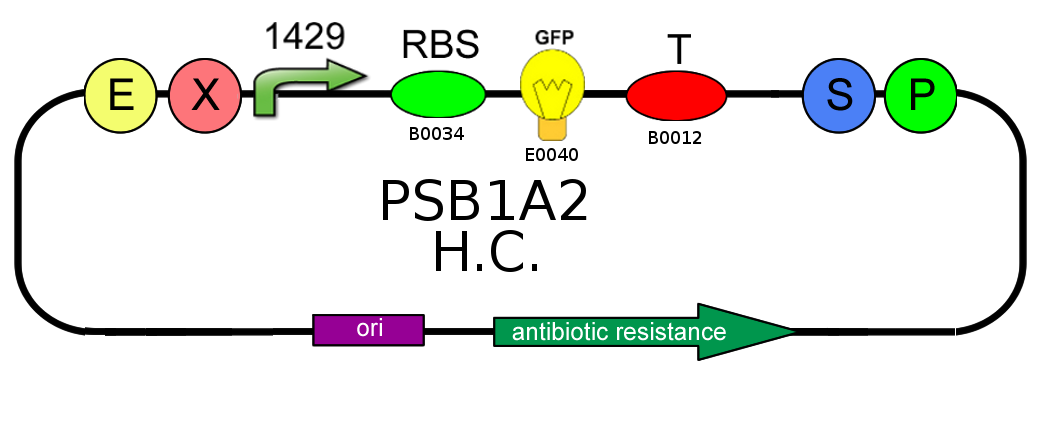
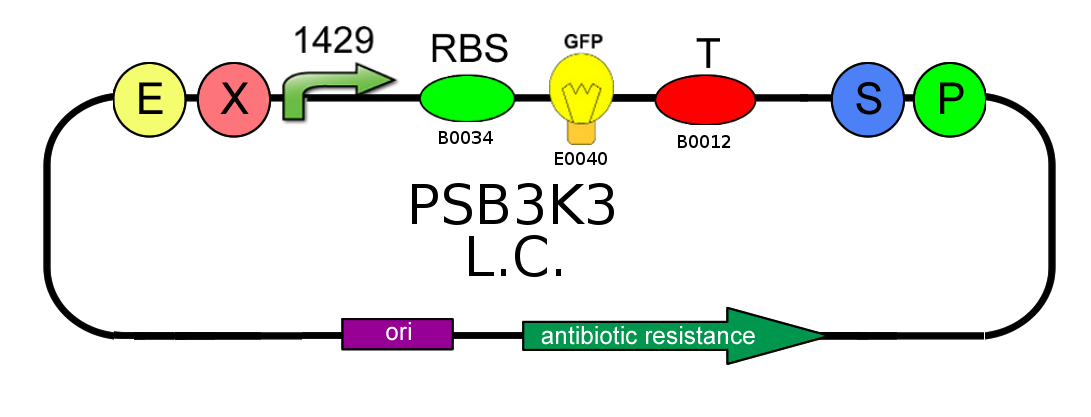
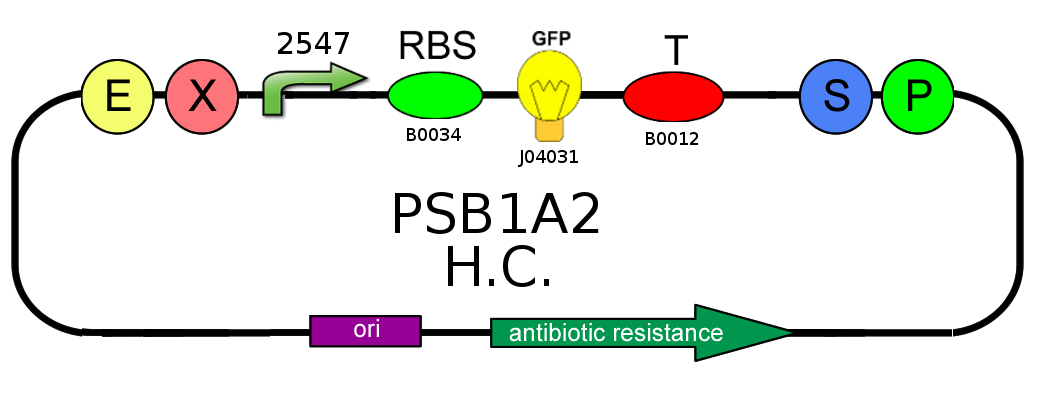
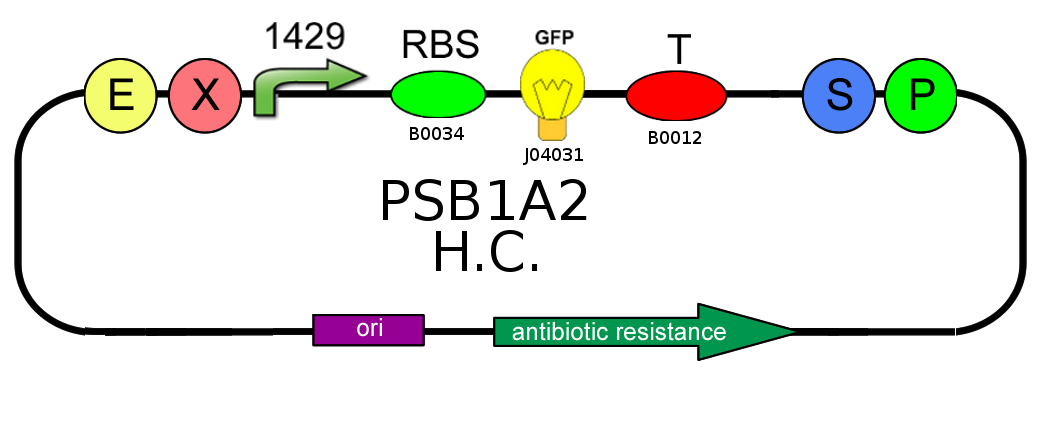
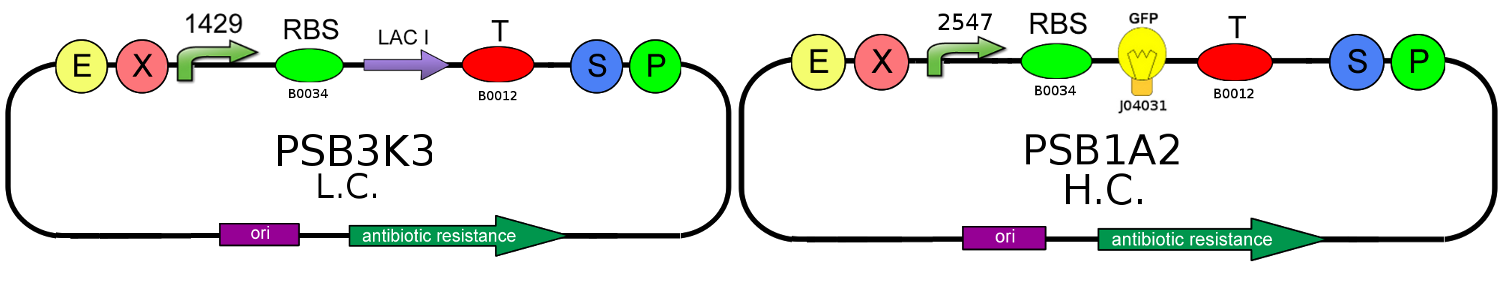
- We started with plasmid copy number characterization. To test the ratio between the production of an high copy number plasmid (PSB1A2) and a low copy number one (PSB3K3), we assembled two circuits. The open loop GFP (no tag) circuits are realized with a 1429 promotor and the standard biobrick I13504:
- Then we analyzed the difference in strength of the two promoters J23100 (2547) and J23118 (1429) studying the ratio of fluorescence. To do this we created these two circuits where the only different element was the promoter. Our goal was to find a ratio of fluorescence between the two circuits as close as possible to 1.78 (ratio between 2547 and 1429).
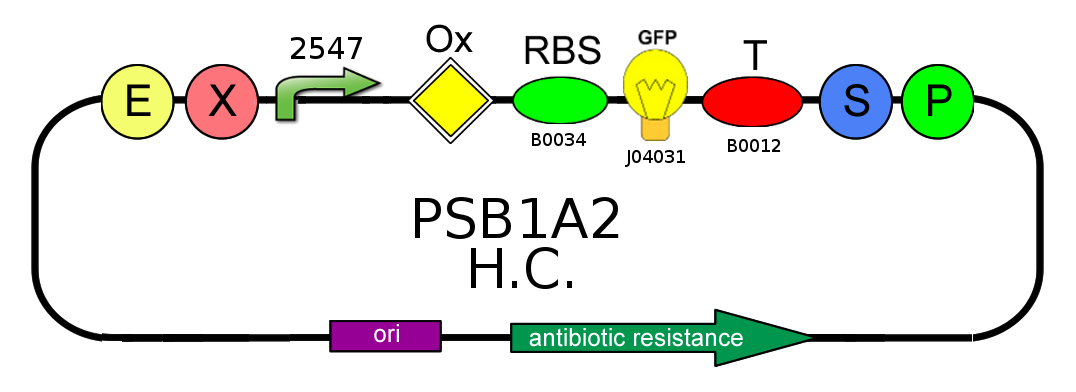
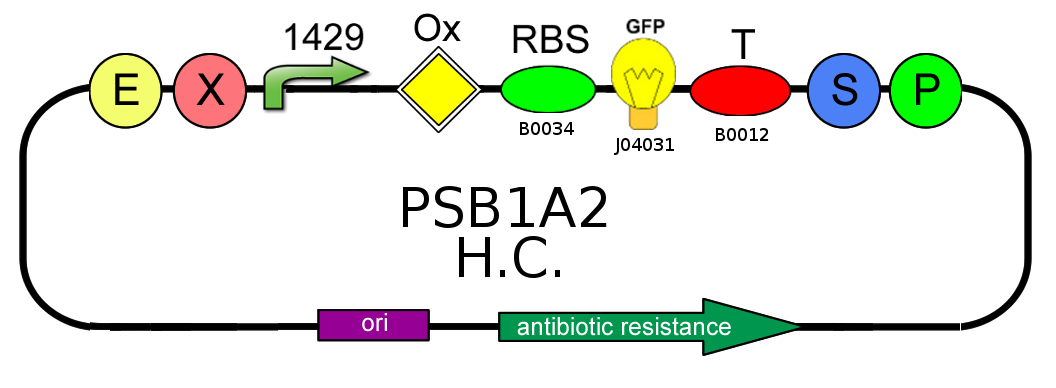
- After that we needed to know if the presence of the Ox operator could influence or not the production of GFP. We tested two open loop GFP circuits for each promoter, one with the operator Ox and another without. We wanted to obtain the same GFP production in the two circuits built with the promoter 2547 and in the two circuits built with the 1429 one.
- We started to assemble circuits with the simultaneous presence of GFP and LACI to assess their operation and their interaction with each other. The first one is a closed-loop configuration where GFP expression is auto-regulated by the synthesis of LACI repressor protein; we used different IPTG induction concentration to evaluate the LACI repression strength. The second is an open-loop configuration lacking the operator site, so we can determine if there is repression in absence of Ox operator.
 "
"